Attribute Monitoring Workflows [Infographic]
Learn how the BioAccord LC-MS System is instrumental in the drug development workflow from sample prep to reporting.
![Attribute Monitoring Workflows [Infographic]](https://www.waters.com/blog/wp-content/uploads/feature-image-768x758.png)
Learn how the BioAccord LC-MS System is instrumental in the drug development workflow from sample prep to reporting.

Combining BioAccord, our most capable MS detection platform, with ACQUITY Premier UPLC, our highest performance LC system, to demonstrate superior performance.

The development of biotherapeutics continues to accelerate with their potential to revolutionize disease treatment in areas not adequately addressed by small molecule therapeutics. However, the complexity of these macromolecules and the range of potential modification variants (attributes) creates an interesting analytical challenge for scientists tasked with their characterization and then with monitoring those attributes important…

To ensure compliance success in a changing regulatory landscape, laboratories must continually adapt. Learn how the compliant-ready Waters BioAccord LC-MS platform helps biopharma address the additional compliance challenges posed by the field’s complex analytical instruments.

Oligonucleotides, or short strands of DNA or RNA, continue to increase in popularity. In addition to their essential role as primers and probes in the expanding clinical DNA testing and diagnostics market, they’re also seeing great success as new gene-based therapeutics that hold tremendous promise. With a string of recent drug approvals, including the first…
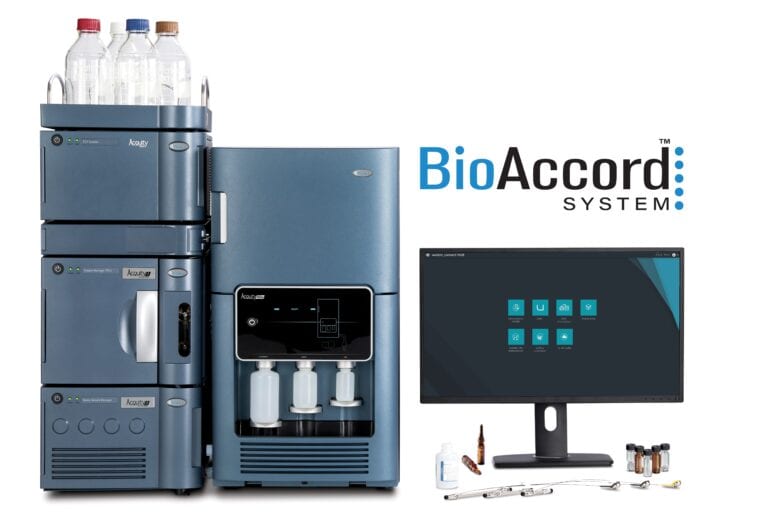
As scientific advances continue to carry researchers into new arenas, it is vital that technology evolves in tandem. At Waters, we understand that it is only by leveraging fit-for-purpose technologies that researchers are able to unleash their creativity and make pioneering scientific breakthroughs. Scientists working within the rapidly growing biopharmaceutical industry are no exception. With…

What does “intelligence” mean in the context of biopharma analysis? Given the complex setting, much of the answer comes down to the task a user needs to complete, along with the question of who exactly that user is. In developing our BioAccord System, we wanted to expand the possibilities of high-resolution LC-MS analysis from characterization…

In late January, I traveled to WCBP (Well Characterized Biological Products), an industry conference that is hosted every year by the CASSS organization. It serves as a forum that brings together biopharmaceutical industry professionals and various regulatory agencies from around the world to review and discuss the current and future state of biopharmaceutical drug development….
Analytical data can be so much more than a historical point in time documented in a single report. In fact, biopharmaceutical organizations can use LC-MS data to build a continuum of compliance.

For biopharmaceutical organizations working with contract labs, it’s important to consider how data and methods will be transferred and how generated data will be managed so that it meets data integrity and compliance requirements.
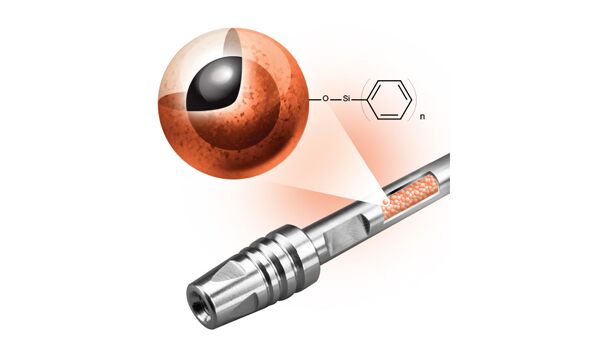
We listened and learned how scientists separate mAbs and ADCs; then we designed a novel column for LC-MS bioseparations A critical step toward the prolific and successful use of monoclonal antibodies (mAb) as biotherapeutics occurred in 1988 when techniques were introduced to humanize these biomolecules, eliminating or reducing the deleterious patient side effects that previous…

Analytical methods transfer. Data integrity. A changing and more stringent regulatory landscape. All this, and more, impacts productivity in upstream and downstream biopharmaceutical processes. How is Waters looking to help address development challenges for biologics and biosimilars?
ACQUITY QDa (17) bioanalysis (12) biologics (17) biopharma (49) biopharmaceutical (58) biotherapeutics (21) case study (18) chromatography (14) compliance (12) data integrity (23) food analysis (12) HPLC (15) LC-MS (26) liquid chromatography (LC) (24) mass detection (16) mass spectrometry (MS) (58) method development (13) particle analysis (19)