Tools for Chromatographic Success: Three Ways to Support Data Quality in Routine Analysis
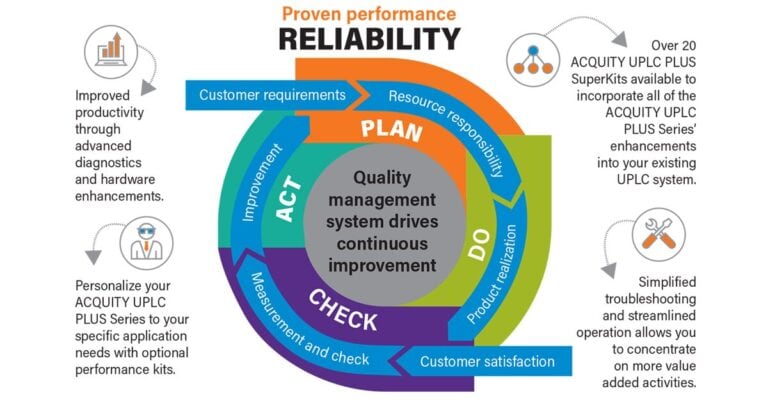
Avoiding the pitfalls of data quality and integrity in the Pharma QC laboratory while running routine analyses is an ongoing challenge. Regulatory violations are frequently a result of a lack of complete data, suspect data, and improper audit trails. A recent report from Deloitte stated that data integrity violations account for over 70% of the…