Embracing Analytical Techniques Into Regulated Laboratories
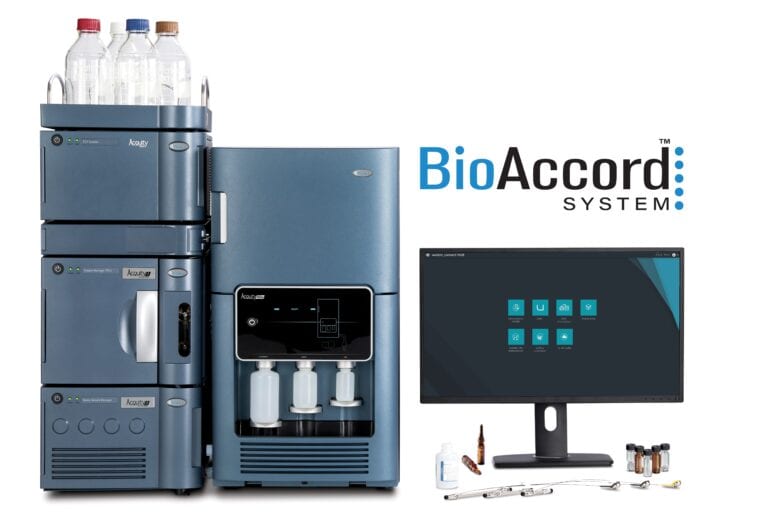
To ensure compliance success in a changing regulatory landscape, laboratories must continually adapt. Learn how the compliant-ready Waters BioAccord LC-MS platform helps biopharma address the additional compliance challenges posed by the field’s complex analytical instruments.