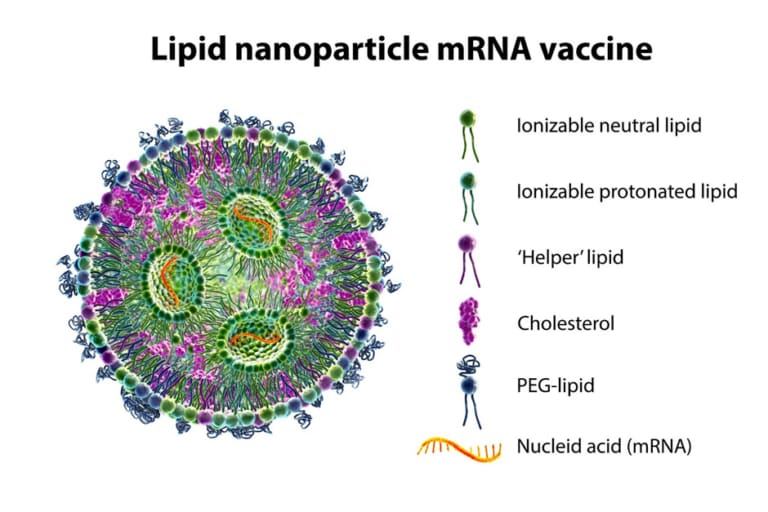
USP<787>: Tailored Guidance for Better Biologics
Understanding US<787> and its Impact on Pharmaceutical Quality with Tailored Guidance for Biologics In the world of pharmaceuticals, quality is paramount. Ensuring the safety and efficacy of medications is not only a regulatory requirement but also a moral obligation to patients. To achieve this, regulatory bodies like the United States Pharmacopeia (USP) lay down guidelines…