Exploring Cell Imaging Techniques – Types, Analysis, and Applications
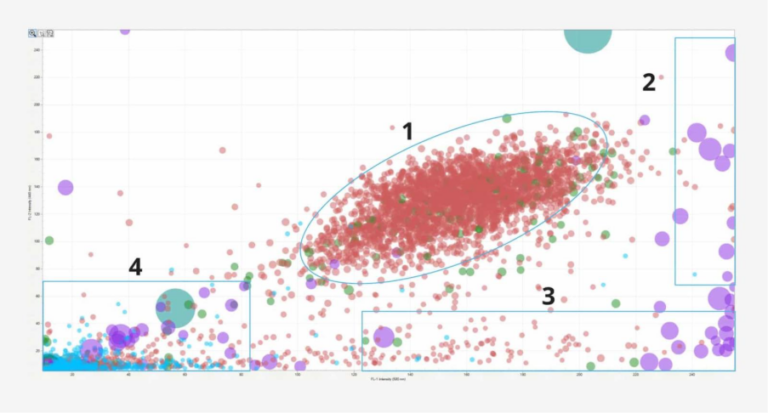
Understanding cellular functions like migration, morphology, and physiological changes in healthy and diseased cells is important for grasping biological processes and using them to optimize the creation of biological therapeutics. Live-cell imaging provides an instant picture of a cell, tissue, or organism’s current state, as close to in vivo as possible, without introducing artifacts seen…