The objective of this application is to demonstrate the use of the ACQUITY QDa Detector as a complementary orthogonal detection technique that enables the integration of optical (UV) and basic mass spectral data within a single workflow using a known peptide mixture.
For established biotherapeutic drugs in the pharmaceutical industry, screening protocols are often employed at different stages during the manufacturing process to ensure regulatory guidelines are met with regards to product efficacy, identity, and purity. Improved productivity and lower development costs are often associated with the implementation of targeted analytical protocols to assess critical quality attributes (CQA). Orthogonal techniques that add value and can be employed with minimal cost and effort without compromising productivity are highly desirable.
The ACQUITY QDa Detector is Waters’ solution to meet the demands of today’s fast-paced pharmaceutical work environment by enabling complementary optical and mass spec analysis techniques to be run on-line in a single screening workflow for improved productivity in data analysis.
The ACQUITY QDa Detector offers the ability to combine straightforward mass spectral data with optical data in a single workflow with minimal effort and cost. The ACQUITY QDa is pre-optimized to work without the need for sample-specific user adjustments that are typical of traditional mass spectrometers, making it simple and easy to use. With a purposeful design that allows for integration into existing UPLC instrumentation (Figure 1), the ACQUITY QDa Detector is well-suited for improving productivity and strengthening quality assurance in the biotherapeutic production environment.

The objective of this application is to demonstrate the use of the ACQUITY QDa Detector as a complementary orthogonal detection technique that enables the integration of optical (UV) and basic mass spectral data within a single workflow using a known peptide mixture.
The ACQUITY UPLC Peptide CSH Column (130Å, C18, 1.7 μm, 2.1 x 100 mm) 1.7 μm) was conditioned prior to use per the care and use manual. Chemical reagents were purchased from Sigma Aldrich and used as received. A peptide mix containing angiotensin (frag. 1-7), bradykinin, angiotensin II, angiotensin I, renin substrate, enolase T37, and melittin was prepared at a concentration of 0.3 mg/mL in initial mobile phase conditions.
|
LC system: |
ACQUITY UPLC H-Class |
|
Vials: |
Total Recovery vial: 12 x 32 mm glass, screw neck, cap, nonslit (p/n 600000750cv) |
|
Column: |
ACQUITY UPLC Peptide CSH 130Å, C18, 1.7 μm, 2.1 x 100 mm (p/n 186006937) |
|
Column temp.: |
60 °C |
|
Sample temp.: |
4 °C |
|
Injection vol.: |
10 μL |
|
UV detector: |
ACQUITY UPLC TUV |
|
Wavelength: |
215 nm |
|
MS detector: |
ACQUITY QDa |
|
MS sample rate: |
2 points/sec |
|
Mass range: |
200 to 1250 Da |
|
Cone voltage: |
7 V |
|
Capillary voltage: |
0.5 kV |
|
Probe temp.: |
600 °C |
|
Mobile phase A: |
H2O, 0.1 % TFA |
|
Mobile phase B: |
Acetonitrile, 0.1 % TFA |
|
Mobile phase C: |
H2O, 0.1 % FA |
|
Mobile phase D: |
Acetonitrile, 0.1 % FA |
|
Time |
Flow (mL/min) |
%A |
%B |
%C |
%D |
|---|---|---|---|---|---|
|
Initial |
0.300 |
19 |
1 |
76 |
4 |
|
2.00 |
0.300 |
19 |
1 |
76 |
4 |
|
22.00 |
0.300 |
11 |
9 |
44 |
36 |
|
25.00 |
0.300 |
3 |
17 |
12 |
68 |
|
28.00 |
0.300 |
3 |
17 |
12 |
68 |
|
28.01 |
0.300 |
19 |
1 |
76 |
4 |
|
30.00 |
0.300 |
19 |
1 |
76 |
4 |
Empower 3 Chromatography Data Software, SR2 with Mass Analysis
Proteolytic digests are often analyzed with optical detectors as a monitoring technique for protein composition of biotherapeutic drugs. The volatility and acidic nature of the mobile phases used in conventional peptide analyses makes the technique amenable to mass spectrometry. Lower m/z values are often observed for peptides because they are often multiply charged during the ionization process, particularly when low TFA or TFA-free mobile phases are employed. With a detection range between 30–1250 m/z, the ACQUITY QDa Detector is well-suited for providing complementary mass data in peptide analyses for multiply charged species.
To demonstrate this, a peptide mixture containing seven peptides was prepared at a concentration of 0.3 mg/mL. With an ACQUITY QDa on-line as a mass detector post optical detection, 3 μg of the peptide mixture was injected onto an ACQUITY UPLC Peptide CSH 130 Å C18 Column. A 20-minute reversed-phase gradient was used to elute the peptides (see Experimental).
As shown in Figure 2A, the ACQUITY QDa Detector provides sufficient sensitivity to clearly detect the individual peptides. Extracted MS spectra (Figure 2A inset) of the detected peaks can be examined in Purity, Combined, or Apex mode (shown) using Empower 3 Software. The complementary mass data are seamlessly integrated into the optical data as shown in Figure 2B, where the mass from the most intense base peak ion is displayed next to the retention time of the identified peak.
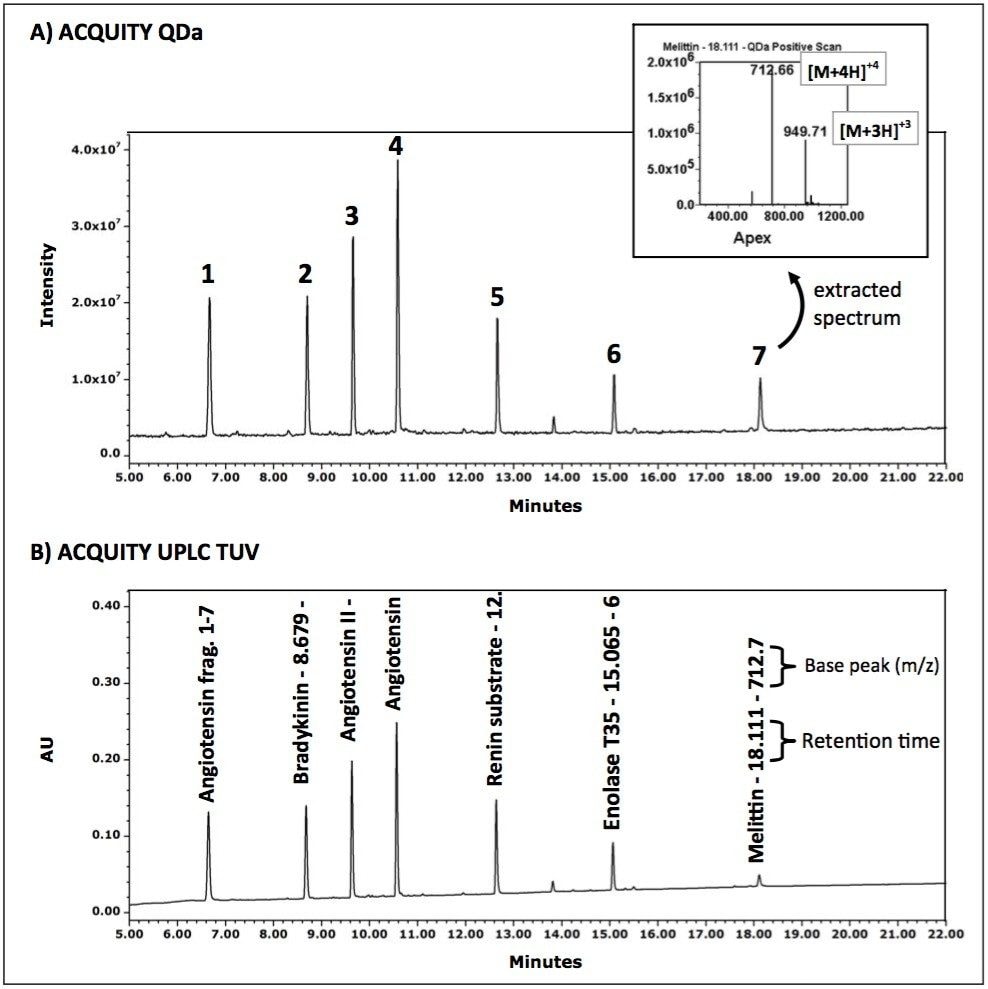
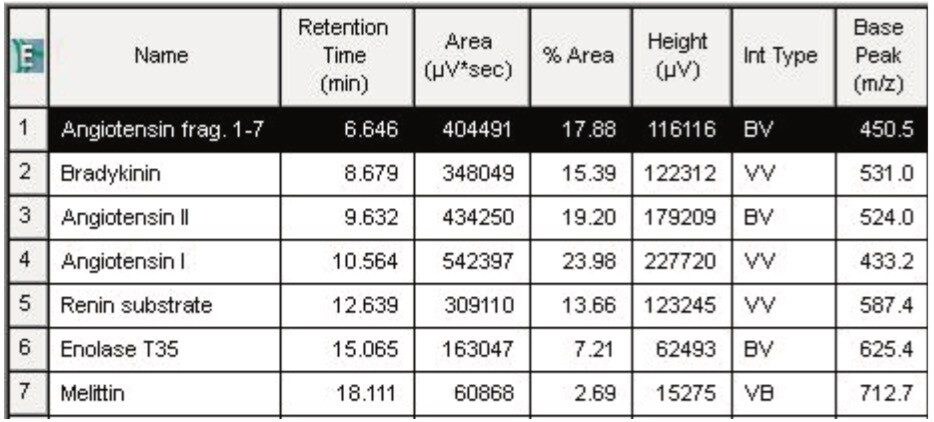
Utilizing Empower’s integrated informatics tools such as the Component Manager, optical peaks are automatically identified and labeled with their corresponding peptide name within the chromatogram (Figure 2B) as well as in tabular format (Table 1) for efficient data review.
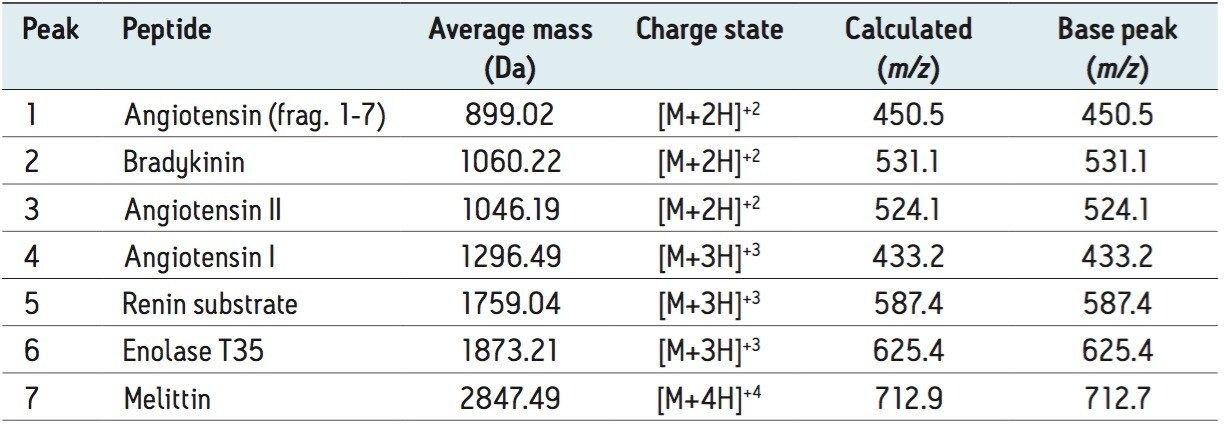
The base peak ion information reported by Empower 3 Software was compared to the calculated m/z for increasing charge states of the peptides using the average mass. As shown in Table 2, the ACQUITY QDa Detector readily detected peptides ranging in mass from 900 – 2800 Da. The base peaks detected corresponded to the peptides at charge states ranging from [M+2H]+2H – [M+4H]+4H. Accuracy of the detector response was tested over three injections and found to be within the instrument specification of ± 0.2 Da.
The performance of the ACQUITY QDa Detector as part of the ACQUITY UPLC H-Class System, combined with Empower 3 Software’s ability to seamlessly integrate mass spectral data with optical data, makes the ACQUITY QDa an ideal solution as an on-line complementary orthogonal detection technique for improving productivity in screening protocols for well characterized biopharmaceutical drugs.
The high costs associated with meeting regulatory guidelines for biotherapeutic drug safety has renewed interest in efficient methods that reduce costs and increase productivity in the manufacturing process. The ACQUITY QDa Detector, used with an ACQUITY UPLC H-Class System, offers the ability to employ mass spec analysis as an orthogonal detection technique in a single workflow, greatly improving monitoring capabilities in the production environment with minimal cost and effort.
With a plug-and-play design, and integrated compliant data acquisition and analysis through Empower 3 Software, the ACQUITY QDa Detector is a powerful tool that can increase productivity and confidence in data analysis within the biotherapeutic production environment.
720005131, August 2014