This is an Application Brief and does not contain a detailed Experimental section.
To demonstrate the value of an intact murine monoclonal antibody standard as both a generic internal standard and as a workflow check/system health standard in protein bioanalysis.
Protein quantification workflows are complex and replete with challenges, not the least of which are choice of internal standard and the ability to demonstrate analyst capability. While ligand binding assays (LBAs) have long been the gold standard for large molecule quantification, LC-MS has risen to the top as a technique of choice due to its improved specificity, improved accuracy and precision, and elimination of long development timelines for often poorly standardized biological reagents. In spite of its obvious advantages, LC-MS for protein bioanalysis is relatively new and is often carried out by scientists with traditional small molecule training. For this reason, we have developed a murine monoclonal antibody for use as both a generic internal standard for discovery studies involving antibody-based drugs as well as for use in evaluating staff proficiency with these new workflows.
For those small molecule bioanalytical scientists now faced with performing bioanalysis studies on proteins, the task may be a difficult one. One of the first challenges encountered is the choice of internal standard (IS). While small molecule workflows have become commonplace and are simple and straightforward, protein quantification involves many unfamiliar processes such as digestion and affinity purification. In order to monitor the efficiency of these steps, an appropriate internal standard must be used. Options such as labeled and/or extended tag surrogate/signature peptides are not effective in compensating for or identifying changes in digestion efficiency or upfront, protein-level pull-down. The optimal IS for protein quantification via the surrogate peptide approach would naturally be a protein. While labeled antibody IS’s exist such as SILu mAb, they can be prohibitively expensive.
In addition, protein bioanalysis workflows not only contain many segments and multiple steps within each segment, but a significant fraction of the workflow is comprised of techniques which are new to typical small molecule bioanalysts. It is the unfamiliar nature of the steps, coupled with the preparation of numerous specialty (some biological) reagents that creates opportunities for error, imprecision, and poor reproducibility. A well characterized workflow test standard with an accompanying “kit” and protocol recipe would be an ideal way to not only learn and validate the process, but also to demonstrate competence.
We have developed and verified an intact murine (mouse) antibody for use as both a generic internal standard and as a tool, in conjunction with ProteinWorks eXpress Digest Kits, to both learn and then test aptitude in protein bioanalysis workflows. This standard can also be used on a periodic basis to confirm system readiness/health for these same workflows. The murine antibody is an ideal internal standard for antibody quantification via the surrogate peptide approach as it is added to the sample prior to any type of sample preparation and, subsequently, efficiently and effectively compensates for changes and or sample spillage throughout every step of these complex multi-stage workflows. This includes its ability to adjust for efficiency, recovery, or other changes in the affinity purification (if used) of the protein drug from plasma. It is also ideally suited to compensate for changes in digestion efficiency or volume as it is essentially an analog of any antibody-based drug, thus making it a suitable generic IS for discovery studies. For use as an IS, we recommend a final concentration in plasma of 20–50 μg/mL.
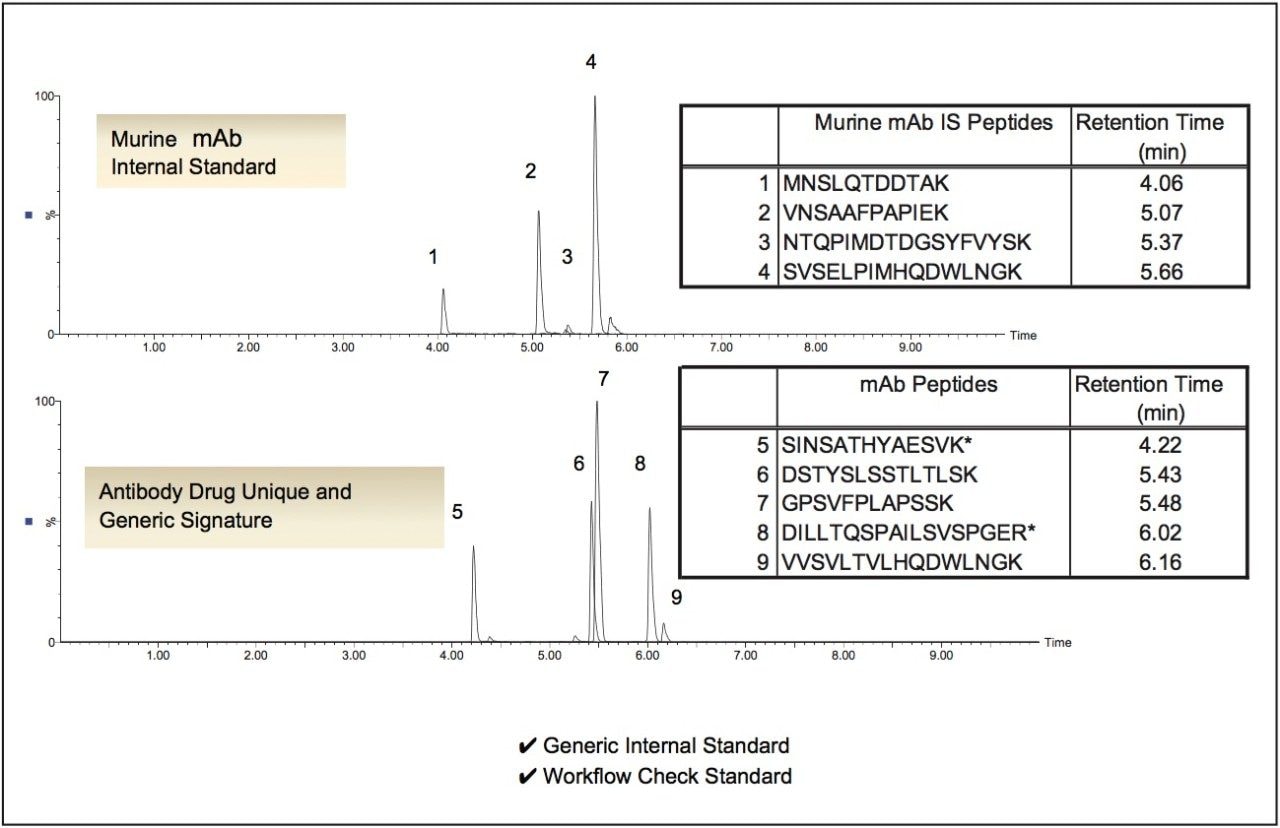
In order to broaden its utility, we have identified several signature peptides from this murine mAb. Depending on the signature peptide used, one can obtain specificity in human, rat, or other species. For example, while the SVSELPIMHQDWLNGK and VNSAAFPAPIEK peptides from the heavy chain are specific in rat, human and mouse plasma, the MNSLQTDDTAK peptide is specific in human and mouse, but not rat plasma. The final peptide, NTQPIMDTDGSYFVYSK , is also specific in human and mouse plasma, but not necessarily in rat. MRM and chromatographic test conditions are provided in order to confirm and monitor the workflow performance. The sequence and available signature peptides from the intact murine mAb IS/check standard are shown in Figure 1. When the murine mAb is used with and according to the directions of the ProteinWorks eXpress Digest Kits, its signature peptides elute in the same gradient window as both unique and generic signature peptides from humanized monoclonal antibody type drugs. For example, Figure 2 demonstrates the elution profile of the murine peptides as compared to multiple representative unique and generic signature peptides from mAb therapeutics such as trastuzumab, infliximab, adalimumab, and bevacizumab, following the recommended chromatographic conditions from the ProteinWorks eXpress Digest Kit. Furthermore, as an internal standard, its performance enabled single digit accuracy and precision and a quantification limit of 10 ng/mL for infliximab in Waters Application Note 720005535.
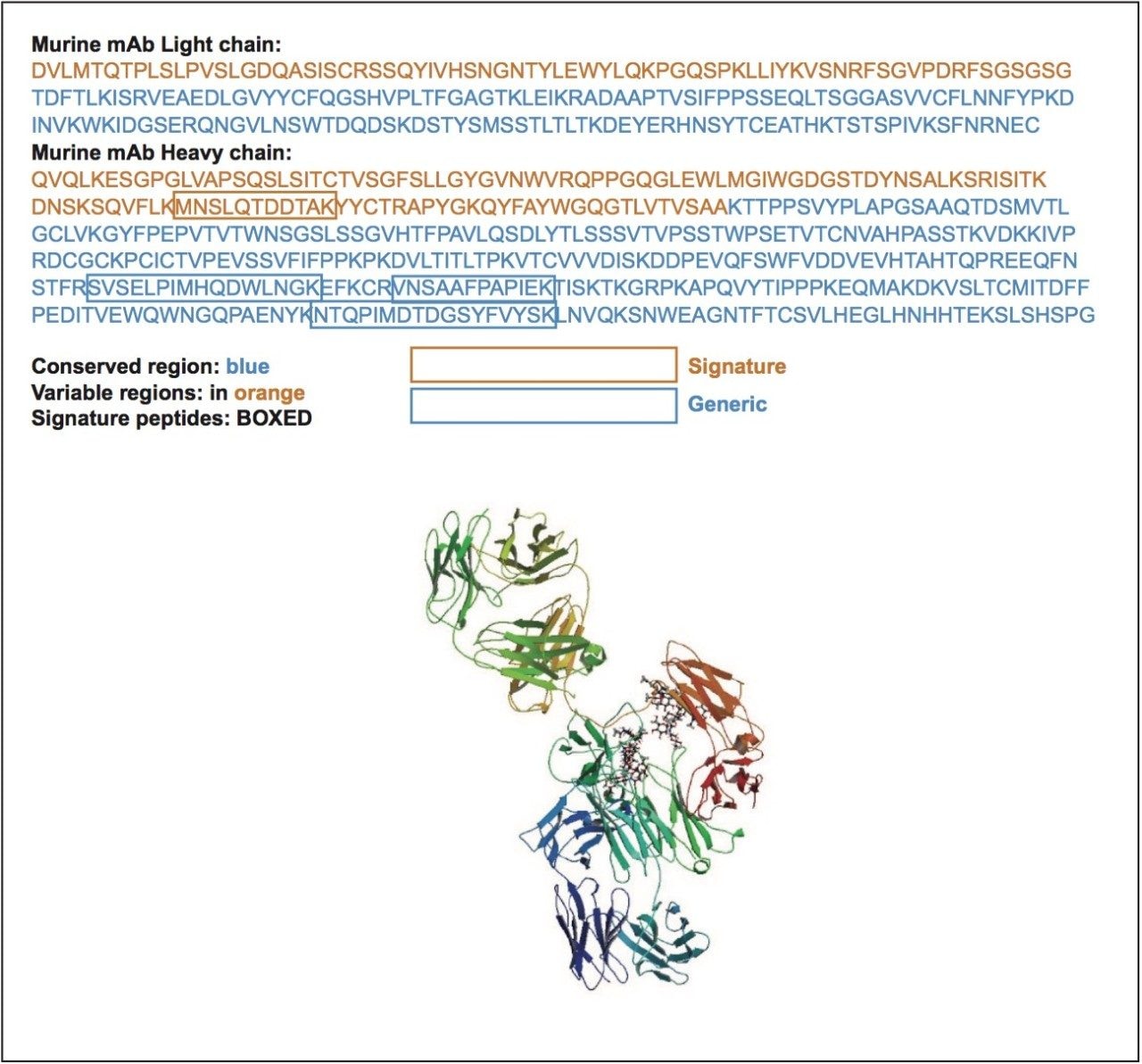
An intact murine (mouse) antibody has been developed and is available for use in protein bioanalysis workflows as either a generic IS or as a workflow test/system health standard, in combination with the specified MRM and LC conditions. In conjunction with a universal, proven protocol and the pre-measured, lot-traceable reagents in ProteinWorks eXpress Digest Kits, one has a recipe which simplifies protein bioanalysis and enables novice users to quickly generate high quality data in discovery studies. In addition, this generic commercially available antibody can be used to evaluate analyst proficiency and to monitor LC-MS system performance.
720005543, December 2015