Bioconjugates are increasingly central to next-generation therapeutics, enabling the precise delivery of potent payloads through targeted molecular design. Optimizing complex, heterogeneous molecules requires robust analytical technologies to fully characterize critical attributes such as conjugation sites, payload distribution, and overall molecular heterogeneity.
As bioconjugated molecules advance from discovery to commercialization, their structural diversity and complexity present significant analytical challenges. Comprehensive characterization is essential to ensure safety, efficacy, and consistent product quality.
Waters provides fit-for-purpose solutions to deliver deep insight into bioconjugate structure and quality. Simplify your complex molecule analysis and accelerate the characterization and monitoring of critical quality attributes, regardless of your bioconjugate structure.





Case Study: How Waters is Helping to Support the Development of Novel Therapies

Accelerate the journey from sample analysis to decision making using waters_connect for biopharmaceuticals, with tailored workflows designed for bioconjugate analysis with integrated, automated calculation of conjugation ratio and distribution
Achieve high-quality LC-based analyses and determine DAR of cysteine-conjugated and highly hydrophobic ADCs using Waters Protein-Pak Hi Res Hydrophobic Interaction Chromatography (HIC) HPLC Columns for fast, efficient separations.
Reversed-Phase Columns for bio-conjugates and Antibody Drug Conjugates with MaxPeak HPS such as the ACQUITY and XBridge Premier Protein BEH C4 Columns are uniquely designed to minimize secondary interactions between ADCs and column hardware and suitable for the analysis of lysine-conjugated ADCs. Unlike cysteine-conjugated ADCs, the intra-chain disulfide bonds that maintain linkages between the heavy and light chains of the mAb for the lysine-conjugated ADCs are intact. Therefore, reversed-phase chromatography is suitable for the analysis of lysine-conjugated ADCs when linker chemistry is not labile at acidic pH. In addition, Waters offers Reversed-Phase Columns with solid-core particle technology, such as BioResolve RP mAb Polyphenyl for intact and subunit analysis to determine the localization of linked drugs at the domain level.
Eliminate the need to create multiple SEC eluent mobile phases with different proteins by using a platform method with phosphate buffered saline (i.e., PBS) for consistently accurate aggregate, monomer, and fragmented data with Waters SEC Columns. In addition, for bio-conjugates which can be hydrophobic creating unwanted secondary interactions, there is no need to add organic solvent with MaxPeak Premier Protein SEC Columns and it’s PEO bonded BEH particles.
Accurately measure charge variant profiles of cysteine-linked and lysine-linked ADCs with the BioResolve SCX mAb Column and BioResolve CX pH Buffer Concentrates for salt or pH gradients with minimal secondary interactions in optical detection. For structural insights with mass spectrometry in ion exchange, our MS-compatible buffers, IonHance CX-MS pH Buffer Concentrates, make online IEX-MS a reality with high-purity, volatile buffers suitable for mass spectrometry.
Antibody oligonucleotide conjugates (AOCs) are commonly used in protein diagnostics and are a promising therapeutic for targeted cell therapy. The net negative charge of an AOC expands with each additional conjugation site, resulting in increased retention on an anion exchange resin. The GenPak FAX Column contains a weak anion exchange sorbent that exhibits controlled retentivity and a unique selectivity for protein and oligonucleotide samples. Recent application work demonstrates that AEX can be coupled with MALS to generate absolute molecular weight information.
Hydrophilic Interaction Chromatography (HILIC) has been widely used to separate small polar compounds, yet its application to large biomolecules, other than released glycans, has been surprisingly limited. With Waters widepore Glycoprotein Columns, it is now possible to use HILIC to glean previously unattainable information from intact proteins (with or without glycosylation), protein fragments, and complex, released glycans.
Waters PeptideWorks Tryptic Protein Digestion Kits for manual or automated workflows uniquely provide your lab with high efficiency, reproducible peptide maps in under 2.5 hours—4x faster than average home-brew methods. Achieve 30-minute digestions with a 1:5 enzyme:peptide ratio, 78% reduction in missed cleavages, and a 98% reduction in contaminated autolysis peaks. PeptideWorks is ideal for drug conjugation site determination, protein structural characterization, protein identification, and protein modification monitoring including post-translation modifications.
Re-imagine a streamlined, fast, and highly sensitive way to profile N-glycans on ADCs with the GlycoWorks Kit. Achieve unprecedented fluorescent and mass spectrometric performance for released n-glycan analysis while maximizing productivity with a solution that is automatable on Waters Andrew+ Pipetting Robot. With just 3 easy steps in as little as 30 minutes, the GlycoWorks RapiFluor-MS N-Glycan Kit reduces complicated, time-consuming sample preparation.
Simplify and accelerate protein digestion with ProteinWorks eXpress Digest Kits. Pre-measured, lot-traceable reagents and standardized protocols for accurate, precise, and sensitive LC-MS quantification of proteins, including ADCs, in plasma and serum. Achieve unmatched performance and flexibility for reproducible protein digestion with kits optimized for various workflows, including protein bioanalysis/DMPK.
Accelerate antibody titer measurements with Waters BioResolve Protein A Affinity Columns featuring MaxPeak Premier Technology. These high-performance columns help you achieve up to 7x greater sensitivity, enabling faster decision-making and better process control. These columns also unlock advanced analytical workflows, including simplified Direct Connect 2D ProA‑SEC and ProA‑MS methods. Streamline your processes and gain unprecedented, in‑depth insights into your antibodies.
Streamline sample preparation for quantitative LC-MS analyses with verified workflows that help minimize variability, improve traceability, and simplify method transfer.
Optimize your laboratory's productivity and success with Waters Global Services to maintain peak system performance, minimize down time, address application challenges, and support stringent compliance requirements.
Maximize resources and minimize risk with payment options from Waters Capital, including upgrading aging equipment, getting customized support, and bundling services into one monthly payment.

Cysteine conjugated ADC analysis using HIC. Drug distribution was determined for three different cysteine-conjugated ADC samples with increasing drug load.
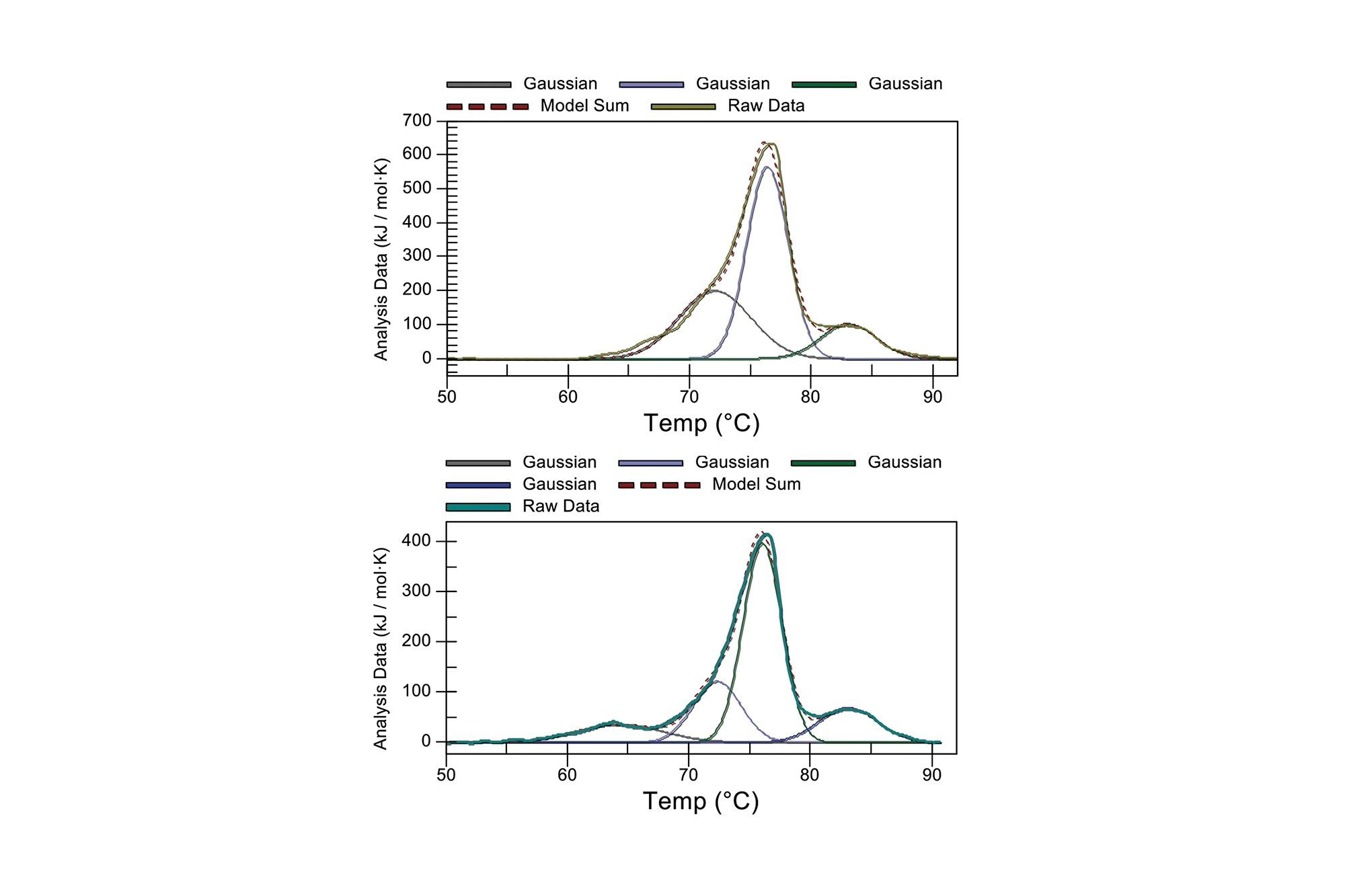
Gaussian-fitted DSC ADC results collected on the BioAccord System for native (top) and a high DAR mAb (bottom).
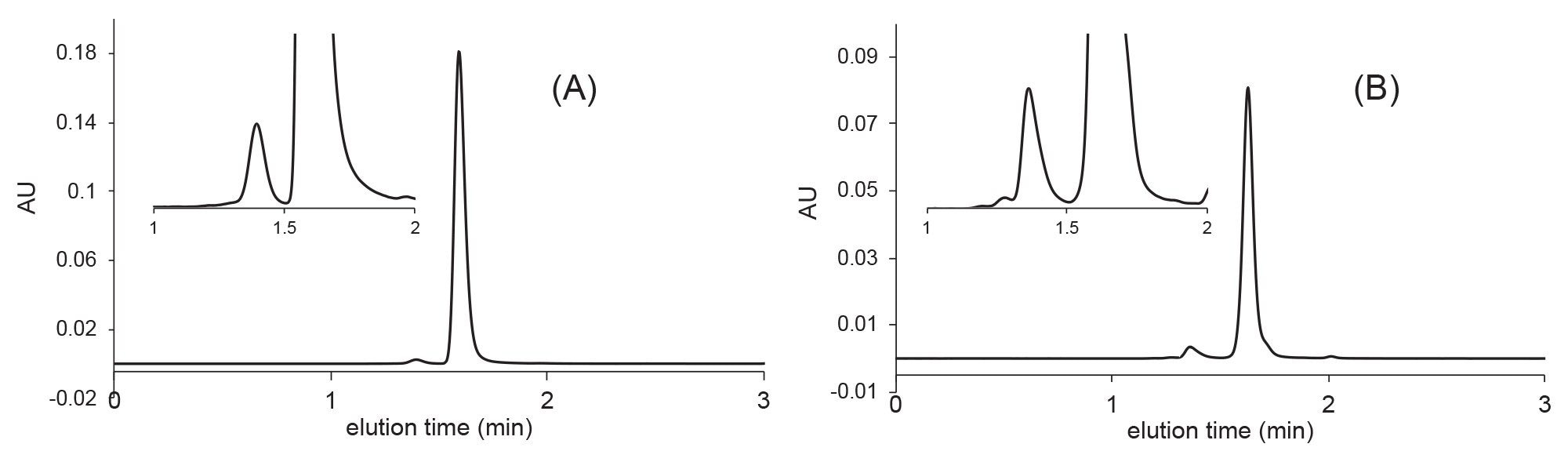
Fast SEC analysis of trastuzumab emtansine (A) and bevacizumab (B) as performed on an XBridge Premier Protein SEC 250 Å 2.5 µm 4.6 x 150 mm column. Mobile phase: 1x PBS, pH 7.4, flow rate F = 0.7 mL/min, temperature T = 25 °C, injected volume 1 μL, detection: 280 nm (UV).
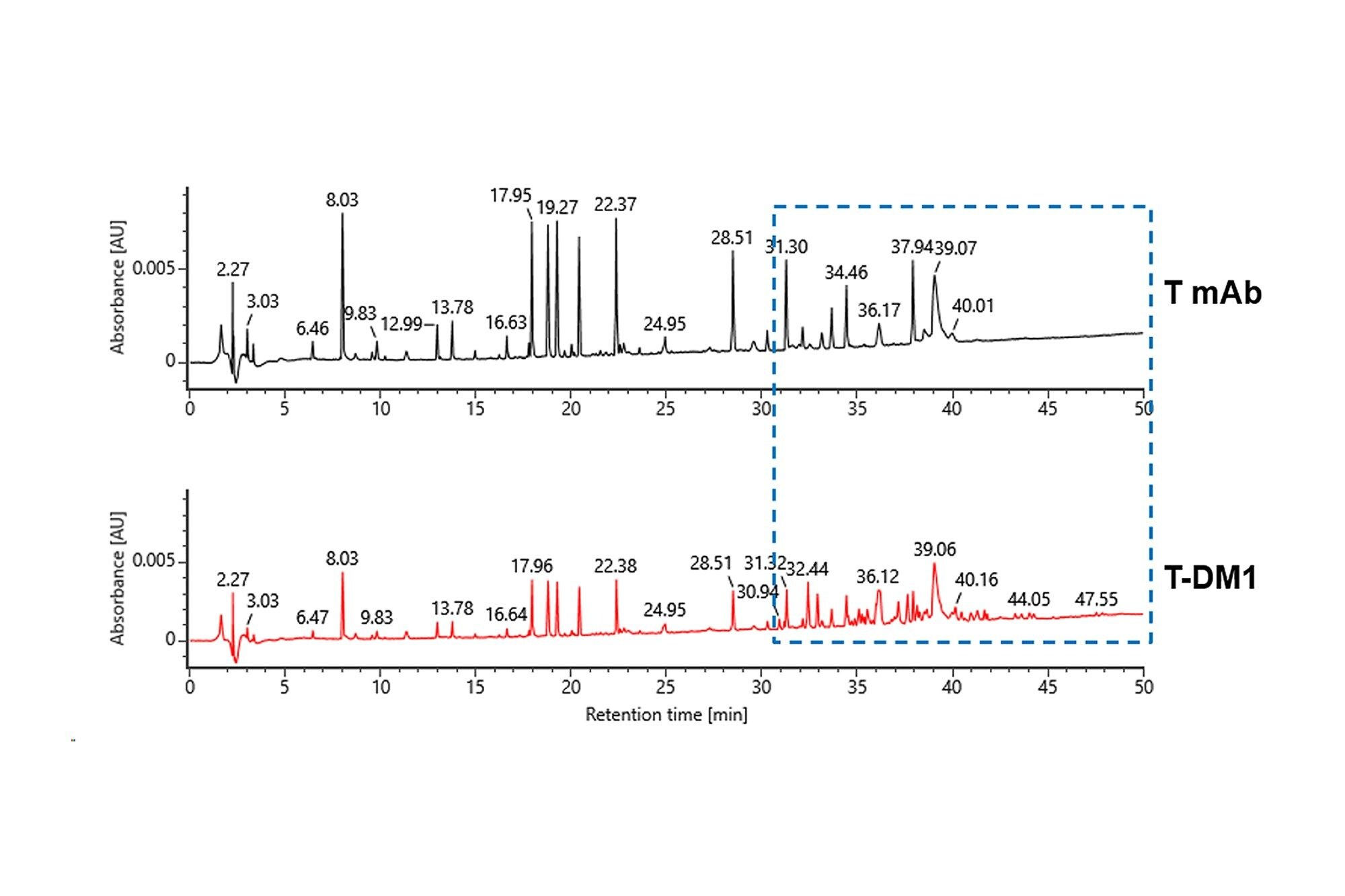
Peptide Mapping (UV chromatograms) comparison of a conjugated and unconjugated Trastuzumab
Top: Trastuzumab biosimilar tryptic digestion.
Bottom: Trastuzumab emtansine (Kadcyla), antibody drug conjugate tryptic digestion.
Both tryptic digestions utilized RapiZyme Trypsin and PeptideWorks Tryptic Protein Digestion Kit with 100 µL injection (~ 10 µg) on XSelect Premier Peptide CSH C18, 2.5 µm, 2.1 x 150 mm column. Additional peaks observed in Kadcyla chromatogram (bottom) compared to unconjugated trastuzumab (top).