For research use only. Not for use in diagnostic procedures.
This application note describes a clinical research method using protein precipitation of whole blood with internal standards. Chromatographic elution was completed within 1.8 minutes using a Waters ACQUITY UPLC HSS C18 SB Column on a Waters ACQUITY UPLC I-Class System followed by detection on a Xevo TQD Mass Spectrometer
Cyclosporine, everolimus, sirolimus, and tacrolimus are commonly used, powerful immunosuppressive agents, with a variety of active mechanisms. High inter and intra-individual variability is known due to a variety of factors,1 therefore, an accurate, analytically sensitive quantitative method may play a role in researching the pharmacokinetic and pharmacodynamic effects of administration of cyclosporine, everolimus, sirolimus, and tacrolimus.
Here we describe a clinical research method using protein precipitation of whole blood with internal standards. Chromatographic elution was completed within 1.8 minutes using a Waters ACQUITY UPLC HSS C18 SB Column on a Waters ACQUITY UPLC I-Class System followed by detection on a Xevo TQD Mass Spectrometer (Figure 1).

Whole blood calibrators and quality control materials containing cyclosporine, everolimus, sirolimus, and tacrolimus were used. Internal standards ascomycin (Cerilliant, USA), 2H12-cyclosporine (Alsachim, France), 2H4-everolimus (Cerilliant, USA) and 2H3-sirolimus (IsoSciences, USA) were also sourced. The calibration range was 25–1500 ng/mL for cyclosporine and 1–30 ng/mL everolimus, sirolimus, and tacrolimus. QC concentrations were 150, 400, and 900 ng/mL for cyclosporine and 2, 8, and 22 ng/mL for everolimus, sirolimus, and tacrolimus.
To 50 µL of sample, add 200 µL 0.1 M ZnSO4(aq),vortex mix, add 500 µL of internal standard (ISTD) containing 2 ng/mL ascomycin and 2H4-everolimus, 25 ng/mL 2H12-cyclosporine and 4 ng/mL 2H3-sirolimus were added and vortex mix. Tubes were centrifuged for 2 minutes at 16,100 g and supernatant analyzed. Each analyte was analyzed individually.
|
System: |
ACQUITY UPLC I-Class (FTN) |
|
Needle: |
30 μL |
|
Column: |
ACQUITY UPLC HSS C18 SB Column, 1.8 μm, 2.1 x 30 mm (p/n 186004117) |
|
Mobile phase A: |
Water + 2 mM ammonium acetate + 0.1% formic acid |
|
Mobile phase B: |
Methanol + 2 mM ammonium acetate + 0.1% formic acid |
|
Needle wash solvent: |
80% aqueous methanol + 0.1% formic acid |
|
Purge solvent: |
Mobile phase A |
|
Seal wash: |
20% aqueous methanol |
|
Column temp.: |
55 °C (pre-column heater active) |
|
Injection volume: |
20 μL |
|
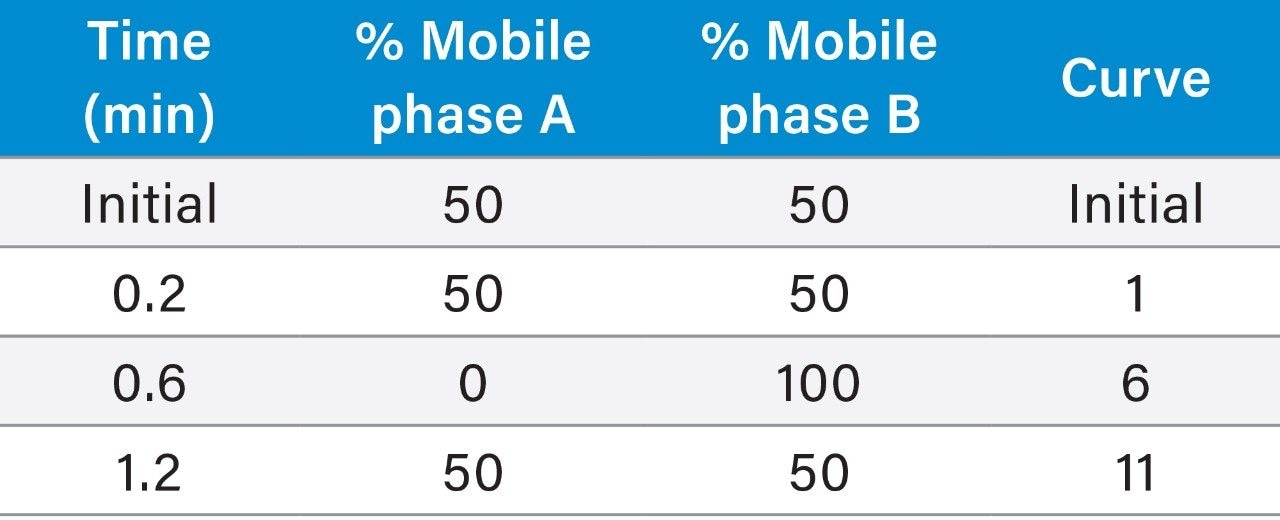
Flow rate: |
0.40 mL/min (equilibrate at 0.6 mL/min from 1.2 to 1.8 min) |
|
Run time: |
1.8 minutes (2.2 minutes injection-to-injection) |
|
System: |
Xevo TQD |
|
Resolution: |
MS1 (0.7 FWHM) MS2 (1.2 FWHM) |
|
Acquisition mode: |
Multiple Reaction Monitoring (MRM) (See Table 2 for details) |
|
Polarity: |
ESI+ |
|
Capillary: |
1.0 kV |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
400 °C |
|
Dwell time: |
0.05 seconds |
|
Inter-scan delay: |
0.02 seconds |
|
Inter-channel delay: |
0.01 seconds |
MassLynx v4.1 with TargetLynx Application Manager
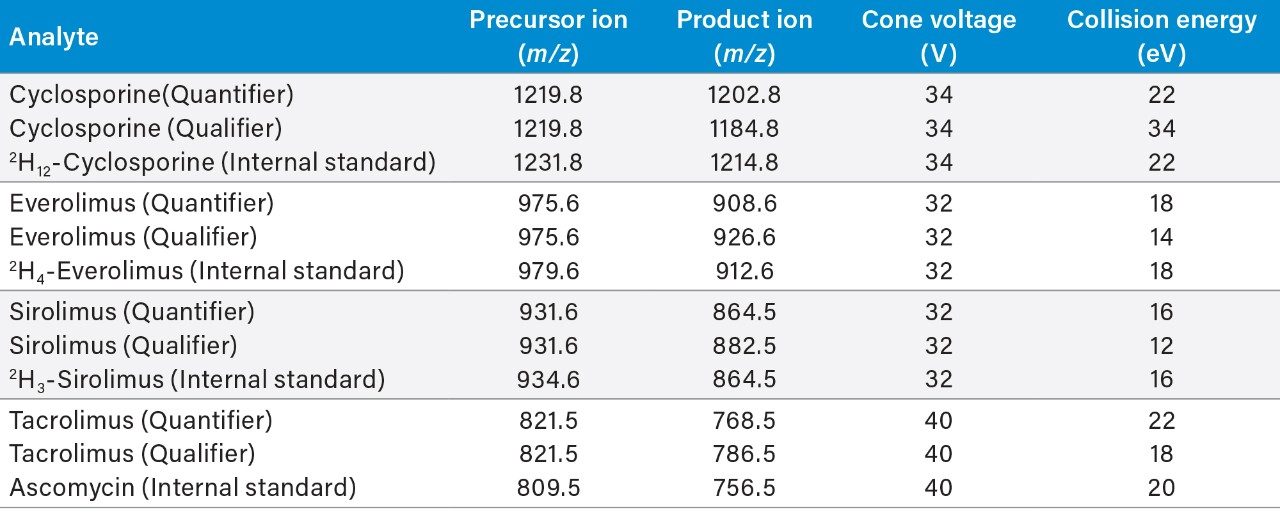
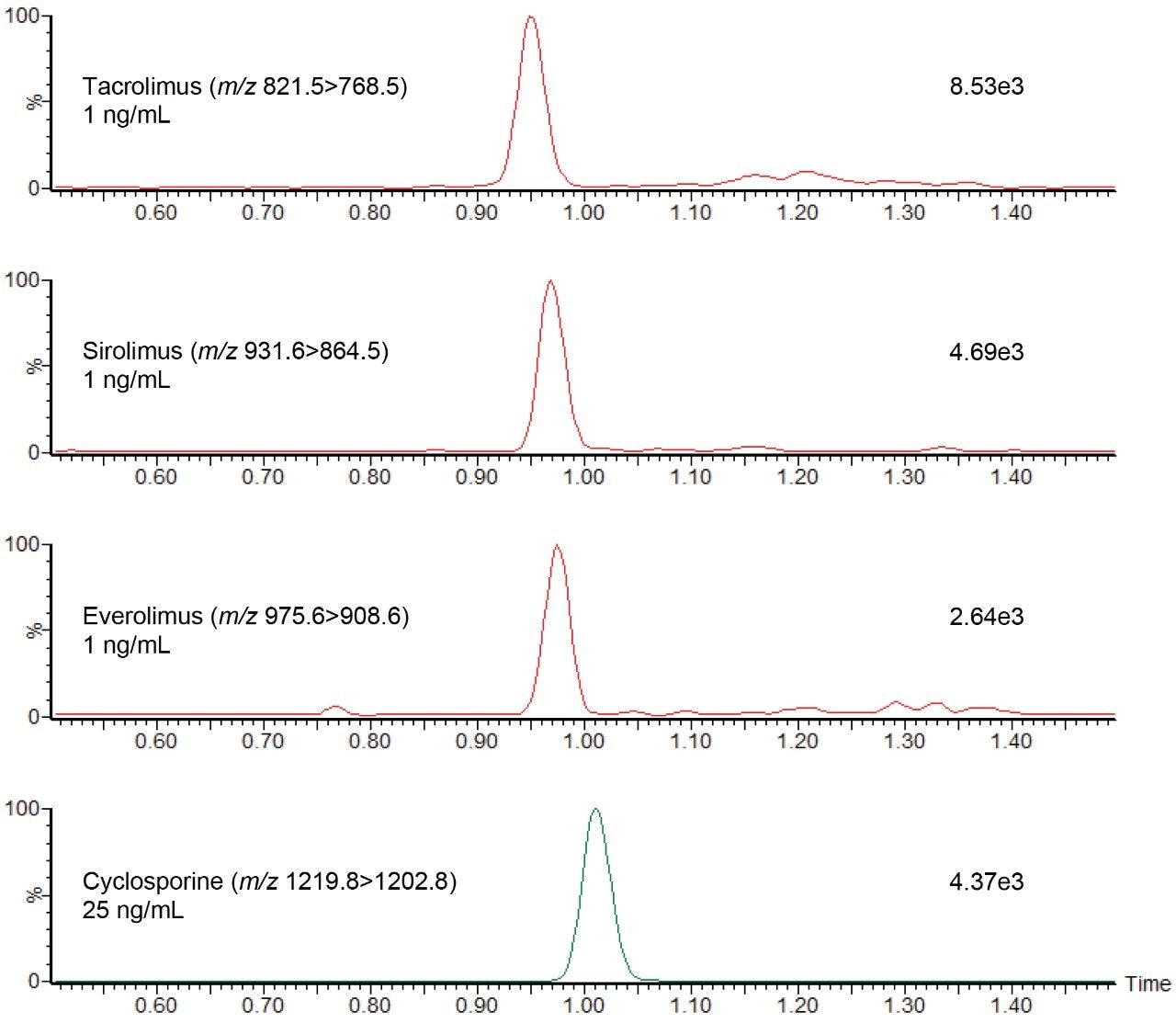
Figure 2 shows an example chromatogram of calibrator 1 (25 ng/mL cyclosporine and 1 ng/mL everolimus, sirolimus, and tacrolimus).
No system carryover was observed following analysis of whole blood samples with 3000 ng/mL of cyclosporine and 60 ng/mL of everolimus, sirolimus, and tacrolimus.
Analytical sensitivity investigations indicate the method would allow for precise quantification (<20% RSD, <15% bias) at 8 ng/mL for cyclosporine, 0.5 ng/mL for everolimus, 1 ng/mL for sirolimus, and 0.5 ng/mL for tacrolimus.
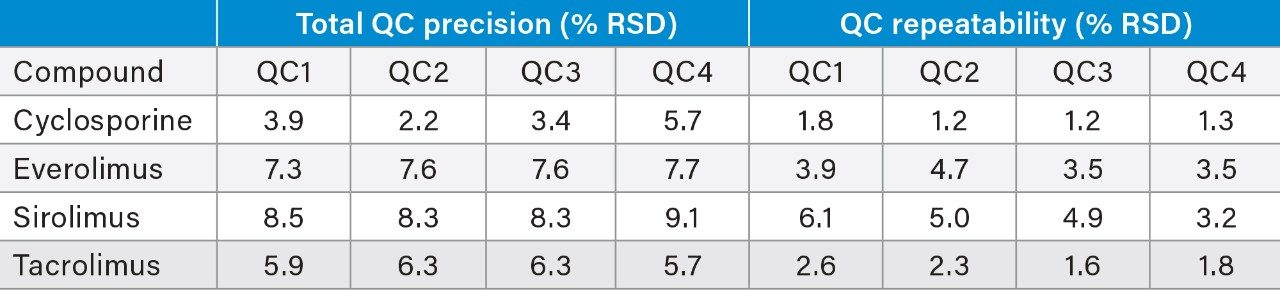
Total precision was determined by extracting and quantifying five replicates of four concentrations of whole blood pools over five separate days (n=25). Repeatability was assessed by analyzing five replicates at each QC level. Table 3 presents results of these experiments, where total precision and repeatability at the four concentrations assessed (50, 350, 800, and 1200 ng/mL for cyclosporine, 3.5, 10, 22, and 28 ng/mL for everolimus, sirolimus, and tacrolimus) was ≤9.1% RSD.
The method was shown to be linear over the range of 19.4–1662 ng/mL for cyclosporine, 1.0–30.1 ng/mL for everolimus, 0.7–30.1 ng/mL for sirolimus, and 0.7–34.8 ng/mL for tacrolimus when different ratios of high and low concentration pools were combined and analyzed.
A bias of less than 10% was achieved after 1:1 dilution (using blank whole blood) of high samples to be within the calibration range of the assay.
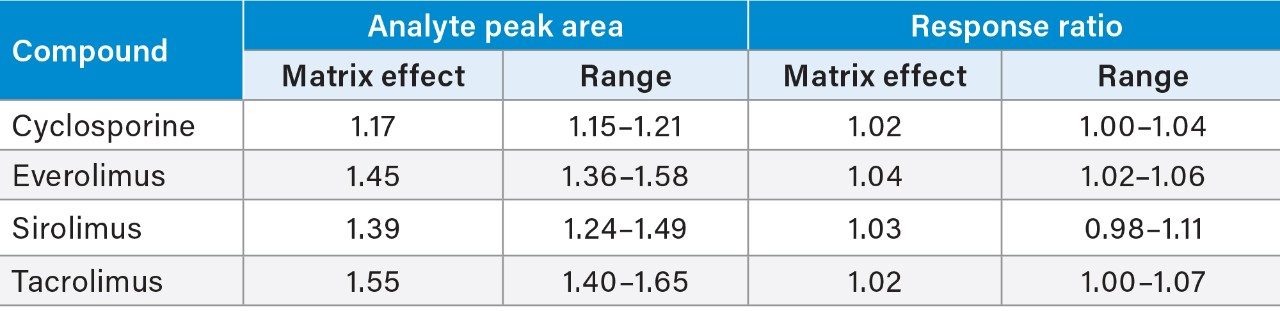
Matrix effects were evaluated as the peak area of extracted post spiked serum samples (n=6) taken as a percentage of extracted solvent samples spiked to equivalent concentrations. Calculation using analyte:internal standard response ratio indicated compensation for signal enhancement by the internal standard. Testing was carried out at 25 ng/mL for cyclosporine, 3 ng/mL for everolimus, 4 ng/mL for sirolimus, and 2 ng/mL for tacrolimus. Table 4 presents the results.
Potential interference from endogenous compounds (albumin, bilirubin, cholesterol, creatinine, triglycerides, uric acid, and vitamin B12) spiked at high concentrations was assessed by determining the recovery (n=3) from low and high pooled whole blood samples (100 and 1200 ng/mL cyclosporine, 4 and 20 ng/mL everolimus, sirolimus and tacrolimus. Recoveries ranged from 89.7–114.3% when assessing the effects of the presence of the other immunosuppressant drugs and internal standards. A substance was deemed to interfere if a recovery range of 85%–115% was exceeded.
NEQAS whole blood samples (n=33) were sourced and analyzed to assess method accuracy. All laboratory trimmed mean LC-MS concentrations spanned from 0–2119 ng/mL for cyclosporine, 0–38.6ng/mL for everolimus, 0–32.4 ng/mL for sirolimus, and 0–48.5 ng/mL for tacrolimus. Mean deviations of -0.2%, -11.9%, -10.7, and 1.6% respectively for cyclosporine, everolimus, sirolimus, and tacrolimus were obtained using this method.
Note: Only two laboratories returned NEQAS results for everolimus.
Using only 50 µL of sample with fast and inexpensive sample preparation, this analytical method provides sufficient analytical sensitivity to analyze low concentrations with excellent precision performance.
The method has been demonstrated as linear over the intended measuring intervals, with neither system carryover nor matrix effects and showed good agreement with an external quality assurance scheme.
720006424, November 2018