Il semplice schema mostrato nella Figura E mostra i componenti di un sistema di cromatografia liquida ad alte prestazioni [HPLC] di base.
Un recipiente contiene il solvente [definito fase mobile, perché si muove]. Una pompa ad alta pressione [sistema di erogazione del solvente o sistema di gestione degli eluenti] viene utilizzata per generare e misurare una velocità di flusso specificata di fase mobile, in genere espressa in millilitri al minuto. Un iniettore [sistema di gestione dei campioni o campionatore automatico] è in grado di introdurre [iniettare] il campione nel flusso continuo della fase mobile che trasporta il campione nella colonna HPLC. La colonna contiene il materiale di impaccamento cromatografico necessario per effettuare la separazione. Questo materiale di impaccamento è denominato fase stazionaria perché è tenuto in posizione dai componenti hardware della colonna. È necessario un rivelatore per vedere le bande di composti separati mentre eluiscono dalla colonna HPLC [la maggior parte dei composti è incolore, quindi non sono visibili a occhio nudo]. La fase mobile esce dal rivelatore e può essere inviata allo scarico o raccolta, in base al caso specifico. Quando la fase mobile contiene una banda di composti separati, l’HPLC consente di raccogliere questa frazione dell’eluito contenente il composto purificato per procedere a ulteriori studi. Questa tecnica è denominata cromatografia preparativa [viene trattata nella sezione relativa alla scala HPLC].
Da notare che vengono utilizzati raccordi e un tubo ad alta pressione per l’interconnessione dei componenti della pompa, dell’iniettore, della colonna e del rivelatore in modo da formare il condotto per la fase mobile, il campione e le bande di composti separati.
![Figura E: Sistema di cromatografia liquida ad alte prestazioni [HPLC]](/content/dam/waters/it/figures/primers/hplc/primer_e_lcsystem.jpeg.82.resize/img.jpeg)
Il rivelatore è collegato alla stazione dati del computer, il componente del sistema HPLC che registra il segnale elettrico necessario per generare il cromatogramma sul display, così come per identificare e quantificare la concentrazione degli elementi costituenti del campione (vedere la Figura F). Poiché il composto campione può avere caratteristiche molto diverse, sono stati sviluppati vari tipi di rivelatori. Per esempio, se un composto è in grado di assorbire la luce ultravioletta, viene utilizzato un rivelatore di assorbanza UV. Se il composto è fluorescente, viene utilizzato un rivelatore a fluorescenza. Se il composto non presenta nessuna di queste caratteristiche, viene utilizzato un tipo di rivelatore più universale, come l’Evaporative Light Scattering Detector (rilevatore evaporativo a luce diffusa) [ELSD]. L’approccio più efficace consiste nell’utilizzo di più rivelatori in serie. Per esempio, un rivelatore UV e/o ELSD può essere utilizzato in combinazione con uno spettrometro di massa [MS] per analizzare i risultati della separazione cromatografica. Con un’unica iniezione, fornisce informazioni più complete su un analita. La pratica di accoppiare uno spettrometro di massa a un sistema HPLC è denominata LC-MS.
![Figura F: Un tipico sistema HPLC [Waters Alliance]](/content/dam/waters/it/figures/primers/hplc/primer_F_AllianceHPLCSystem.jpeg.82.resize/img.jpeg)
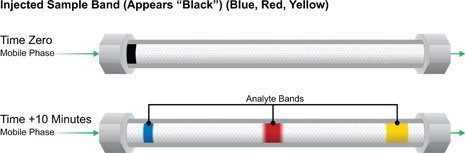
Per comprendere con facilità come si ottiene la separazione dei composti contenuti in un campione, vedere il grafico presente nella Figura G.
La fase mobile entra nella colonna da sinistra, attraversa il letto di particelle ed esce a destra. La direzione del flusso è rappresentata da frecce verdi. In primo luogo, considerare l’immagine in alto, che rappresenta la colonna al tempo zero [il momento dell’iniezione], quando il campione entra nella colonna e inizia a formare una banda. Il campione mostrato in questo caso, una miscela di coloranti giallo, rosso e blu, ha l’aspetto di un’unica banda nera all’ingresso della colonna. [In realtà, questo campione potrebbe essere qualsiasi cosa che possa essere dissolta in un solvente; in genere i composti sono incolori e le pareti della colonna sono opache; pertanto, sarebbe necessario un rivelatore per visualizzare i composti separati durante l’eluizione.]
Dopo alcuni minuti [immagine in basso], durante i quali la fase mobile scorre in maniera ininterrotta e costante attraverso le particelle del materiale di impaccamento, è possibile osservare che i singoli coloranti si sono spostati in bande separate a velocità diverse. Ciò è dovuto al fatto che esiste una competizione tra la fase mobile e la fase stazionaria per l’attrazione di ciascuno dei coloranti o analiti. Da notare che la banda del colorante giallo è quella che si sposta più velocemente e sta per uscire dalla colonna. Il colorante giallo preferisce [è attratto da] la fase mobile più degli altri coloranti. Pertanto, si sposta a una velocità superiore, simile a quella della fase mobile. La banda del colorante blu preferisce il materiale di impaccamento alla fase mobile. A causa della maggiore attrazione per le particelle, il suo movimento è notevolmente più lento. In altre parole, è il composto a ritenzione maggiore all’interno di questa miscela di campioni. La banda del colorante rosso esercita un’attrazione intermedia per la fase mobile e pertanto si sposta a una velocità intermedia attraverso la colonna. Poiché ogni banda di colorante si muove a una velocità diversa, siamo in grado di separarla a livello cromatografico.
Quando le bande di coloranti separati lasciano la colonna, passano subito nel rivelatore. Il rivelatore contiene una cella di flusso che vede [rivela] ciascuna banda di composto separato su un fondo di fase mobile [vedere la Figura H]. [In realtà, le soluzioni di molti composti a concentrazioni analitiche HPLC tipiche sono incolori.] Un rivelatore adeguato è in grado di rivelare la presenza di un composto e di inviare il corrispondente segnale elettrico alla workstation di un computer. È possibile scegliere tra molti tipi diversi di rivelatori, a seconda delle caratteristiche e delle concentrazioni dei composti da separare e analizzare, come indicato in precedenza.
Un cromatogramma è una rappresentazione della separazione che ha avuto luogo a livello chimico [cromatografico] nel sistema HPLC. Viene tracciata una serie di picchi rispetto a una linea di base su un asse del tempo. Ciascun picco rappresenta la risposta del rivelatore per un composto diverso. Il cromatogramma viene tracciato dalla workstation del computer [vedere la Figura H].
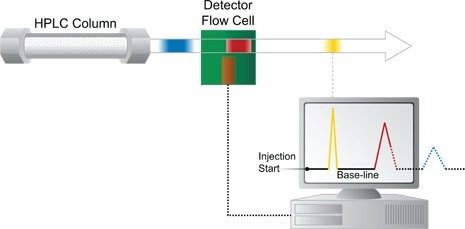
Nella Figura H, la banda gialla ha attraversato completamente la cella di flusso del rivelatore; il segnale elettrico generato è stato inviato alla workstation del computer. Il cromatogramma risultante inizia a essere visualizzato a schermo. Da notare che il cromatogramma inizia quando il campione viene iniettato per la prima volta e inizialmente è una linea retta presente nella parte inferiore dello schermo. Prende il nome di linea di base e rappresenta la fase mobile pura che attraversa la cella di flusso nel tempo. Quando la banda gialla dell’analita attraversa la cella di flusso, al computer viene inviato un segnale più forte. La linea curva prima verso l’alto e quindi verso il basso in misura proporzionale alla concentrazione del colorante giallo nella banda del campione. In questo modo si crea un picco nel cromatogramma. Dopo che la banda gialla è uscita completamente dalla cella del rivelatore, il livello del segnale ritorna sulla linea di base; a questo punto la cella di flusso contiene, di nuovo, soltanto fase mobile pura. Poiché la banda gialla si sposta più velocemente, è la prima a eluire dalla colonna: si tratta del primo picco tracciato.
Poco dopo, la banda rossa raggiunge la cella di flusso. Il segnale sale dalla linea di base quando la banda rossa entra per la prima volta nella cella, iniziando a tracciare il picco che rappresenta la banda rossa. In questo grafico la banda rossa non ha attraversato completamente la cella di flusso. Il grafico mostra come apparirebbero la banda rossa e il picco rosso nel caso di interruzione del processo in questo momento. Poiché la maggior parte della banda rossa ha attraversato la cella, la maggior parte del picco è stata tracciata, come mostrato dalla linea continua. Potendo riavviare, la banda rossa attraverserebbe completamente la cella di flusso, completando il picco rosso [linea tratteggiata]. La banda blu, quella a ritenzione più forte, si sposta alla velocità più bassa ed eluisce dopo la banda rossa. La linea tratteggiata mostra come apparirebbe il cromatogramma completato se avessimo lasciato continuare l’analisi fino alla conclusione. È interessante notare che l’ampiezza del picco blu sarà la più ampia perché l’ampiezza della banda blu dell’analita, pur essendo la più stretta sulla colonna, diventa la più ampia mentre eluisce dalla colonna. Questo perché si muove più lentamente attraverso il letto di materiale di impaccamento cromatografico e richiede più tempo [e volume di fase mobile] per un’eluizione completa. Poiché la fase mobile fluisce ininterrottamente a una velocità fissa, la banda blu si allarga ed è più diluita. Poiché il rivelatore risponde in misura proporzionale alla concentrazione della banda, il picco blu ha un’altezza inferiore, ma un’ampiezza maggiore.
Cos’è l’HPLC (cromatografia liquida ad alte prestazioni)
Come Funziona la Cromatografia Liquida ad Alte Prestazioni (HPLC)?