
This is an Application Brief and does not contain a detailed Experimental section.
This application brief describes how the use of ion mobility-mass spectrometry (IM-MS) can facilitate enhanced mass spectral clean up, producing spectra that are both retention time and ion mobility aligned. This simplifies structural elucidation and improves the efficiency of location and identification of target analytes.
The use of Waters UltraPerformance Liquid Chromatography (UPLC) in combination with high resolution mass spectrometry has increased significantly in food and environmental laboratories in recent times, offering unique advantages for screening and profiling complex mixtures. Waters’ introduction of ion mobility mass spectrometry (IM-MS), which combines high resolution mass spectrometry and high efficiency ion mobility based measurements and separations, has taken the quality and volume of information that can be obtained from food and environmental samples to a new level. One of the benefits of IM-MS enabled solutions is the ability to clean up the mass spectra of compounds using ion mobility.
Ion mobility spectrometry (IMS) is a rapid, orthogonal, gas phase separation technique that provides another dimension of separation within an LC timeframe, allowing compounds to be differentiated based on size, shape, and charge. In addition, precursor ions (with isotopes), fragment ion information, and Collision Cross Section (CCS) values can be acquired in a single acquisition.
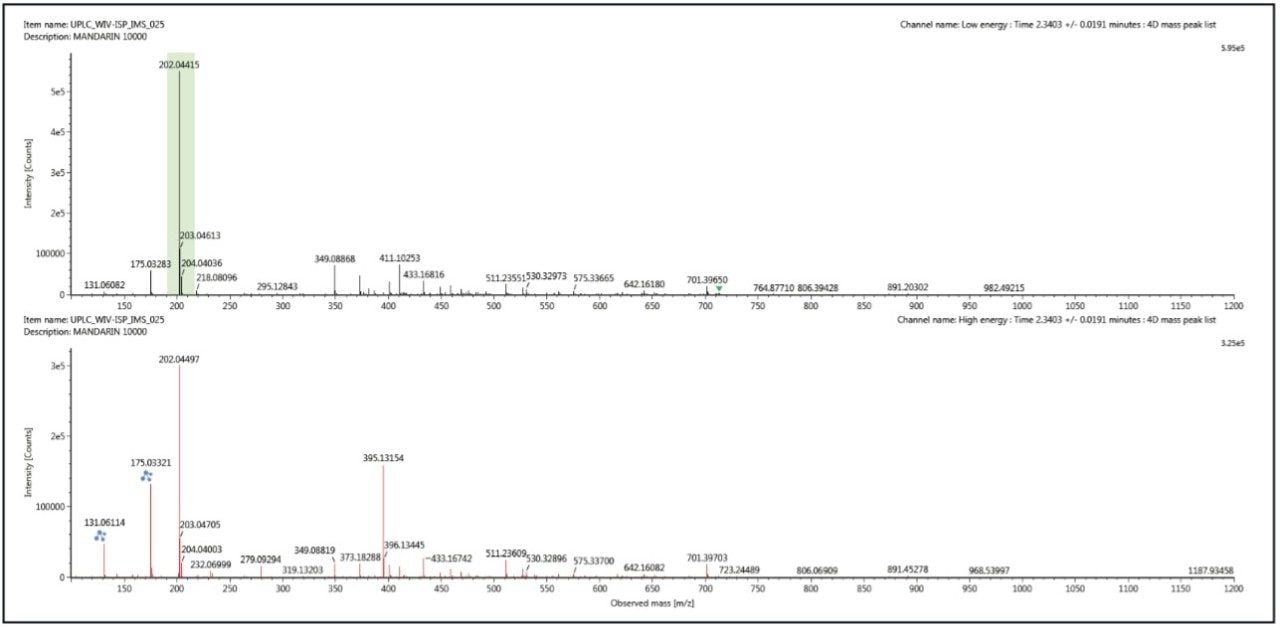
In food and environmental analysis one of the major challenges is the variety of sample matrices encountered and overcoming the subsequent analytical matrix effects. One of those matrix effects is the presence of components in the matrix that can interfere with the confirmation and identification of target compounds and unknowns. Typically mass spectra are generated by integrating chromatographic peaks using a variety of peak detection algorithms, then extracting a representative spectrum at the retention time corresponding to the maximum intensity of the peak. Complex mixed mass spectra are often generated when matrix components co-elute with the compound of interest. To attempt to overcome this, background subtraction routines are often used with varying success, depending on the complexity of the sample.
Figure 1 shows the conventional retention time aligned precursor and fragment ion spectra for thiabendazole (m/z 202) generated using 3D peak detection algorithm within UNIFI Software, which typically generates cleaner spectra compared to other available peak detection algorithms. Many other ions can be seen in the spectrum, particularly above the ion corresponding to thiabendazole (m/z 202).
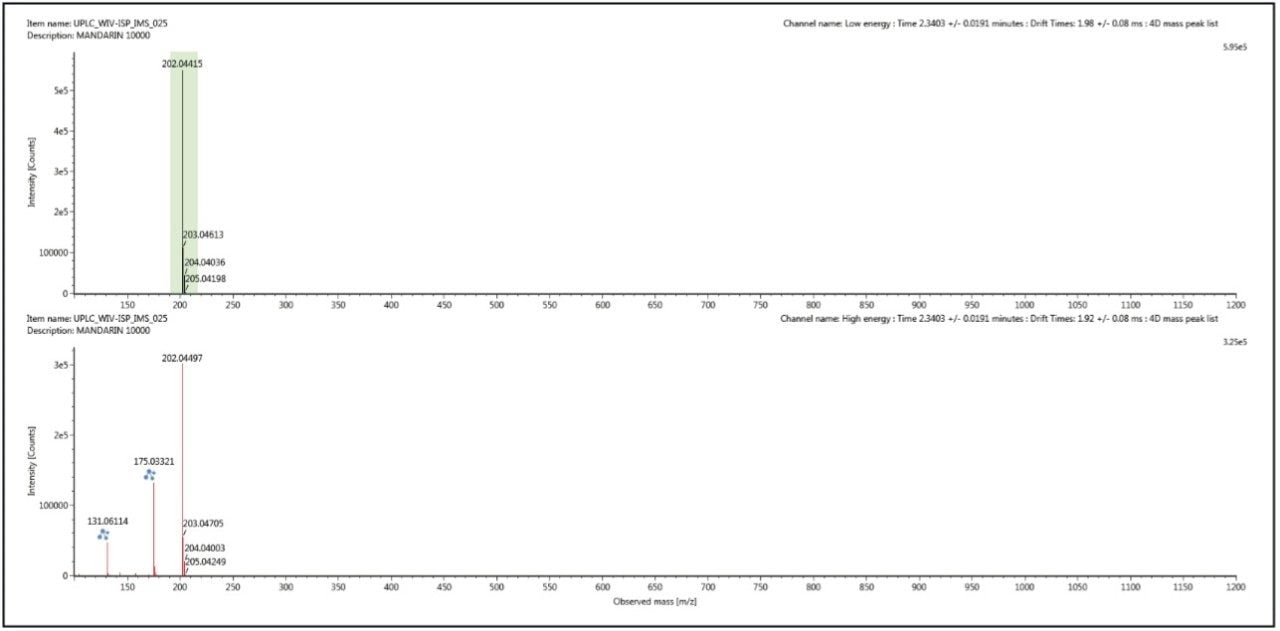
When acquiring ion mobility data using Waters’ IM-MS systems mass spectra obtained by the process described can be cleaned up further. Using UNIFI’s Ion Mobility Resolve function, the multi-component spectrum is completely “cleaned up” using UNIFI’s 4D peak detection algorithm, as shown in Figure 2. The subsequent precursor ion and fragmentation spectra are both retention time aligned and ion mobility aligned giving an extra dimension of selectivity and more confidence in compound confirmation and identification.
Examples showing the use of ion mobility for mass spectral cleanup can be found here:
The use of ion mobility is an extremely powerful analytical tool using Waters’ Ion Mobility-Mass Spectrometry (IM-MS) systems. One benefit of this technique is to facilitate enhanced mass spectral clean up producing spectra that are both retention time and ion mobility aligned. This simplifies structural elucidation and improves the efficiency of location and identification of target analytes.
720005375, April 2015