For forensic toxicology use only.
This is an Application Brief and does not contain a detailed Experimental section.
This application brief investigates the utility of the innovative data acquisition mode MSE for the screening of toxicants in human specimens.
Laboratories are frequently required to perform broad screening techniques on complex biological samples to identify drugs of abuse and other toxicants. In recent years there has been an increased interest in the use of Time-of-Flight (Tof) instruments for this purpose owing to the high level of specificity offered by accurate mass data.
While theoretical or exact mass libraries can be automatically generated without reference material i.e., from molecular formulae, the lack of additional information can lead to false positive results in the analysis of authentic samples. Thus, where possible, additional information e.g., an associated retention time (RT) and confirmatory fragment ions should be used to increase confidence in drug identification and to improve the ease and speed of data review and reporting.
MSE is a novel, patented mode of data acquisition that permits the seamless collection of a comprehensive catalogue of information for both precursor and fragment ions in a single analysis.1–3 This is achieved by rapidly alternating between two functions i.e., the first, acquired at low collision energy provides an accurate mass measurement of the precursor ion. The second, at elevated energy provides accurate masses of the fragment ions. In addition to providing increased confidence in identification, fragmentation can help to differentiate between isobaric compounds.
This application brief includes examples of MSE data for toxicological compounds and summarizes some of the key benefits of this acquisition mode in comparison to conventional data-dependent techniques. We describe the flexibility around data processing and summarize the contents of the UNIFI™ Waters™ Forensic Toxicology Library.
ACQUITY™ UPLC™ I-Class PLUS (FTN) System in combination with the Xevo™ G2 XS QTof Mass Spectrometer
|
Column: |
ACQUITY UPLC HSS C18, 1.8 µm, 2.1 x 150 mm (p/n: 186003534) |
|
Run time: |
15 min |
|
Ionization mode: |
ESI+ |
|
Acquisition range: |
m/z 50–1000 |
|
MSE conditions |
Collision energy function 1:6 eV Collision energy function 2:ramp 10–40 eV |
UNIFI Software was used in combination with the Waters Forensic Toxicology Scientific Library.
Certified reference material (CRM) for toxicologically-relevant compounds were obtained from Merck (Dorset, UK) and were analysed using UPLC-Tof in MSE mode. Figure 1 shows the MSE data obtained following analysis of a representative substance, buflomedil. The figure illustrates how a confident identification can be obtained from the ability to measure the mass of the precursor ion to four decimal places (precursor mass is shown in the low energy spectrum). When MSE is utilized, even greater confidence in identification can be achieved by additional incorporation of the masses of the specific fragment ions which are generated when the collision energy is ramped (high energy spectrum). Furthermore, RT can also be included in the identification criteria.
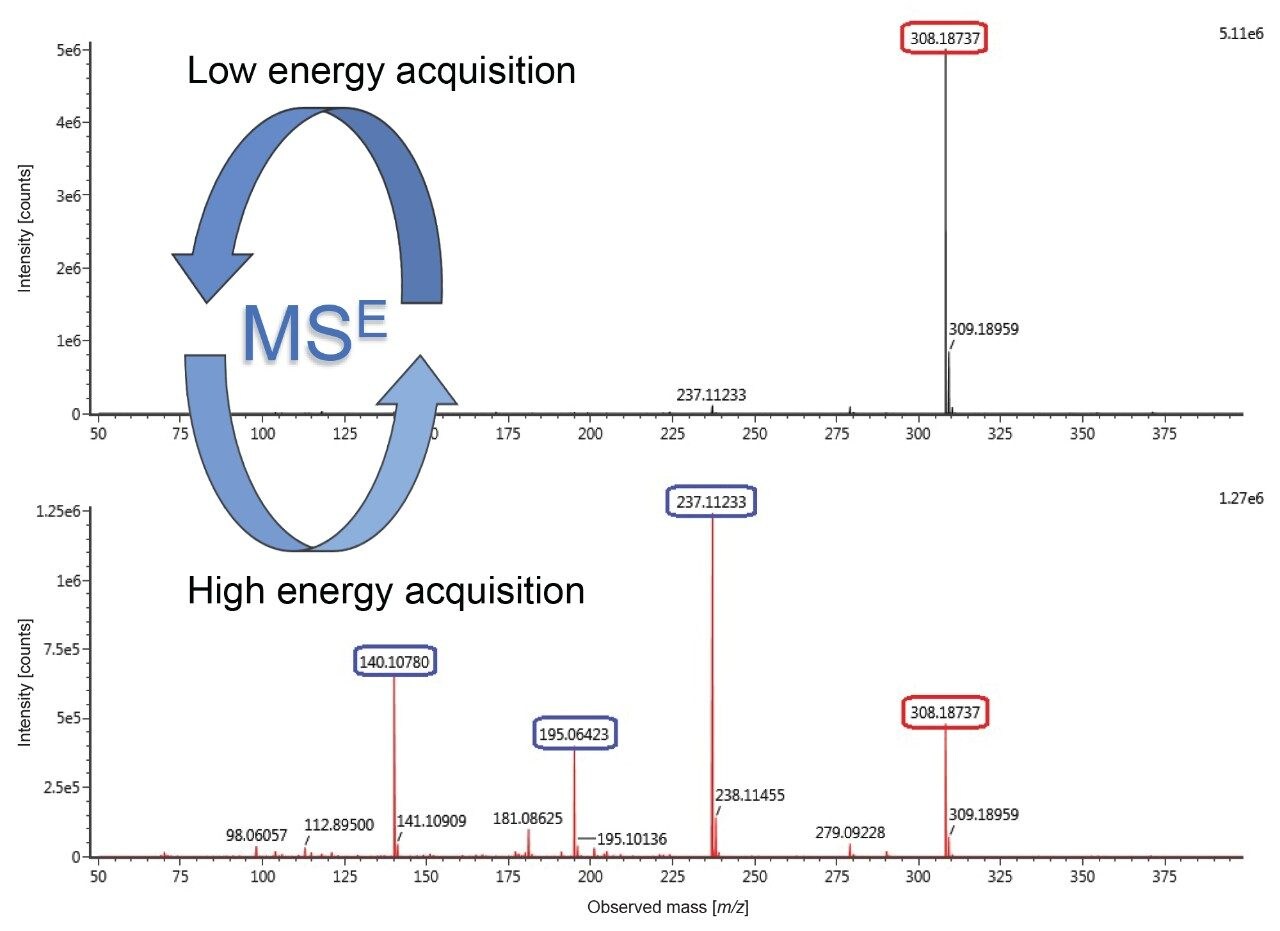
MSE is a data-independent technique meaning that full accurate mass data is being acquired continually under both low and high energy conditions throughout the analysis. This is in contrast to data-dependent (or information-dependent) techniques, where typically the instrument commences collecting full accurate mass data at low energy to provide mass information of the intact molecules until a 'trigger' is received which then instructs the instrument to switch to collecting data in tandem mass spectrometry (MS/MS) mode. The trigger could be any precursor ion exceeding a response threshold, or alternatively the instrument may be set to trigger on detection of specifically targeted precursor ion(s). The disadvantage of data-dependent approaches can be that while the instrument is collecting MS/MS - it is not collecting full scan MS data, thus the data is incomplete. In forensic toxicology a complete and unrestricted dataset is particularly advantageous as it provides the ability to retrospectively examine the data without fear that potentially relevant data has been omitted by use of targeted acquisition techniques such as data-dependent analysis such as conventional MS/MS. In other words, the user can reprocess existing data without the need to reanalyze or reacquire additional data for the sample.
However, the key benefit of having the complete data means that it opens up the ability to process data using three complementary workflows as summarized in Figure 2.
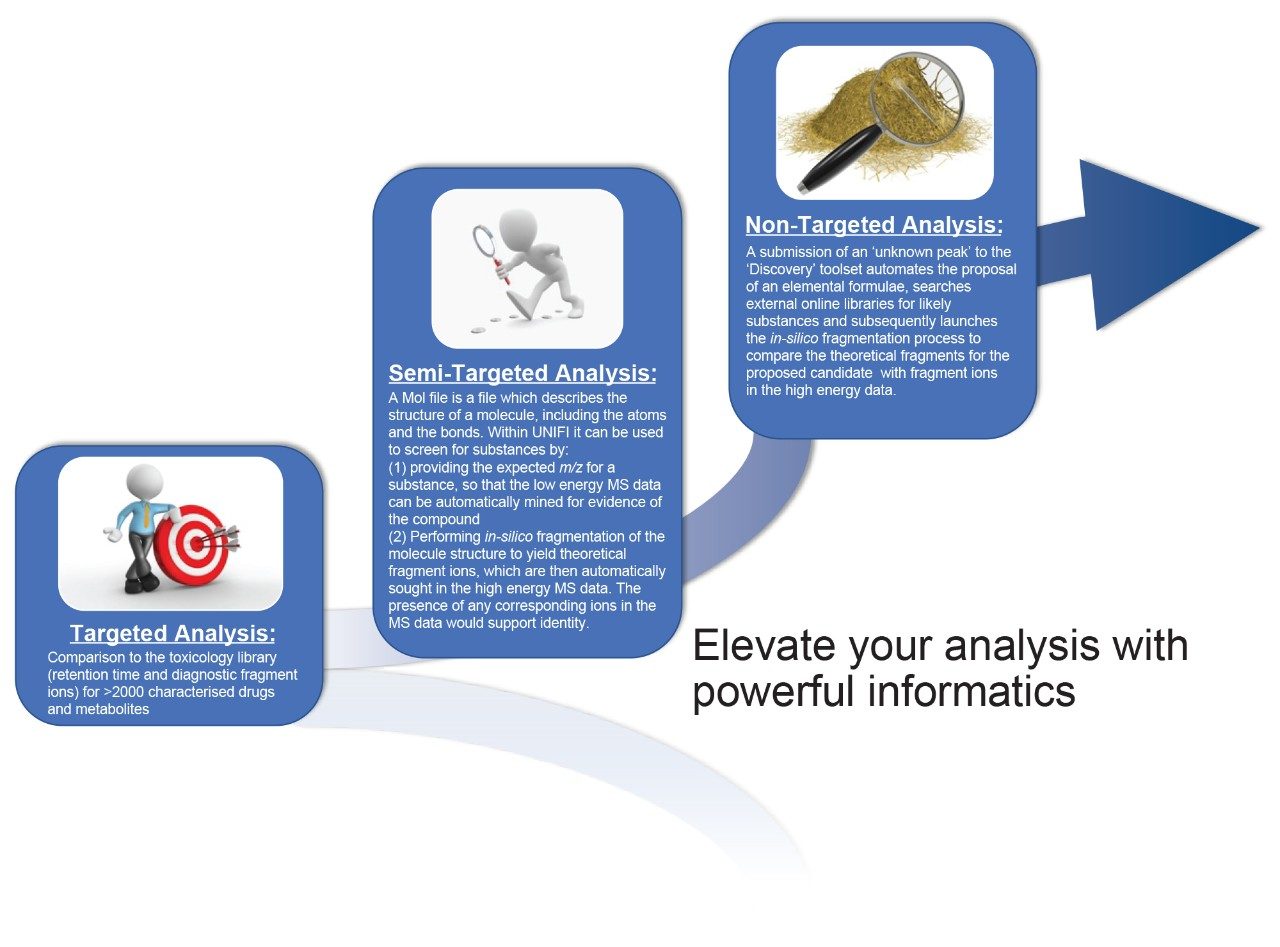
Targeted analysis is the most straightforward approach, where the acquired data is simply matched against a reference library. The UNIFI Forensic Toxicology Screening Solution includes a comprehensive library, in which each library entry comprises a reference RT together with the exact mass of the precursor ion and verified diagnostic exact mass fragment ions. Figure 3 shows an image of a representative entry from the Waters Forensic Toxicology Library.
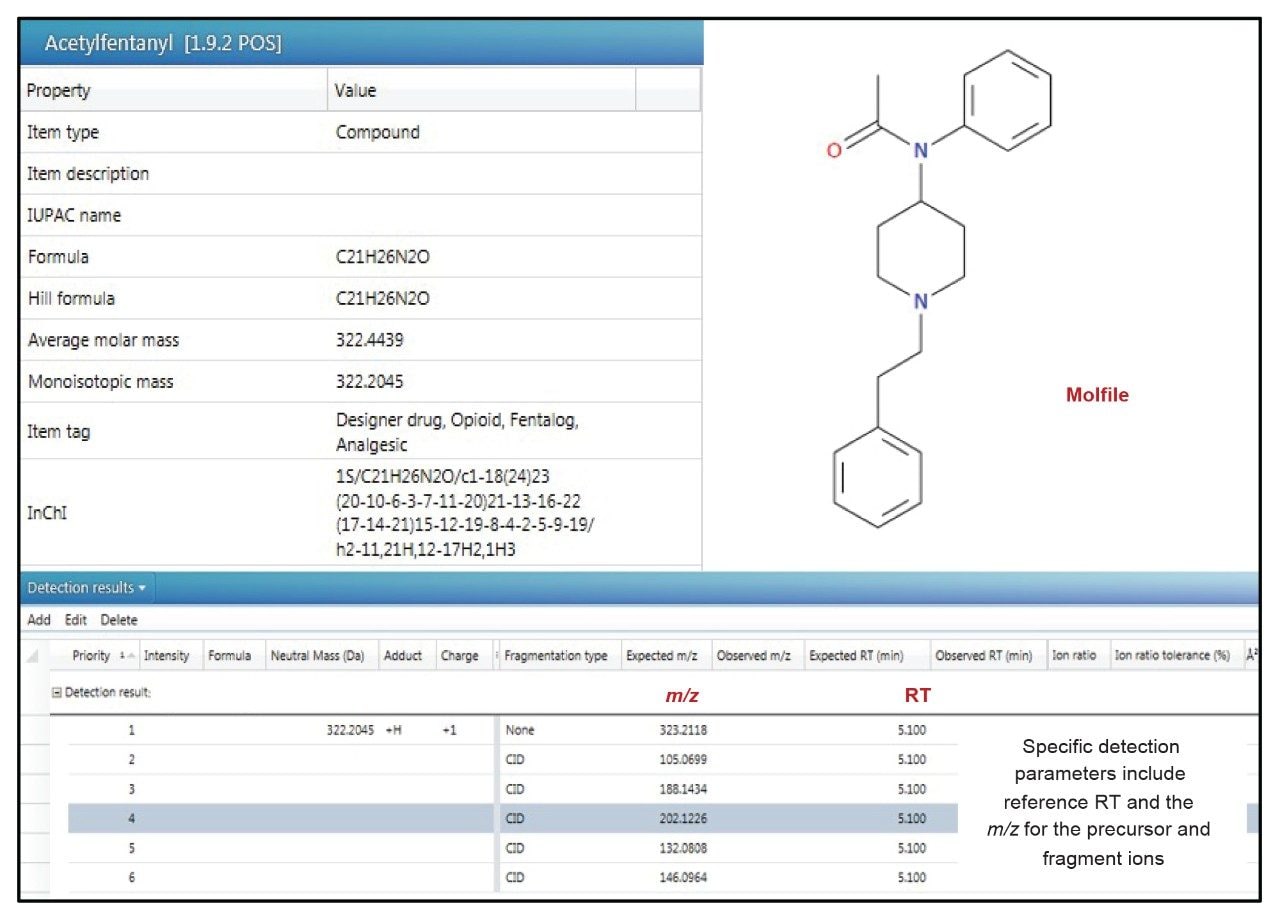
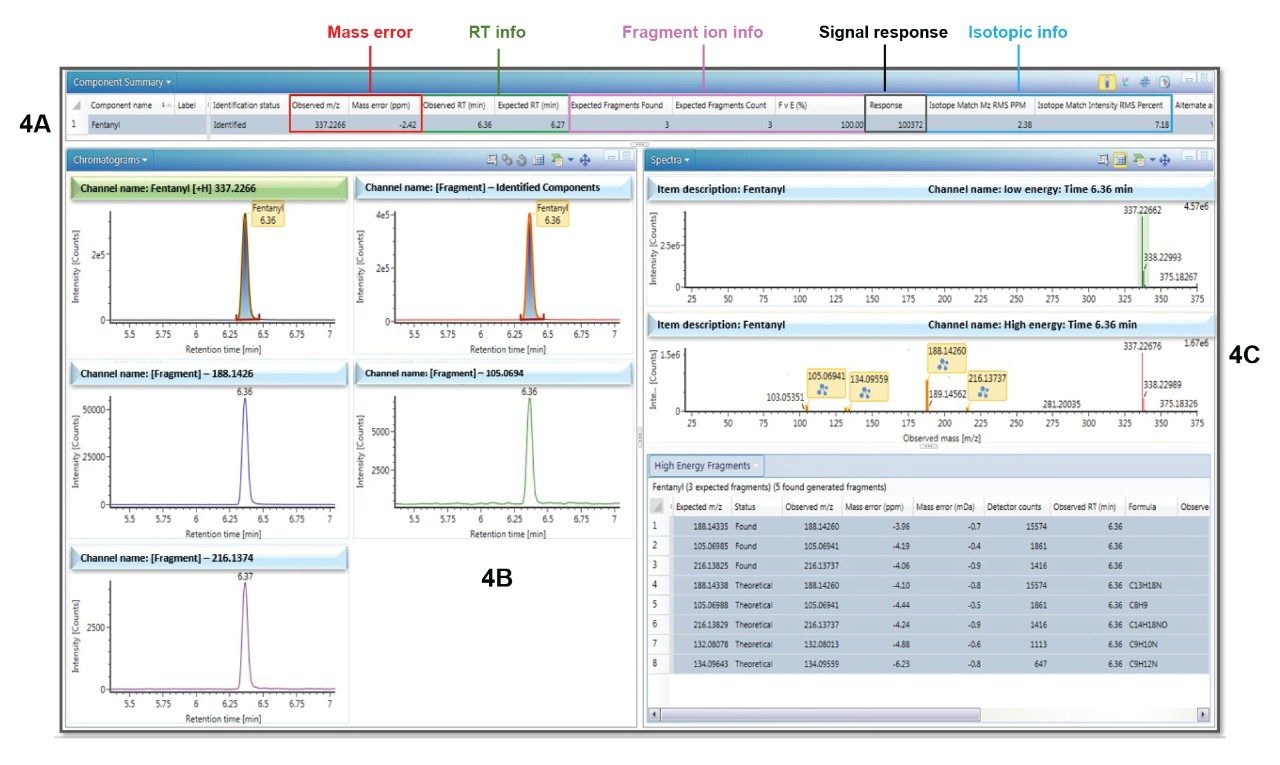
Figure 4 shows the results browser for a typical sample processed in this way and demonstrates the wealth of information that is available for use in the library matching process, and which provides the user with a fast, clear, and confident identification.
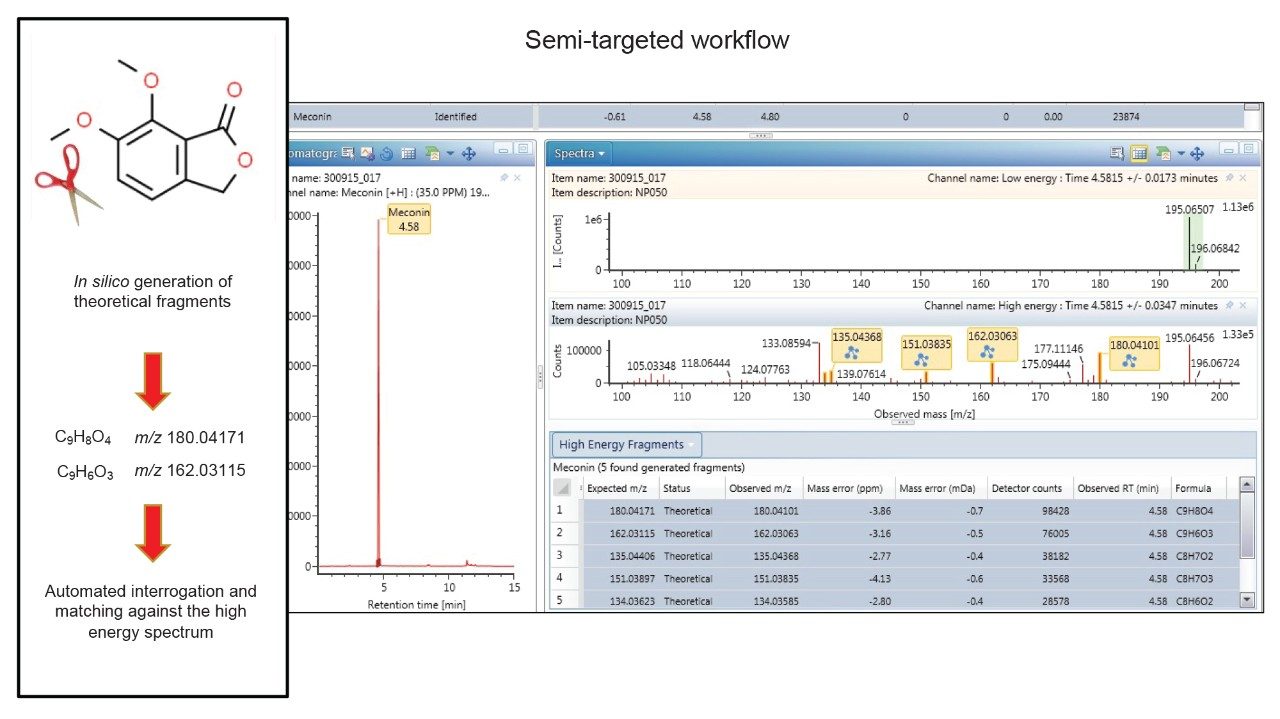
One of the key benefits of high resolution/accurate mass instruments is that even in the absence of a fully characterized library entry, the user still has the ability to screen for drug substances on the basis of their exact mass. This is especially beneficial for forensic toxicology laboratories as it enables the user to screen for novel or emerging drug substances without the requirement of CRM; this is invaluable as access to commercial reference material for new analogues is often delayed. In semi-targeted processing, a Molfile is utilized; this file describes the elemental formula and overall arrangement of the bonds of the compound of interest. During the automated processing, evidence of the m/z for the precursor ion (as determined from the Molfile) is sought in the low energy trace (Figure 5). While this information alone, is useful for a tentative identification, in addition UNIFI performs in-silico fragmentation of the Molfile to generate theoretical fragment ions which are then sought in the high energy data. A sample containing evidence of both precursor and theoretical fragment ions for a particular drug substance, demonstrates a higher confidence in likely identity.
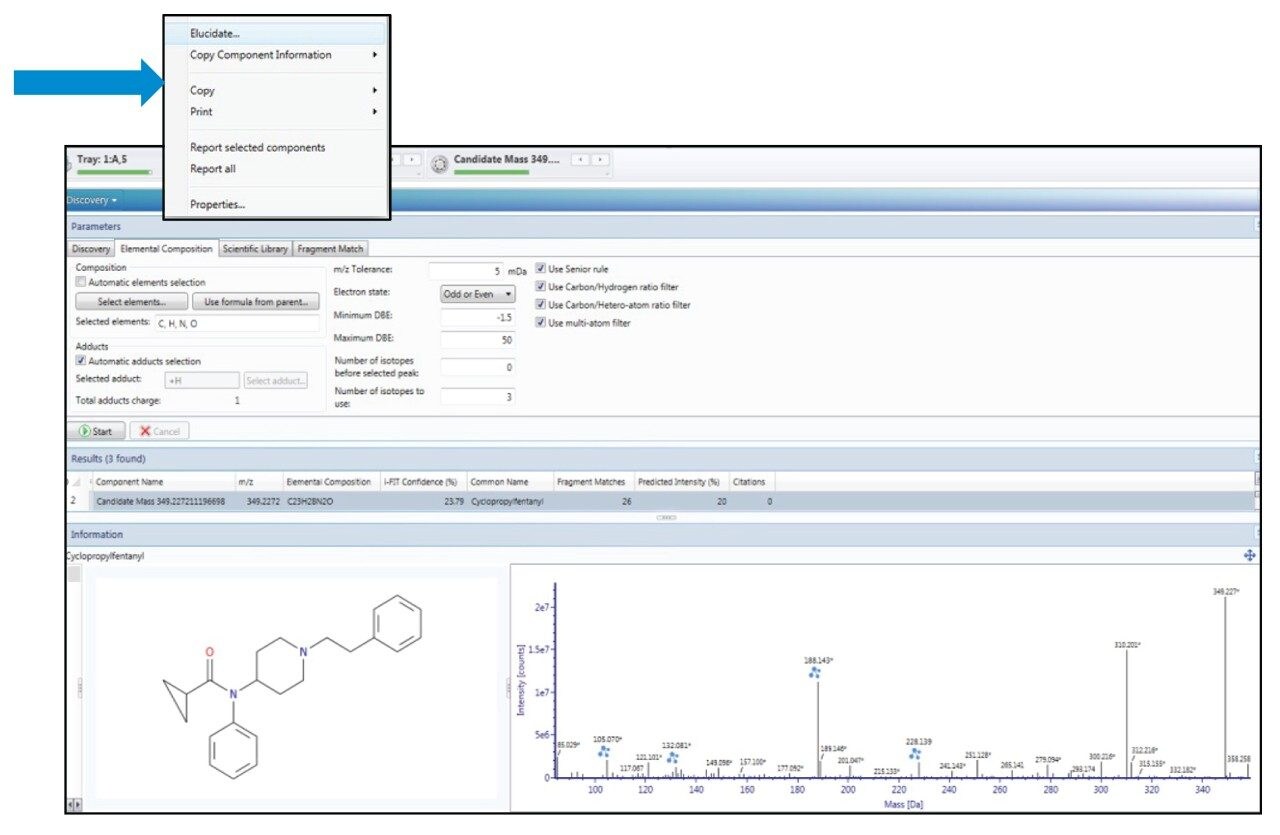
Discovery workflows can be applied where there is an unknown peak in the data that is not identified by either targeted, or semi-targeted, workflows. Under these circumstances, UNIFI offers a full suite of discovery tools that can be used to elucidate the structure of the unknown. The first step in the discovery process is to determine the likely elemental formula(e) of the substance; UNIFI achieves this based on the accurate mass and isotopic information of the precursor mass in the low energy trace of the MSE data. The second step is to assign any likely substances that correspond to that measured formula. UNIFI achieves this by searching online chemical databases such as those contained within Chemspider and simultaneously accessing the Molfile associated with that substance. In the third step and final step, UNIFI performs the in-silico fragmentation process for any proposed substances and compares the theoretical ions with the fragment ions observed in the high energy trace of the unknown substance. The greater the similarity of the acquired to the theoretical, the higher the confidence in the proposed identity. Further confirmation of this type of preliminary tentative identification would require verification of retention time and fragment ions through analysis of CRM. Figure 6 shows an example of a tentative identification of cyclopropylfentanyl using the discovery workflow. A more detailed description of the discovery workflow, with illustrated examples, is available in another Application Note.4
MSE is a powerful acquisition mode that provides a complete catalogue of accurate mass data. Identification by this technique is based on a combination of retention time and an accurate mass fingerprint of the analyte. While the use of Tof instruments provide the capability to assign masses to four decimal places and offers improved specificity over nominal mass data, the additional information of the diagnostic fragments provided by use of MSE represents further specific identification parameters and minimizes false positives. Ion ratios of the diagnostic can also be incorporated into the user’s identification criteria.5 Furthermore, fragment ions can be useful to differentiate isomers. Together, these capabilities translate into faster, easier data review for the user, and an overall high confidence in identification.
As a complete, non-restricted catalogue of accurate mass information MSE also enables discovery workflows which can be applied to facilitate identification of substances that may be new, emerging psychoactive substances and analogues.
1. Plumb R, Johnson K, Rainville P, Smith B, Wilson I, Castro-Perez J, Nicholson J. UPLC/MSE; A New Approach for Generating Molecular Fragment Information for Biomarker Structure Elucidation. Rapid Commun. Mass Spectrom. (2006) 20: 1984–1994.
2. Ni S, Qian D, Duan J, Guo J, Shang E, Shu Y. UPLC–QTof/MS-Based Screening and Identification of the Constituents and Their Metabolites in Rat Plasma and Urine After Oral Administration of Glechoma Longituba Extract. Journal Chromatogr. B (2010) 878: 2741–2750.
3. Hernandez F, Bijlsma L, Sancho JV, Dıaz R, Ibanez M. Rapid Wide-Scope Screening of Drugs of Abuse; Prescription Drugs With Potential for Abuse and Their metabolites in Influent and Effluent Urban Wastewater by UHPLC-QTof-MS. Analytica Chimica Acta (2011) 684: 96–106.
4. Goshawk J and Wood M. Analysis of Plant Alkaloids Through Accurate Mass Screening and Discovery. Waters Application Note 720005461 (2015).
5. Goshawk J and Wood M. Evaluation of Ion Ratios as an Additional Level of Confirmation in Accurate Mass Toxicology Screening. Waters White Paper 720005866 (2016).
720005198, Revised March 2022