In this application note, we demonstrate the feasibility of successfully isolating a synthetic pharmaceutical compound from a crude mixture using AutoPurify on an AutoPurification System configured with an ACQUITY QDa Detector.
Pharmaceutical compounds synthesized in chemistry laboratories almost always require purification before they can be used in experimental studies. Preparative chromatography is often the technique used to clean up synthetic mixtures, but the multi-step purification process can be cumbersome and time-consuming. AutoPurify, a feature provided within the FractionLynx Application Manager of MassLynx Software, streamlines the purification process with flexible, automated strategies that reduce or eliminate user intervention between isolation steps. While the benefits of AutoPurify have been discussed in detail previously,1 the integration of the ACQUITY QDa Detector into the AutoPurification System makes mass-directed purification more readily accessible and provides the added assurance that mass spectral data brings to chromatographic separations. With its pre-optimized hardware and automated calibration routine, the ACQUITY QDa Detector can be easily added to purification systems. In this application note, we demonstrate the feasibility of successfully isolating a synthetic pharmaceutical compound from a crude mixture using AutoPurify on an AutoPurification System configured with an ACQUITY QDa Detector.
Dry pharmaceutical intermediate was dissolved in dimethylsulfoxide to a concentration of 50 mg/mL.
|
LC system: |
Waters AutoPurification System |
|
Detectors: |
ACQUITY QDa (mass); 2998 Photodiode Array |
|
Analytical column: |
XSelect CSH C18, 4.6 x 50 mm, 5 μm (p/n 186005287) |
|
Preparative column: |
XSelect CSH C18 OBD Prep, 19 x 50 mm, 5 μm (p/n 186005420) |
|
Mobile phase A: |
Water with 0.1% formic acid |
|
Mobile phase B: |
Methanol, neat |
|
Column temperature: |
Room |
|
Sample temperature: |
Room |
|
Injection volume: |
Reported in figures |
|
Flow rate: |
Reported in figures |
|
Gradient: |
Reported in figures |
|
Ionization mode: |
Electrospray + |
|
Data: |
Centroid |
|
Mass range: |
100-850 amu |
|
Scan time: |
10 min |
|
Cone voltage: |
15 |
|
Sampling frequency: |
5 Hz |
|
Capillary voltage: |
0.8 |
|
Probe temperature: |
600 |
|
Detector gain: |
1 |
|
Makeup solution: |
90:10 water:acetonitrile with 0.01% formic acid |
Note: Makeup solution is only used for preparative separations
MassLynx v4.1
FractionLynx Application Manager
Before beginning the isolation process, the AutoPurify method was configured in FractionLynx. The AutoPurify method defines the set of parameters that will be used for analyzing the crude sample, selecting the purification method, running the isolation protocol, and evaluating the collected fractions. In high throughput laboratories where there is broad sample diversity, the system administrator may choose to define multiple AutoPurify methods to adequately address the different types of molecules that require purification. For example, there may be a specific method for acids, bases, or hydrophobic molecules. While AutoPurify can be configured to run automatically from analytical crude sample screening and prep isolation to fraction analysis, the three stages may also be executed in a semi-automated fashion. The software can be programmed to run the analytical screening and generate the prep sample list. The user can then review the FractionLynx browser report, make changes to the purification strategy if desired, and then manually start the system to perform the compound isolation. In the same manner, the user may review the newly generated purification results in the browser report and edit the sample list before fraction analysis occurs. Thus, the chemist can interact with AutoPurify as much or as little as he deems necessary for a given set of samples. Detailed descriptions of the AutoPurify process have been communicated previously.2,3
Once the AutoPurify method was defined to perform the completely automatic purification process from analytical screening through fraction analysis, the synthetic crude mixture of the pharmaceutical intermediate was placed in the sample manager and the system was started. After completing the analysis of the crude material, the FractionLynx browser report showed that the sample was approximately 67% pure. According to the parameters defined in the AutoPurify method, the sample required purification. The software selected an appropriate gradient for purification (the focused gradient named Narrow B in Figures 1 and 2), and immediately started the isolation.
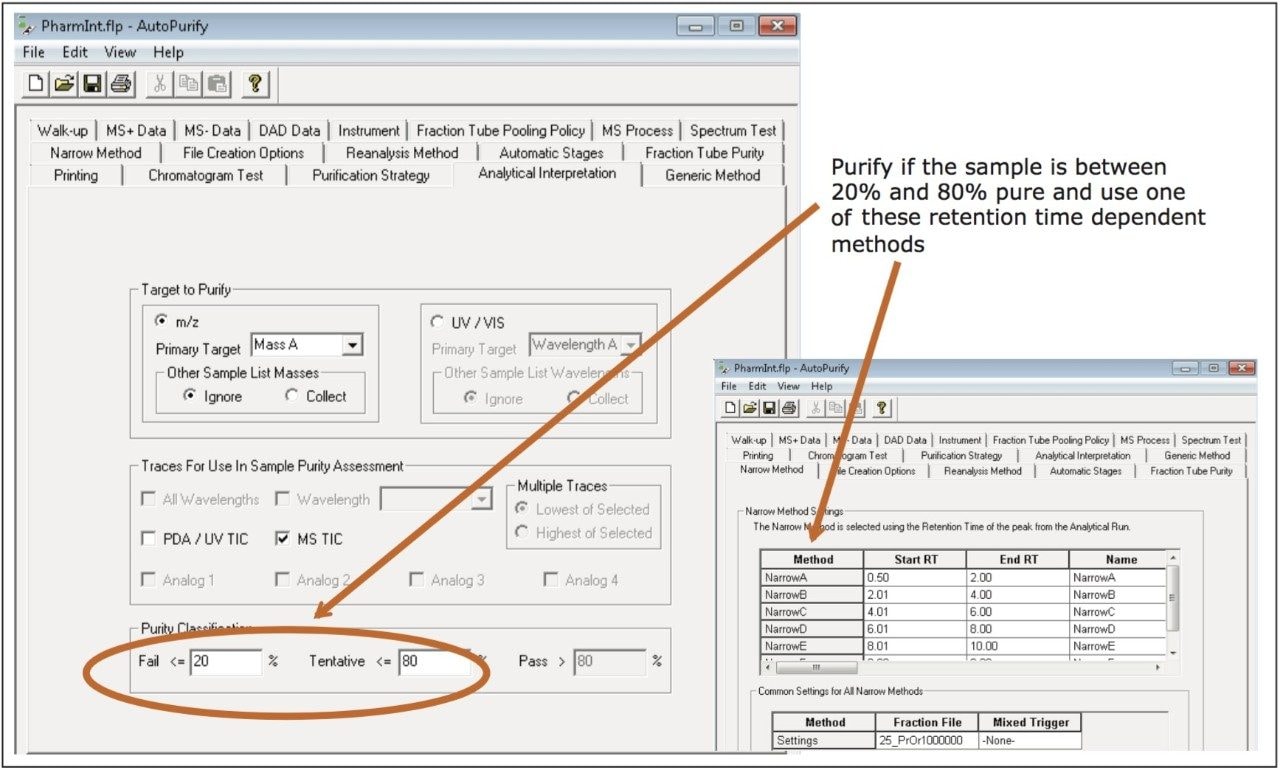
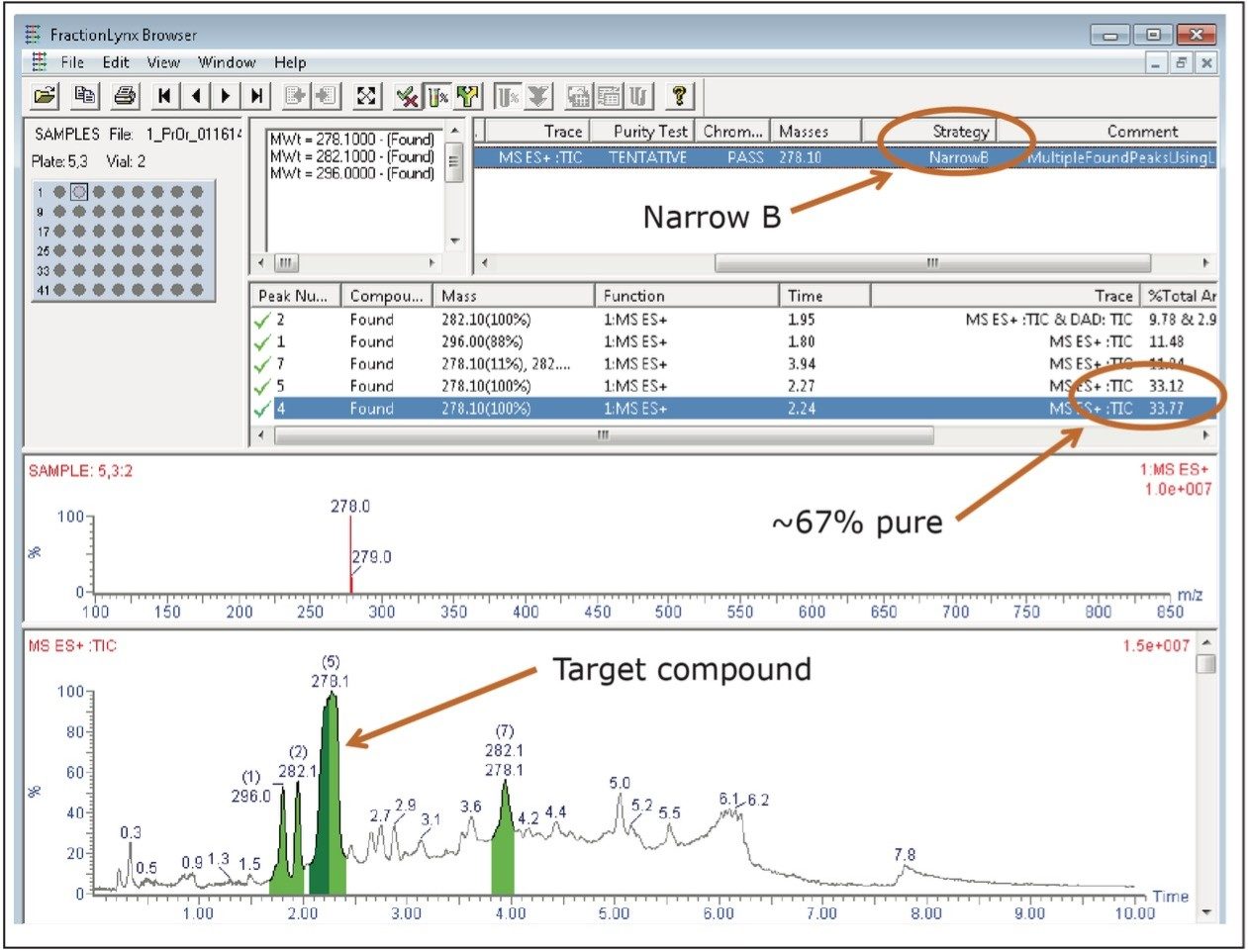
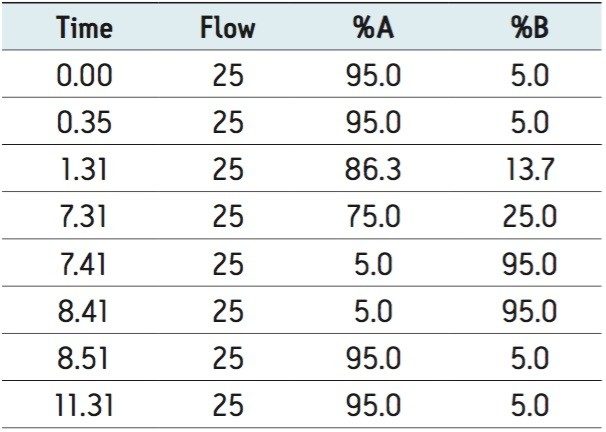
AutoPurify uses the retention time of the target peak to select one of the predefined narrow gradients. Narrow gradients, also known as focused gradients,4 are useful in preparative chromatography because they effectively increase the resolution between the target peak and its closely eluting neighbors without increasing run time. Increased resolution ultimately leads to higher loading and greater product purity. Figure 3 shows the preparative chromatography of the isolation using the Narrow B gradient method described in Table 1.
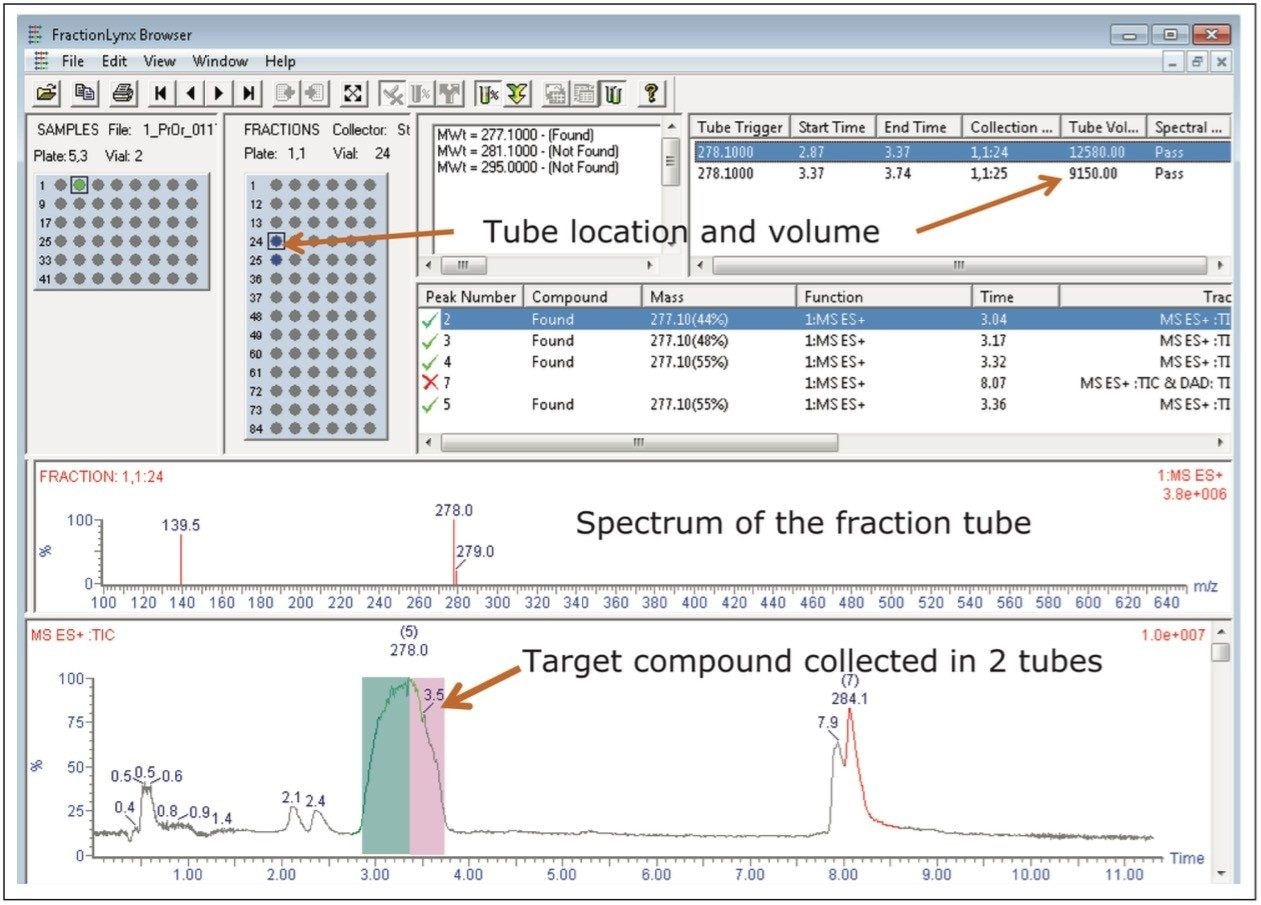
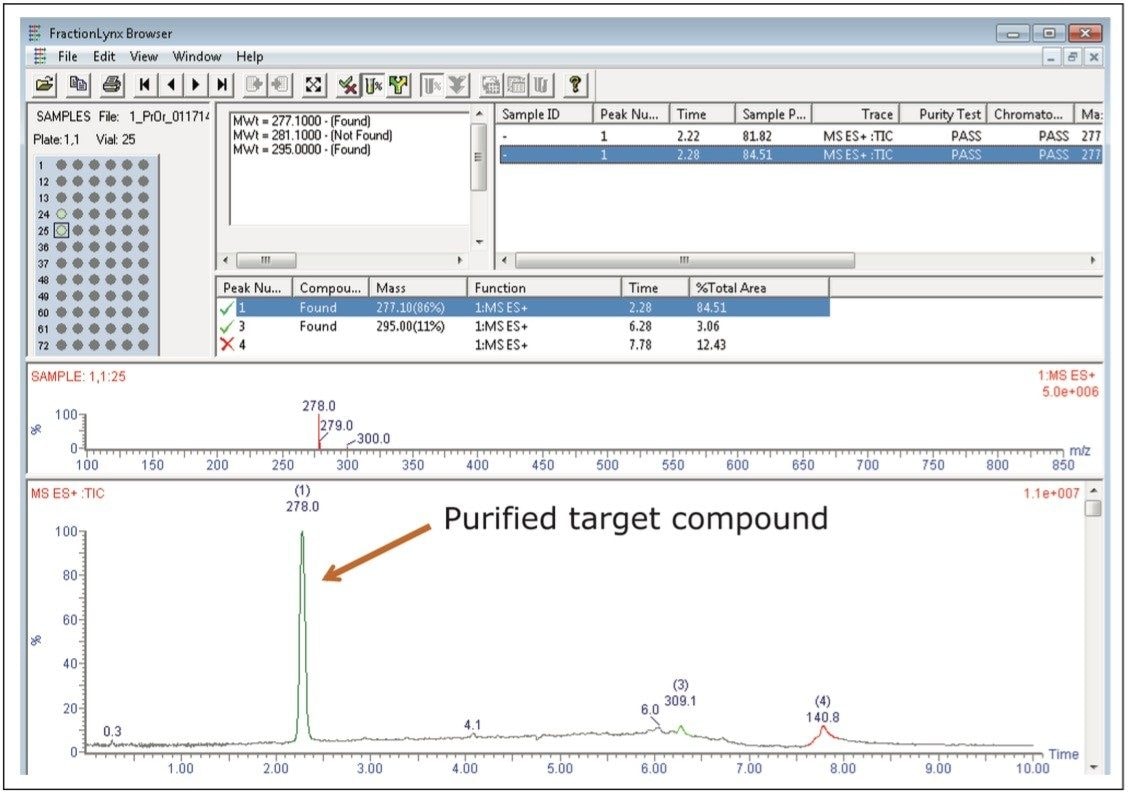
Without user intervention, analysis of the collected fractions was performed immediately following the completion of the preparative isolation. The analysis method used was the same one as the original screening gradient, running from 5–95%B in 6 minutes. The estimated purity of the fraction shown is 97%, as the peak in red is actually part of the column washout. The results of the fraction analysis are shown in Figure 4.
720005106, August 2014