
This is an Application Brief and does not contain a detailed Experimental section.
This application brief determines system characteristics of the ACQUITY UPLC I-Class System and the Xevo G2-S QTof combination, including resolution, sensitivity, and linear dynamic range based on seven commercially available pharmaceutical compounds in human plasma.
The sensitivity of the high resolution Xevo G2-S QTof approaches tandem quadrupole MS capabilities and demonstrates it to be fit-for-purpose for a large number of routine assays and analytical workflows.
Traditionally, qualitative and quantitative information for a given analyte are obtained using two separate mass spectrometer platforms, including a tandem quadrupole (also referred to as triple quadrupole, or QqQ) instrument for quantitative information, and time-of-flight or ion trap for qualitative information. Many researchers are evaluating high resolution MS (HRMS) platforms to see if they can generate comparable data to traditional QqQ technologies while simultaneously generating high quality qualitative data.
There are many advantages to such an approach. It can save sample volume by avoiding duplication of sample analysis on both QqQ and HRMS platforms, which also saves the analyst valuable time. Data acquisition using MSE (simultaneous acquisition of full scan and all fragment ions) by employing Waters QTof MS technologies is also available. This can provide valuable information on potential biotransformation pathways often present in in vivo samples at a very early stage. This leads to faster, more comprehensive, and improved decision making within DMPK groups.
The characteristics of HRMS instrumentation, including resolution, sensitivity, and linear dynamic range, are important factors in determining its utility in drug discovery and development processes. Here, the mass resolution, sensitivity, and linear dynamic range of multiple pharmaceutical compounds are analyzed in human plasma with the Xevo G2-S QTof. Compound structures with masses ranging from 260 to 530 are shown in Figure 1.
The seven compounds, analyzed as a mixture, were quantified using the ACQUITY UPLC I-Class System and Xevo G2-S QTof in ESI+ resolution mode with UNIFI Software for data acquisition and processing.
Figure 2 shows a representative chromatogram of the mixture at a concentration of 1.56 ng/mL. The concentration and % deviation data for two representative compounds, clopidogrel and buspirone, are summarized in Table 1 and data plotted in Figure 3.
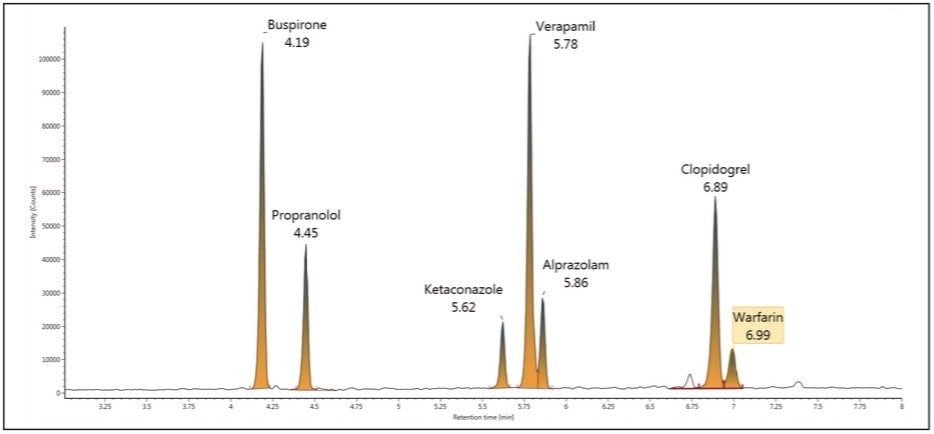
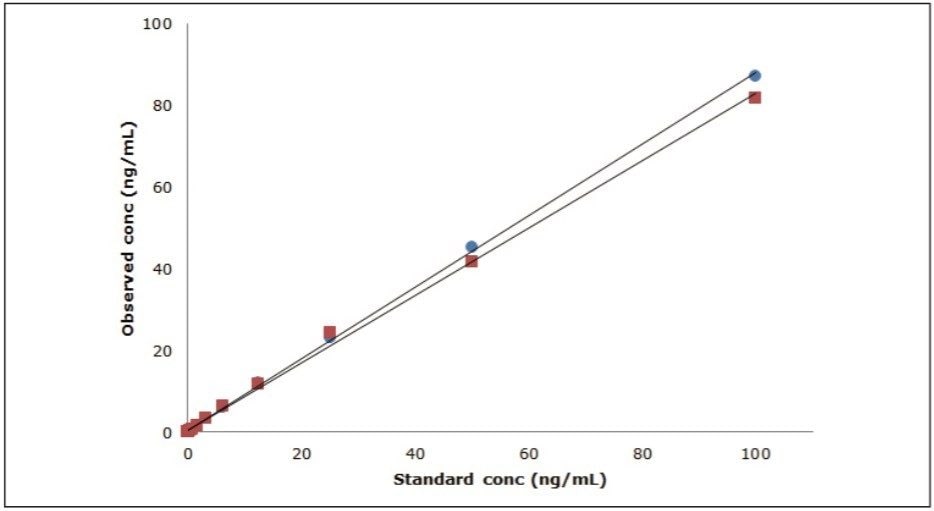
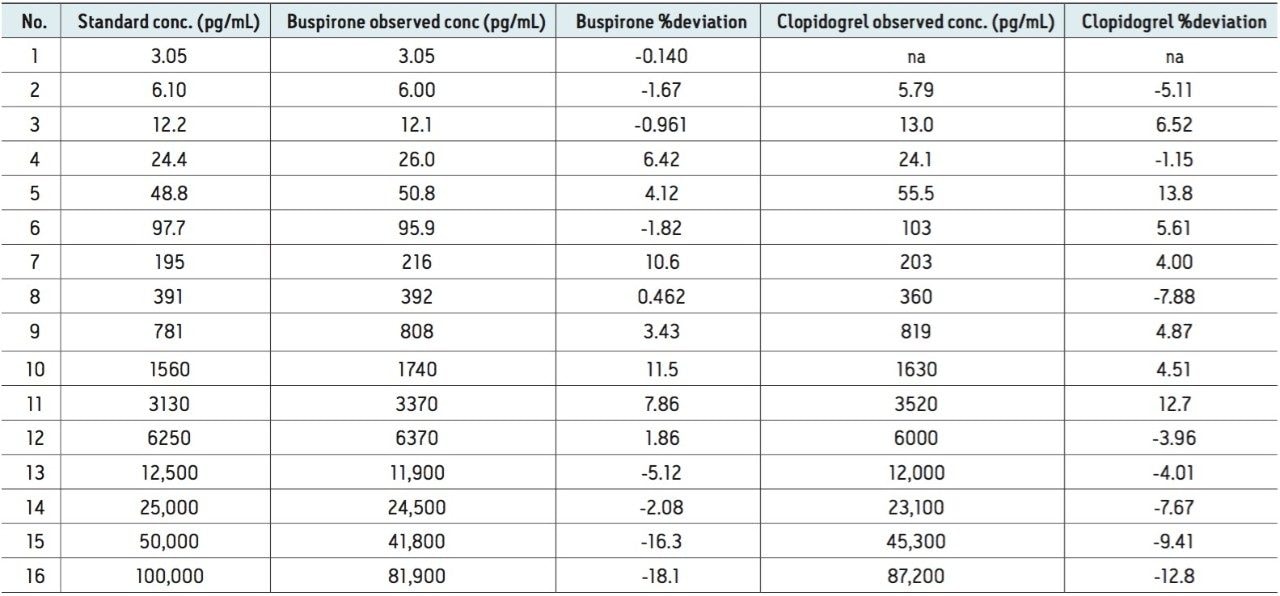
A compilation of data for all seven compounds in this study is shown in Table 2. The peak width at half height under UPLC conditions ranges from 1.3 seconds to 2.1 seconds. The measured mass resolution was excellent with compounds ranging from 29,000 to 40,000 (at a scan rate of 5 Hz) resolution across the entire mass range tested. A minimum linear dynamic range from 24.4 to 25,000 pg/mL, or 3.0 log units, was observed for all seven compounds. Range was compound-dependent with buspirone showing the highest linear dynamic range from 3.05 to 100,000 pg/mL or 4.5 log units. The lower detection limit was also compound-dependent, ranging from a low of 3.05 pg/mL for buspirone and propranolol to a high of 24.4 pg/mL for ketoconazole and warfarin.
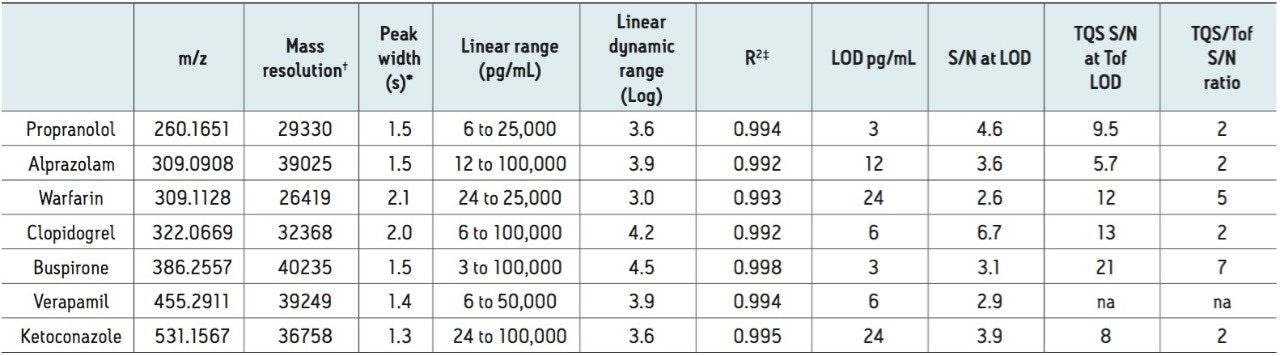
For purposes of comparison, the same samples were also analyzed by MRM using the Xevo TQ-S Mass Spectrometer. The relative sensitivity of the Xevo TQ-S compared with the Xevo G2-S QTof was then estimated, based on a comparison of the ratio of the signal-to-noise value at the QTof LOD concentration. As expected, the Xevo TQ-S is the more sensitive platform, but the HRMS provides compelling data with comparable LODs ranging from two- to sevenfold less sensitivity based on the S/N comparison.
The Xevo G2-S QTof is capable of quantifying small pharmaceutical compounds in biological matrices. Excellent resolution, sensitivity of 3 to 24 pg/mL, and excellent linear dynamic range of 3.0 to 4.5 were obtained. Compared to the Xevo TQ-S, already the most sensitive QqQ instrument on the market, the sensitivity of the Xevo G2-S QTof approaches tandem quadrupole MS capabilities and demonstrates to be fit-for-purpose for a large number of routine assays and analytical workflows.
When combined with UNIFI Software, an all-in-one powerful, flexible, and user-friendly interface for HRMS data collection, the ACQUITY UPLC I-Class and Xevo G2-S QTof systems will play a central role in meeting the challenges of modern drug discovery and development.
720004724, June 2013