
Two fast, selective, and sensitive methods have been developed for the analysis of furans and the passivator benzotriazole in transformer oil. One method utilizes the ACQUITY UPLC H-Class System coupled to the ACQUITY UPLC PDA Detector for the rapid four-minute analysis of furans alone. The second method utilizes the ACQUITY UPLC H-Class System coupled to Xevo TQ MS if furans and additional transformer oil additives are required, the passivator benzotriazole was considered within this application.
This application note illustrates increased sample throughput for the identification and quantification of furans and the passivator benzotriazole in transformer oil offering:
Transformer oil is usually a highly-refined mineral oil and due to its stability at high temperatures, it has excellent electrical insulating and heat transfer properties. Because of these properties transformer oil has many uses including: oil-filled transformers, high-voltage capacitors, fluorescent lamp ballasts, high-voltage switches, and in circuit breakers.
Furanic compounds can originate from the thermal depolymerization of cellulose solid insulation used within electrical equipment, which can then migrate into the transformer oil.
Electrical and mechanical stresses and the presence of water and oxygen increase the degree of cellulose degradation, eventually rendering the transformer ineffective. These changes can occur either due to normal aging or the presence of a fault.
Therefore an effective preventative maintenance program is critical to aid the planning of system servicing and, as a result, reduce costly instrument downtime. Periodic analysis of transformer oil for furans can be used to assess the degree of depolymerization of the cellulose, which does not require taking the unit out of service in order to take a sample.
Various additives can be incorporated into transformer oil to boost the quality of the oil, for either short or long term. All additives present need to be declared, therefore methodology to confirm the presence of declared additives, for example the passivator benzotriazole (BTA) – but also to detect undeclared additives, for example passivator Irgamet 301 – could be very valuable.
The presence of corrosive sulfur in transformer oil could potentially cause expensive power transformer failures. Passivators2 (metal deactivators), such as BTA, act as long term corrosion inhibitors when added to transformer oil. They react with reactive metal surfaces both on metal surfaces and with dissolved metals (copper and silver) therefore reducing the rate of reaction with other compounds in the oil such as corrosive sulfur. But there are passivators, for example Irgamet 30,1 that are not declared but are added to transformer oils to temporarily boost the oils quality to aid the passing of acceptance testing, therefore giving a false indication about the quality of the transformer oil.
Transformer oil analysis
|
LC system: |
ACQUITY UPLC H-Class |
|
Runtime: |
4.00 min |
|
Column: |
ACQUITY UPLC BEH C18 1.7 μm, 2.1 x 150 mm |
|
Column temp.: |
40 °C |
|
Sample temp.: |
10 °C |
|
Mobile phase A: |
Water:(0.1% formic acid) |
|
Mobile phase B: |
Acetonitrile:(0.1% formic acid) |
|
Flow rate: |
0.45 mL/min |
|
Injection volume: |
10.0 μL |
|
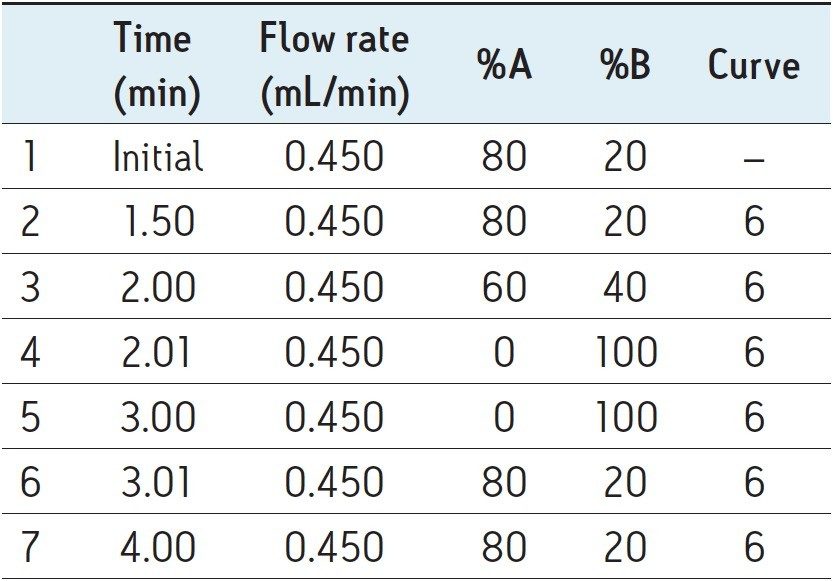
Mobile phase gradient is detailed in Table 1. |
|
UV system: |
ACQUITY UPLC PDA Detector |
|
Range: |
190 to 350 nm |
|
Resolution: |
1.2 nm |
|
Sampling rate: |
20 pts/sec |
|
Filter time constant: |
Normal |
|
MS system: |
Xevo TQ MS |
|
Ionization mode: |
APCI positive |
|
Corona current: |
20 μA |
|
Source temp.: |
150 °C |
|
APCI probe temp.: |
400 °C |
|
Desolvation gas: |
1000 L/hr |
|
Cone gas: |
100 L/hr |
|
Acquisition: |
Multiple Reaction Monitoring (MRM) |
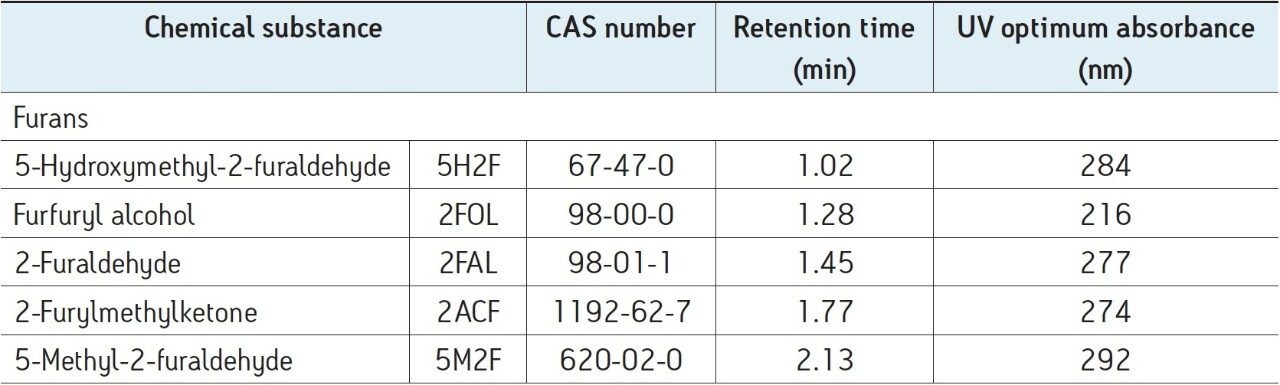
Optimum UV absorbances were established for each furan, detailed in Table 2, for the analysis of furans.
The standard method for the analysis of furanic compounds in electrical insulating liquids is ASTM D5837-12,3 which uses HPLC with UV detection. Other example methodologies for the analysis of furanic compounds include: UPLC with UV detection,4 HPLC analysis using various detection methods including ultraviolet (UV),5 mass spectrometry (MS),6 refractive index,5 spectrophotometric7 analysis, and gas chromatography-mass spectrometry (GC-MS).
Many previously used methods for furans consider only one furan (2-furaldehyde6) and lack reproducibility, selectivity, and sensitivity: they also require lengthy, costly, and time-consuming pre-treatments (SPE, concentration stages).
This application note describes two methods for the analysis of furans and the passivator benzotriazole in transformer oil. One method utilizes Waters ACQUITY UPLC H-Class System coupled to an ACQUITY UPLC PDA Detector for the rapid four-minute analysis of furans alone. The second method utilizes Waters ACQUITY UPLC H-Class System coupled to Xevo TQ MS if the passivator benzotriazole analysis is also required.
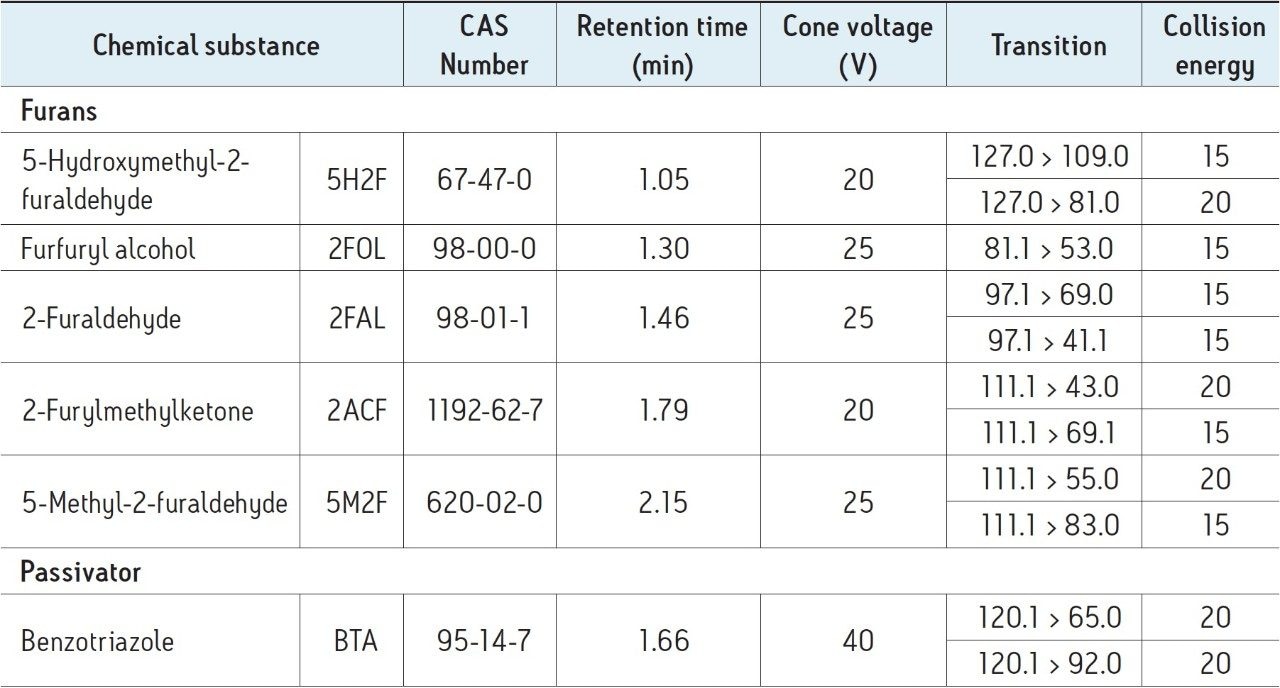
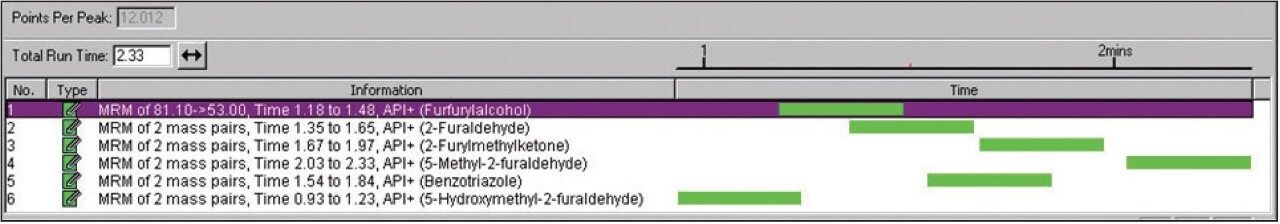
MS conditions were also optimized, detailed in Table 3, for the analysis of furans and the passivator benzotriazole. The established MRM method is illustrated in Figure 1.
Empower 3 Software was used for the UPLC-UV method.
MassLynx Software v.4.1 and TargetLynx Application Manager were used for the UPLC-MRM method.
The analysis of five furans was achieved using an ACQUITY UPLC H-Class System with an ACQUITY UPLC PDA Detector.
Optimum UPLC and UV conditions were developed, with the elution of all compounds within a four-minute run. When compared to the standard ASTM D5837-122 method with a minimum run time of 28 min, greater than seven times increase in sample throughput was achieved, with an excess of 88% less solvent usage.
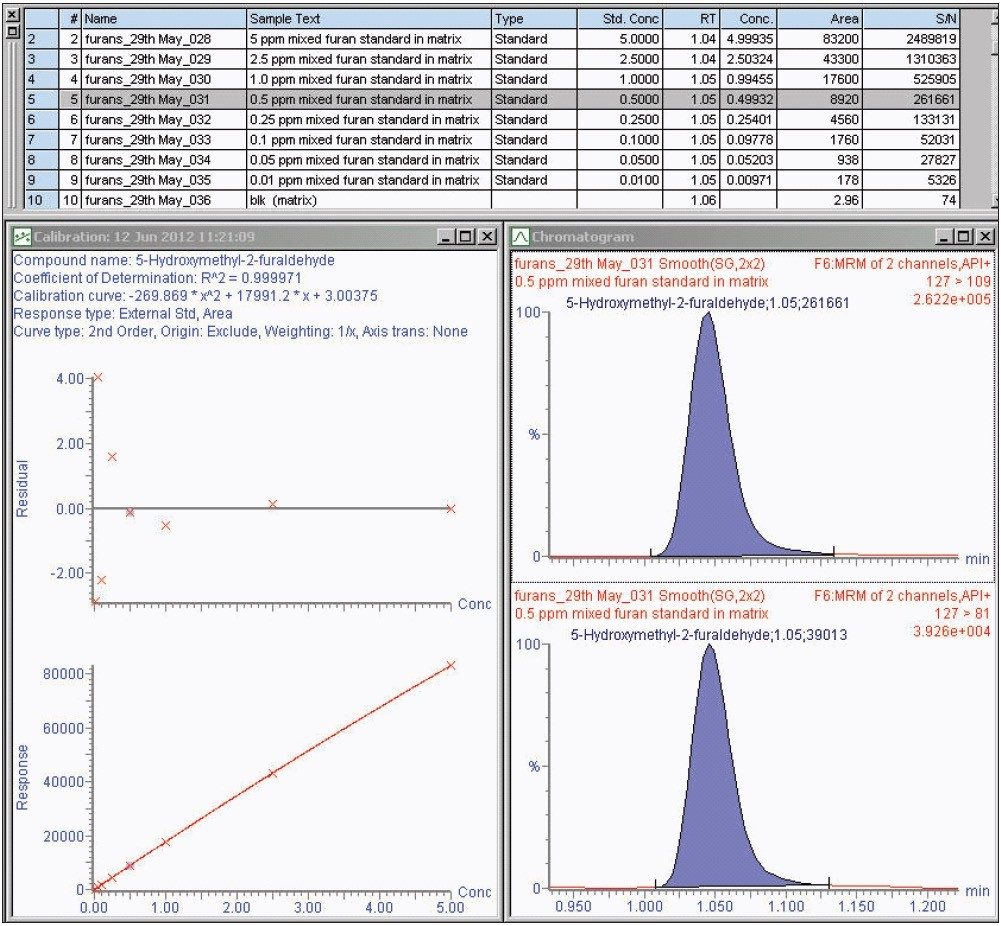
Matrix matched mixed calibration standards, 0.01 to 5.00 μg/mL, were prepared and analyzed for all the compounds considered (equivalent range of 2 to 100 mg/kg in transformer oil using the developed method).
The calibration curve results generated by Empower Software for 5-hydroxymethyl-2-furaldehyde are shown in Figure 2, and the UV chromatograms, including associated UV spectra for each compound, are shown in Figure 3.
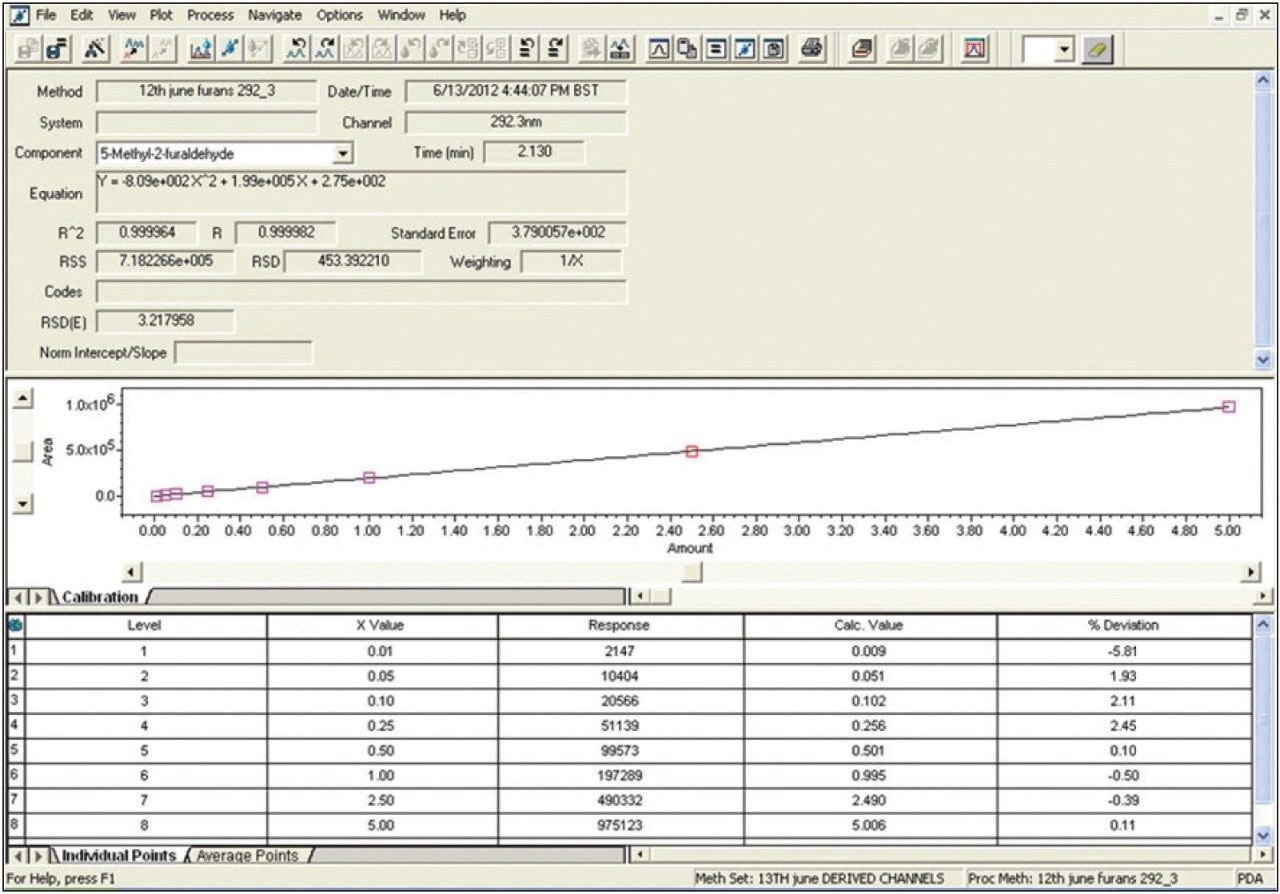
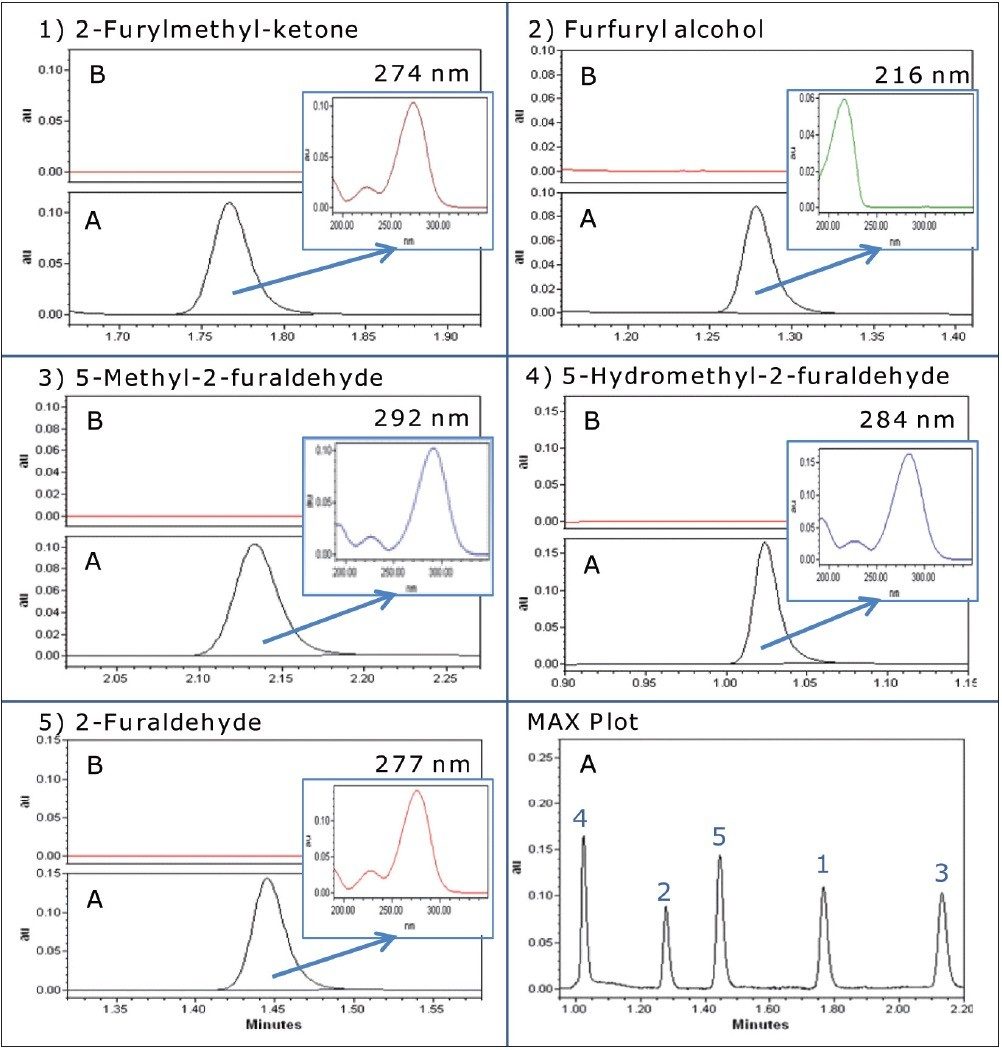
Using the transformer oil analysis extraction and instrument parameters as detailed, the furan results obtained for the analysis of spiked transformer oil are detailed in Table 4. Recoveries were obtained by comparing extracted spiked transformer oil against matrix matched calibration standards.
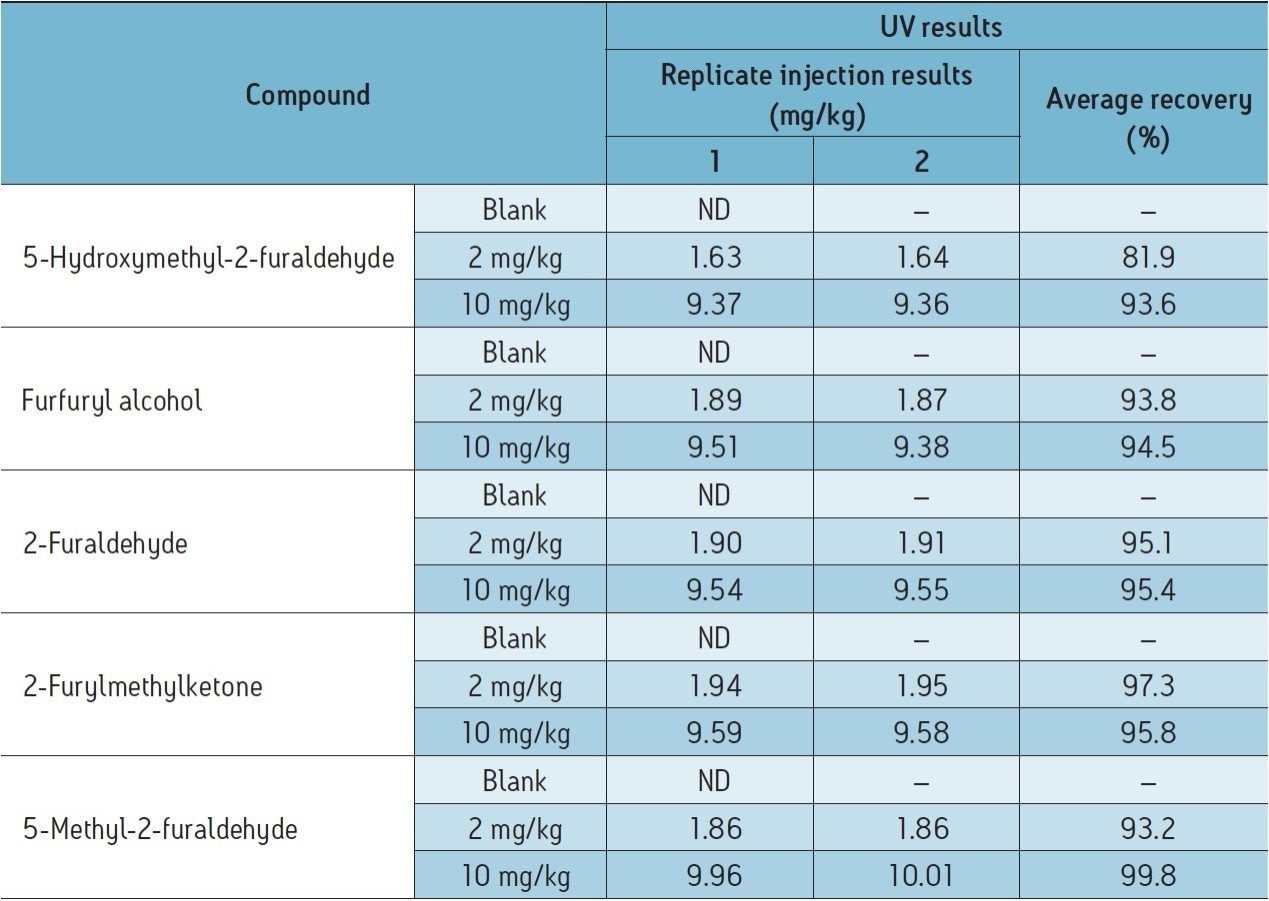
Efficient recoveries were obtained, ranging between 82% to 100% for two replicates, no evidence of matrix interference was observed.
The analysis of six compounds (five furans and the passivator benzotriazole) was achieved using Waters Xevo TQ MS in MRM mode with APCI ionization, coupled to an ACQUITY UPLC H-Class System.
Optimum UPLC and MRM conditions were developed, with the elution of all compounds within a four-minute run.
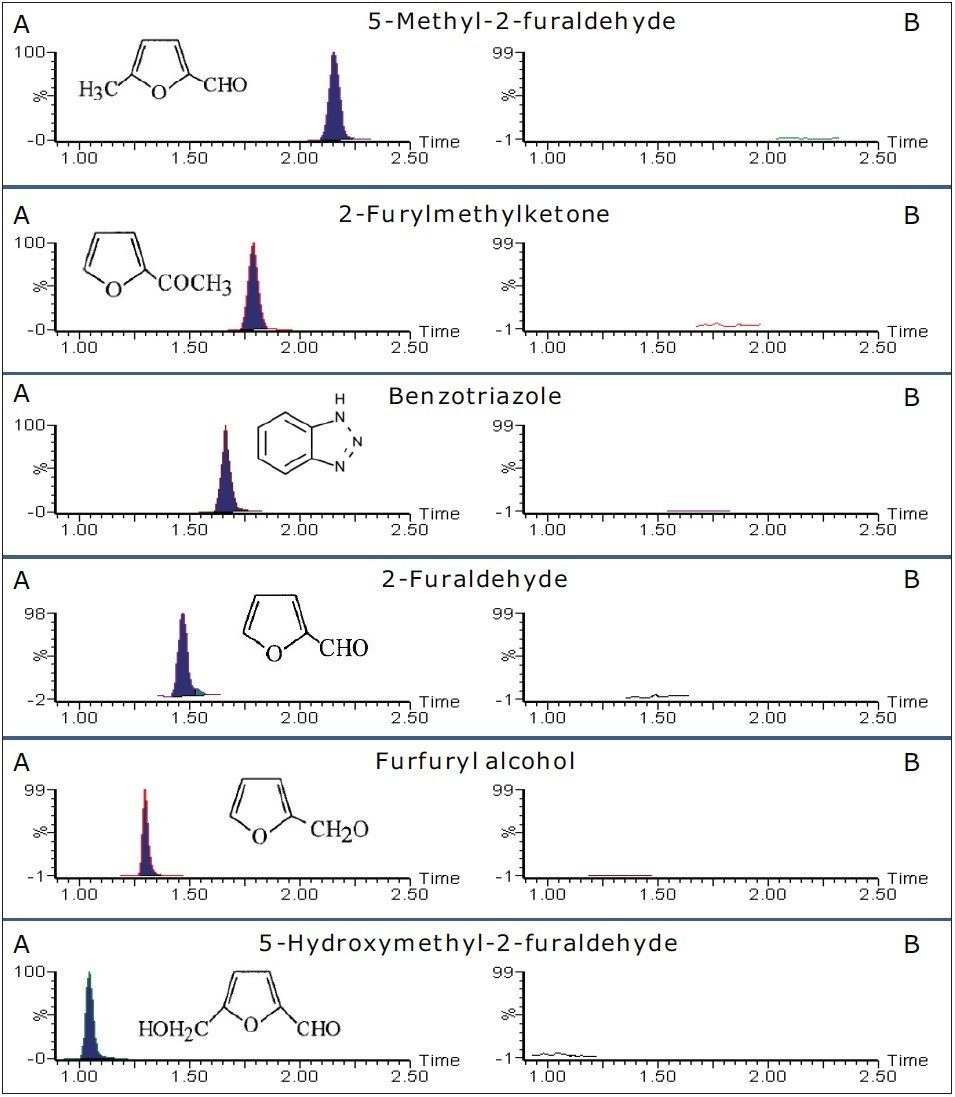
The MRM mass detection method detailed in Figure 1 was used after appropriate sample preparation to quantify for furans in transformer oil (containing furans and the passivator benzotriazole).
Using the transformer oil analysis extraction and instrument parameters as detailed, the furan results obtained for the analysis of spiked transformer oil are detailed in Table 5. Recoveries were obtained by comparing extracted spiked transformer oil against matrix matched calibration standards.
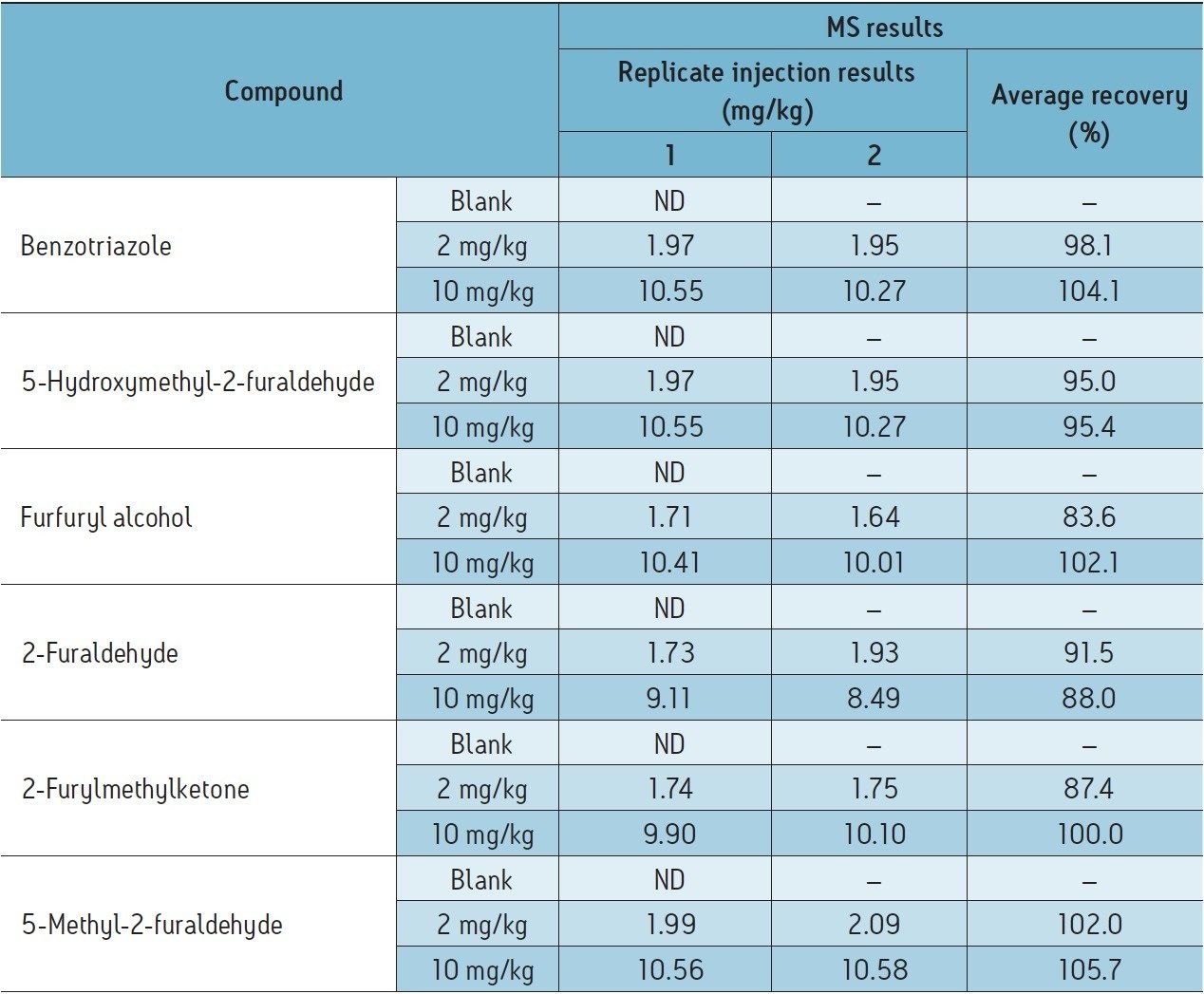
Efficient recoveries were obtained, ranging between 84% to 105% for the two replicates. Additional benefits include improved selectivity and sensitivity for the analysis of furans and transformer oil additives (the passivator benzotriazole was considered within this application) using the Waters Xevo TQ MS in MRM mode.
Matrix matched mixed calibration standards, 0.01 to 5.00 μg/mL, were prepared and analyzed for all the compounds considered (equivalent range of 2 to 100 mg/kg in transformer oil using the developed method). The TargetLynx Quantify results for 5-hydroxymethyl-2-furaldehyde are shown in Figure 4, and the MRM chromatograms for each compound are shown in Figure 5.
Two fast, selective, and sensitive methods have been developed for the analysis of furans and the passivator benzotriazole in transformer oil.
One method utilizes the ACQUITY UPLC H-Class System coupled to the ACQUITY UPLC PDA Detector for the rapid four-minute analysis of furans alone. The second method utilizes the ACQUITY UPLC H-Class System coupled to Xevo TQ MS if furans and additional transformer oil additives are required, the passivator benzotriazole was considered within this application.
The described approach offers many benefits when compared to the standard method:
APCI provides an efficient ionization mode for the analysis of the broad distribution of diverse polarities of the compounds being analyzed.
Quantitative furan analysis as detailed, using a small amount of insulating oil sampled from the transformer (without taking it out of operation), can be used as a representation of the degree of depolymerization and hence establish the condition of the transformer.
720004426, August 2012