
This is an Application Brief and does not contain a detailed Experimental section.
This application brief details selective extraction and enrichment of ingenol from a complex natural product matrix using Waters MV-10 ASFE System, enabling further method optimization for downstream chromatographic analysis and purification.
Supercritical fluid extraction (SFE), as done with the MV-10 ASFE System, is a highly efficient and specific method for extracting ingenol from the euphorbia plant latex.
Natural products have been a highly productive source of leads for drug discovery and development. Untapped biological resources, combined with technological advances in screening, separation, and synthesis, are driving the revival of new natural product drug discovery efforts.
One of the key steps in natural product research is to isolate bioactive compounds, which often exist at low concentrations and are inundated by complex sample matrices. This complexity presents a challenge for ensuing chromatography, especially preparative chromatography. As target compounds and matrices compete for binding sites on the column, the sample loading and purification throughput can be severely limited. A repetitive process is then required until the desired amount of material is accumulated. The process is often time-consuming and labor intensive.
Ingenol, shown in Figure 1, is a naturally occurring bioactive compound being developed as a potential therapeutic agent for Acquired Immune Deficiency Syndrome (AIDS). Current ingenol isolation involves the extraction of euphorbia plant latex using hexane, followed by chromatographic purification. The overall process suffers from extremely low yield arising from the sample complexity.
All SFE extractions were performed on a Waters MV-10 ASFE System. A total of 200 mg of latex pellets were loaded into a 5-mL vessel. The total flow rate was 5 g/min with 10% hexane used as the co-solvent.
Figures 2A and 2B show the SFC chromatograms of an ingenol standard and a latex extract using solvent extraction. Despite the strong MS response, the percentage of ingenol in the original sample was estimated to be less than 0.5% by weight. There is a striking difference between the solvent extract displayed in Figure 2B and the SFE extract shown in Figure 2C. In the SFE extract seen in Figure 2C, the majority of the peaks were removed after 4 min, presenting a much cleaner, simpler chromatogram. It is noted that the peaks in Figure 2C only correspond to a narrow co-solvent range in SFC (15% to 30%). Further method optimization with gradient focusing can be readily carried out to ensure the complete resolution of ingenol from the sample matrix. Furthermore, the ingenol content in the SFE extract was significantly enriched compared to the solvent extract; thus, enabling a more efficient chromatographic purification.
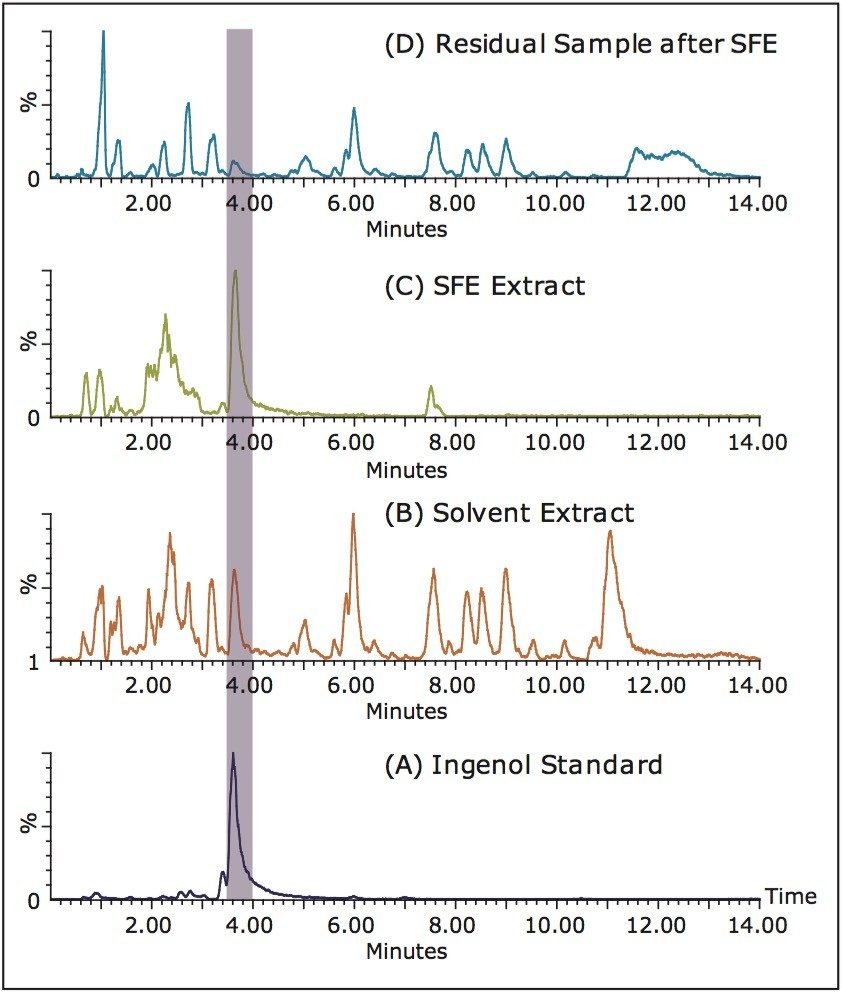
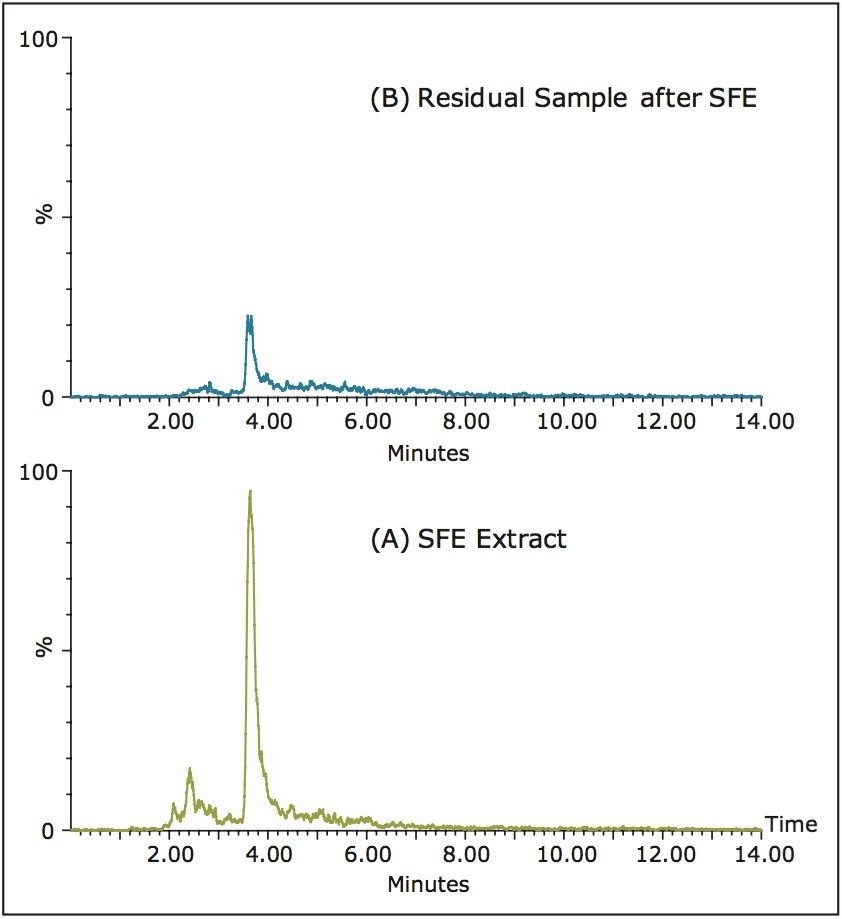
Figure 3 shows the normalized extracted ion chromatograms (XICs) (m/z=347, de-protonated ingenol) of the SFE extract and the residual sample after SFE extraction. After 1.5 hr dynamic extraction, the ingenol remaining in the vessel seen in Figure 3B was approximately 20% of those in the extract shown in Figure 3A. Prolonged extraction time can further improve the extraction efficiency.
Under carefully selected conditions, more than 80% of the ingenol present in the euphorbia plant latex was extracted by SFE. By comparing the SFC-MS chromatograms of the extracts from solvent extraction and SFE, it was demonstrated that SFE was much more specific and efficient than solvent extraction. As a result, the sample complexity was significantly alleviated, allowing for chromatographic method optimization with relative ease. The content of the target compound in the SFE extract was also enriched, enabling more efficient chromatographic purification of the target compound.
720004273, April 2012