This is an Application Brief and does not contain a detailed Experimental section.
Every aspect of the Xevo G2 QTof applies the design ethic of Engineered Simplicity to efficiently obtain the right results every time. Equipped with QuanTof technology, users obtain unheralded mass accuracy over a wide dynamic range of detection that leaves no room for doubt for biotherapeutic characterization studies.
With the biopharmaceutical pipeline expanding faster than the organizations supporting biotherapeutic development, researchers have to cope with biotherapeutic protein complexity in better ways. Large biomolecules with a complex array of major and minor product variants are being developed into therapeutic products for more recalcitrant diseases. Samples are therefore complex and scientists are expected to rapidly determine fine-grained detail over a wide dynamic range of variants.
It is vital for health and safety, and for regulatory compliance, that biotherapeutic product variation is accurately measured. In a peptide map, this means knowing mass accuracy will be predictable over the full dynamic range of component detection.
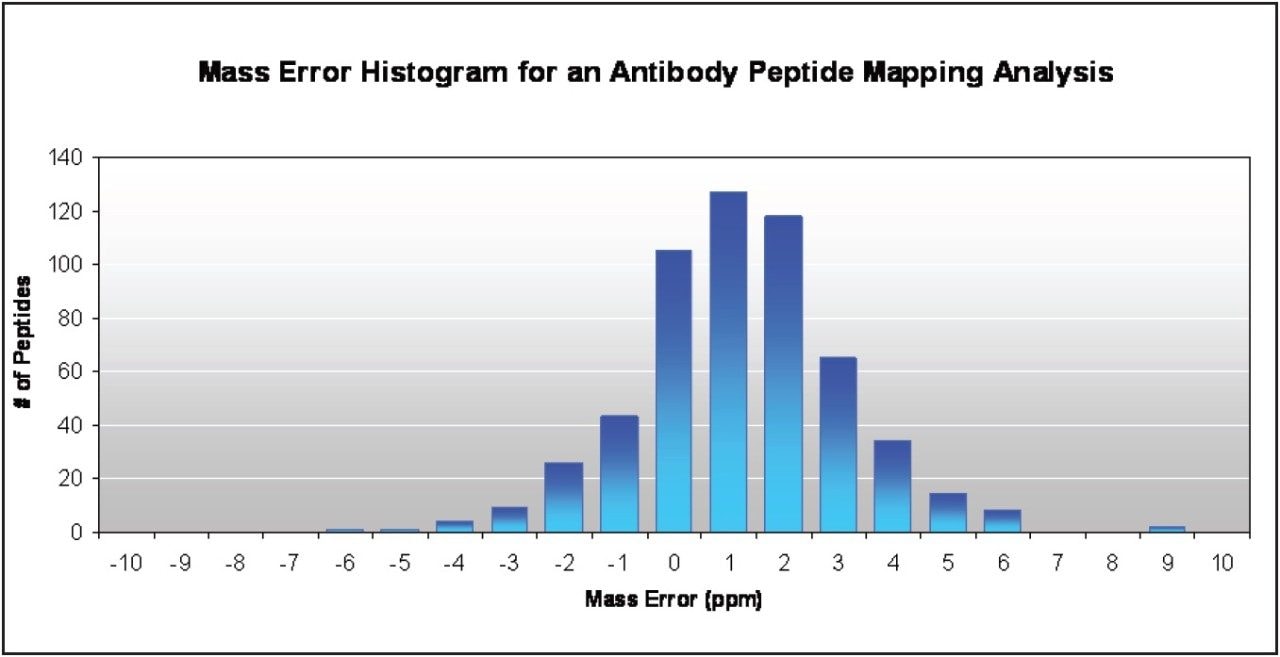
As demonstrated in Figure 1, mass accuracy has been established for a 148 kDa mAb on the Xevo G2 QTof by plotting the mass accuracy distribution for all 550+ antibody peptides assigned by BiopharmaLynx Software, an application manager for MassLynx Software, over a 90-min UPLC-MSE peptide map acquired in ESI positive resolution mode. The distribution of mass accuracy is narrow, with 89% of the peptides in a ± 3 ppm window, and 75% within 2 ppm. Detection of these tryptic, semi-tryptic, and modified peptides requires the full 4+ orders of dynamic range provided by the system’s QuanTof Technology MS detector.

In peptide maps, such a degree of routine mass accuracy for peptides spanning the highest and lowest masses delivered by a benchtop instrument redefines possibilities for routine applications. This level of performance, combined with the usability of both the Xevo G2 QTof System and BiopharmaLynx informatics, allows scientists who are not experts in mass spectrometry to know that their assignments are correct.
Figure 2 shows a histogram of the mass errors for a peptide map of a tryptically digested mAb of 148 kDa. The mass accuracies cluster around the 1 ppm error. The value of tight mass accuracy is that users will not need to re-check assignments, leaving them more productive in an organization testing for comparability where lot release is cost-critical.
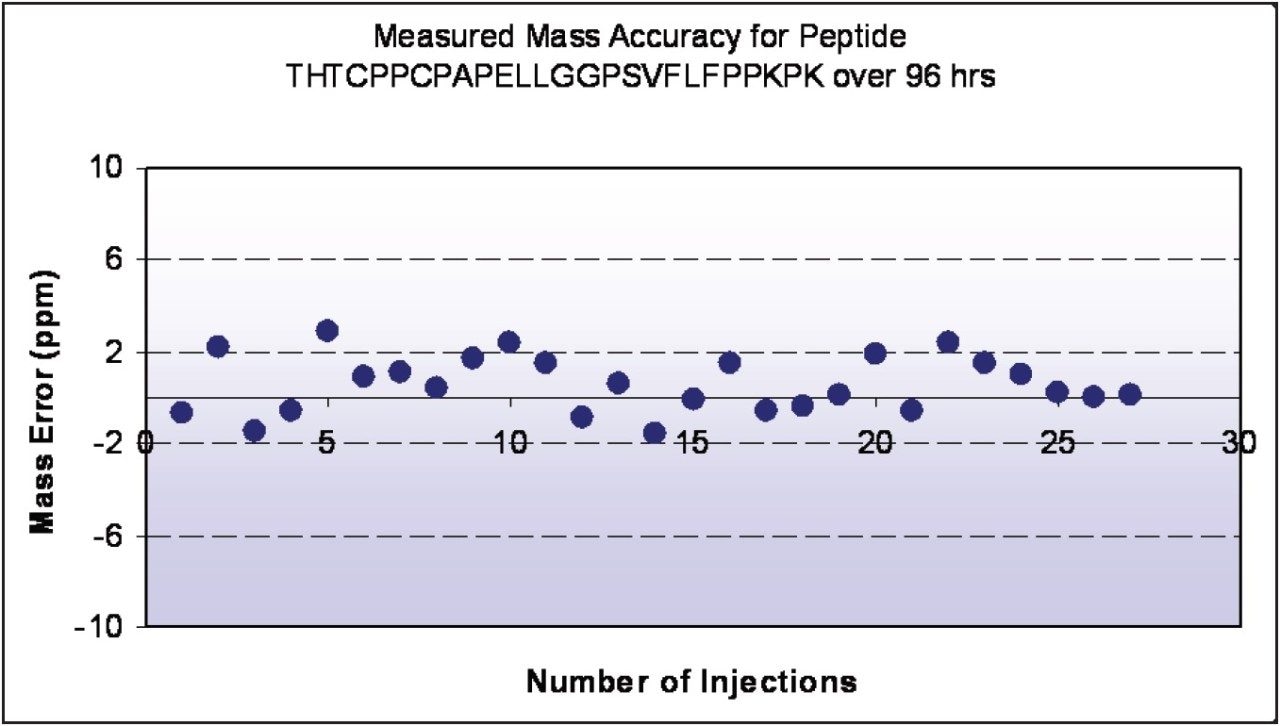
The Xevo G2 QTof maintains mass accuracy within 2 ppm of theoretical across days of experiment time. Figure 3 shows the narrow and consistent accuracy profile for a 2730 Da tryptic antibody peptide over 27 sequential injections (96 hours, or four days of continuous operation). Large peptide mapping studies can be accomplished with minimal effort for data acquisition, freeing scientists for higher-value activities.
The stability of mass accuracy demonstrated for the series of peptide maps is equally critical for studies of intact protein mass. Biotherapeutics organizations need to comply with regulatory authority guidelines to show that they consistently produce comparable biotherapeutics. Intact protein mass is measured as an overall indicator of protein identity and to monitor the glycoforms that account for some of the heterogeneity of the product.
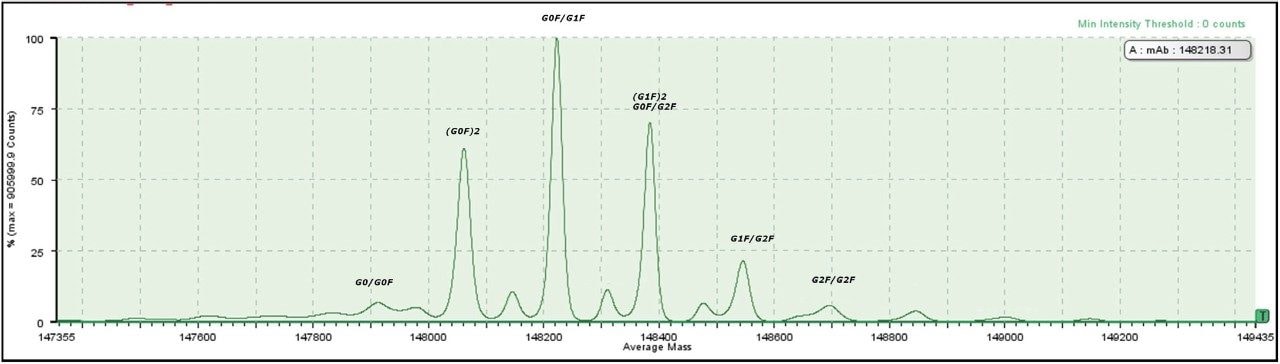
Figure 4 shows the deconvoluted MaxEnt1 Spectrum in the BiopharmaLynx browser with major and minor glycoforms. Six of the most intense peaks are labeled with their attributable glycoforms according to mass.
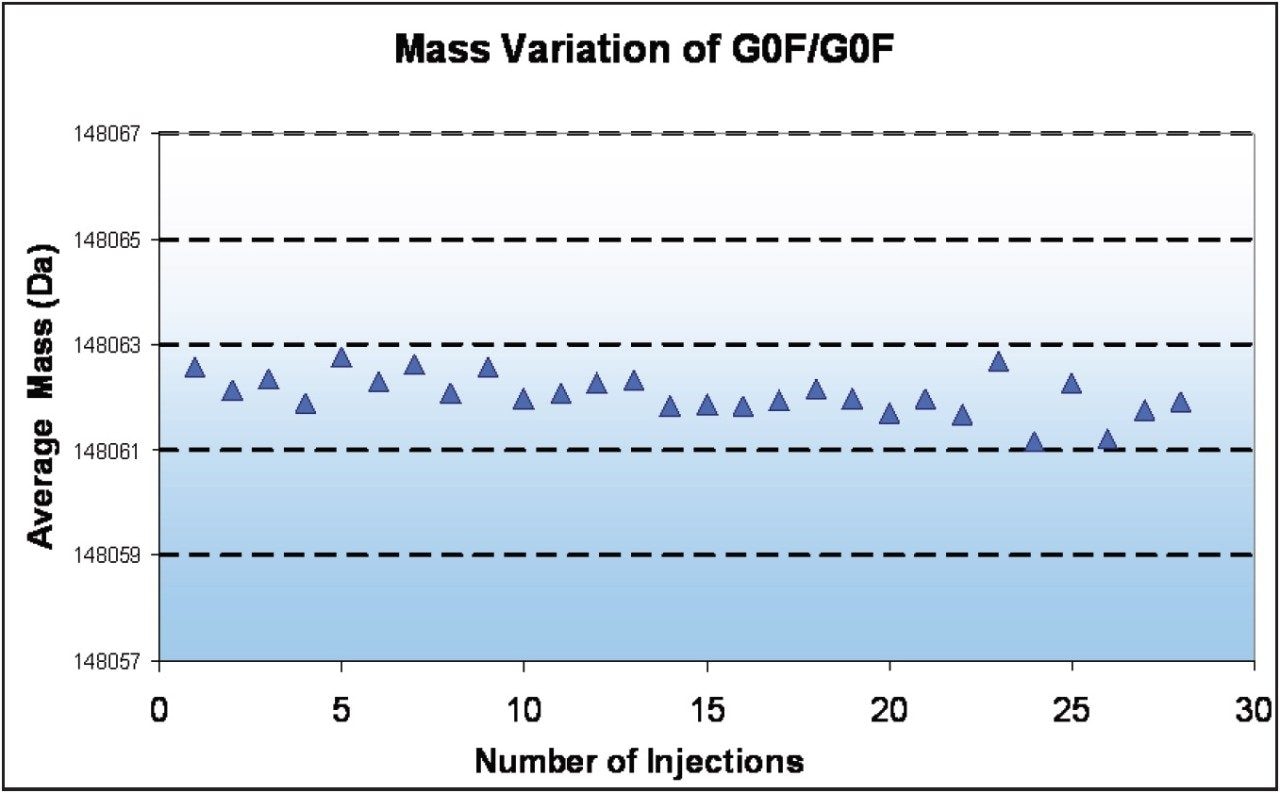
Repeated desalting LC-MS analyses of the same antibody using the Xevo G2 QTof produced MaxEnt1 deconvoluted spectral results showing a narrow range of mass variation (Figure 5). The G0F/G0F glycoform, with an average mass of 148,061 Da, varies within a narrow 2 Da window. Having the confidence of such consistently high mass precision for species even at the intact level allows biotherapeutic manufacturers to pick up quality and comparability issues with greater clarity.
The combination of Engineered Simplicity on the Xevo G2 QTof System equipped with QuanTof Technology provides consistently dependable mass accuracy at all mass ranges relevant to biotherapeutics. The mass accuracy is provided reproducibly over extended periods without intervention by the user, allowing an organization to make the most of its capital and human investment.
720003493, May 2010