This is an Application Brief and does not contain a detailed Experimental section.
The ACQUITY UPLC H-Class System is used for the systematic optimization of peptide mapping conditions in a fully automated protocol using four-solvent blending.
The ACQUITY UPLC H-Class System and its four-solvent blending capability enables the user to focus on the quality of the analysis so that reliable results can be obtained efficiently.
Peptide mapping is used to confirm the primary structure of a protein, identify post-translational modification (PTM), and analyze impurities. Any difference in structure of a protein is reflected in a change in retention time for the peptide containing the modification.
Complete structural characterization of a protein requires resolution of 100 or more peptides that span a wide range of size and molecular properties. The experiments to develop the required separation conditions include changes in the mobile phase modifier and organic solvent composition. Each condition tested requires the preparation of a pair of mixed solvents involving two or three exact measurements.
With the four-solvent blending of the ACQUITY UPLC H-Class System, these solvent mixtures are generated by the instrument from bottles of pure solvent. In this way the labor of preparing many solvent mixtures is reduced, and the possibility of error is minimized. Intermediate compositions can also be easily tested.
The ACQUITY UPLC H-Class System simplifies the development of reliable peptide maps that can be used to characterize the structure of a protein.
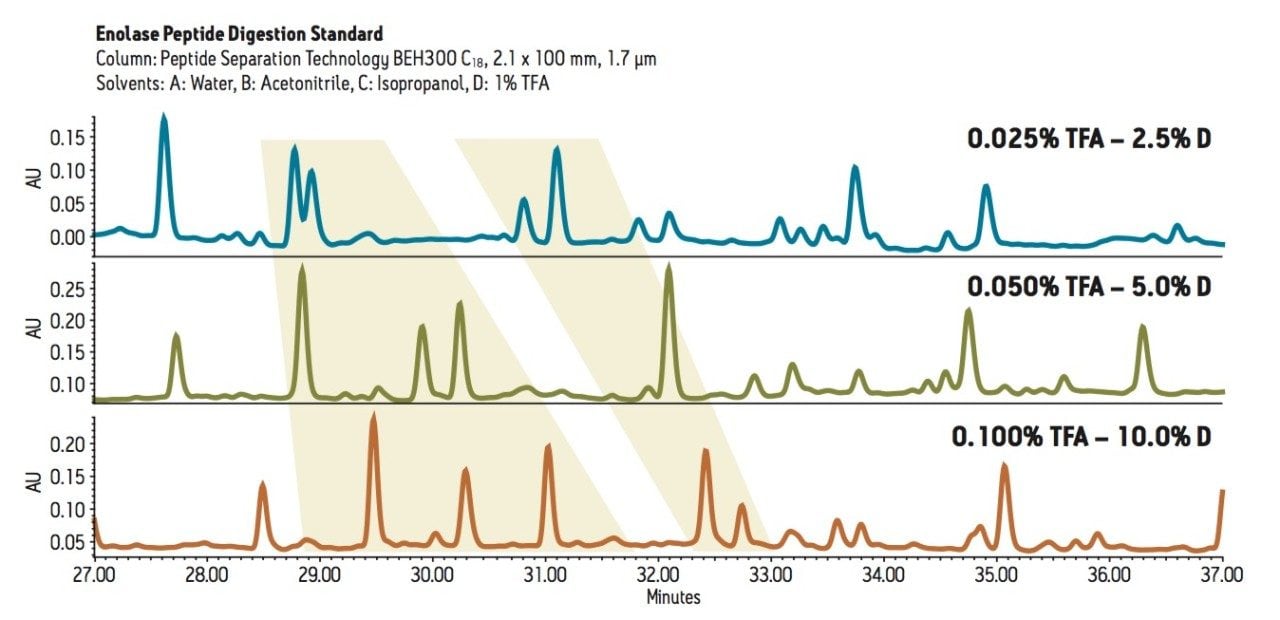
MassPREP Enolase Digestion Standards were separated on a Peptide Separation Technology ACQUITY UPLC BEH 300 C18 Column with various mobile phase combinations. The ACQUITY UPLC H-Class System was configured with water as Solvent A, acetonitrile as Solvent B, isopropanol as Solvent C, and 1% TFA in water as Solvent D. To assess the effect of acid concentration, three identical gradients of increasing acetonitrile were run with 2.5%, 5%, and 10% D, respectively, corresponding to 0.025%, 0.05%, and 0.1% TFA.
The chromatograms in Figure 1 show significant changes in selectivity as a function of acid concentration in the mobile phase. The experiment can be extended by repeating the same experiments but using a gradient of increasing isopropanol. This is conveniently performed by running the gradient from Solvent A (water) to Solvent C (isopropanol) with the same constant percentage of D as in the previous experiments. The change from acetonitrile to isopropanol reduces retention while also giving substantial changes in selectivity.
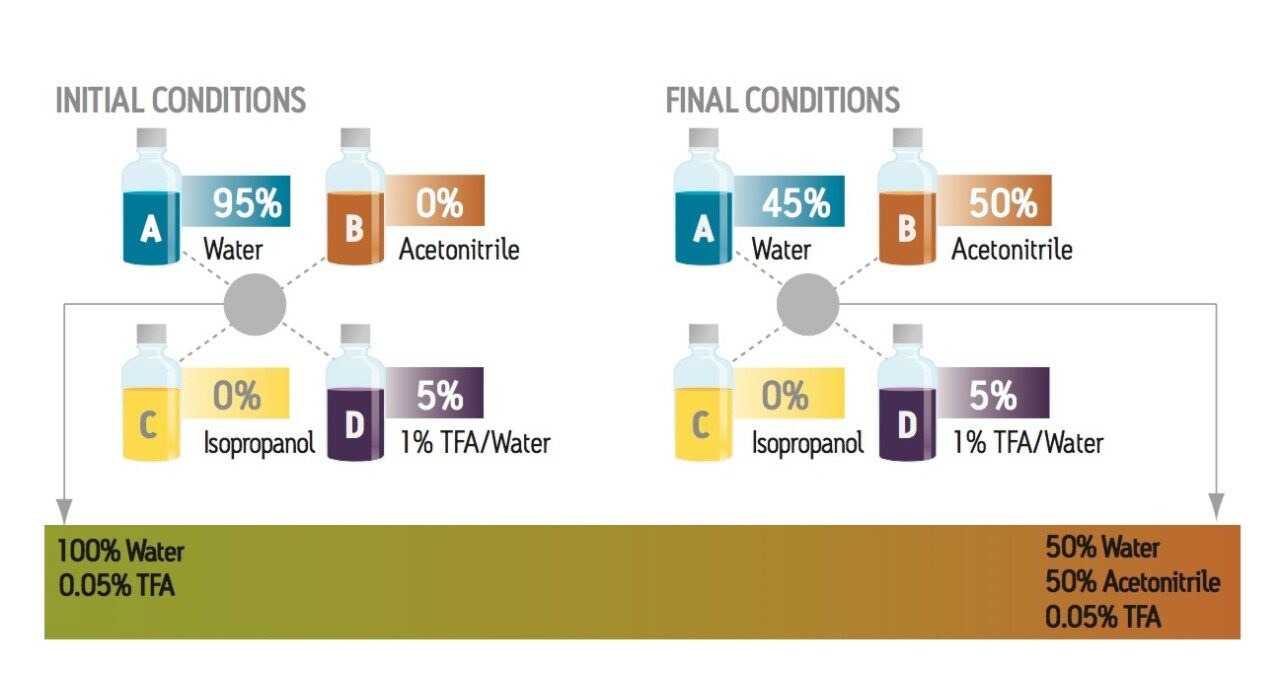
This set of experiments evaluates six different mobile phase formulations to identify the optimum peptide map. The changes in selectivity can be used to obtain the best resolution of the critical peptides in the mixture. To perform these experiments with a binary system or with a switching valve, the scientist would need to prepare 12 bottles of mobile phase with at least 24 volume measurements. With the ACQUITY UPLC H-Class System, only one mixture was used, with two measured volumes, in conjunction with three bottles of pure solvents (Figure 2). The ACQUITY UPLC H-Class System makes it possible to reduce the amount of work, time, and potential for error in reaching the optimum separation conditions.
Biochemists can use the ACQUITY UPLC H-Class System to efficiently develop peptide maps for protein characterization. The system’s easy-to-use four-solvent blending capability enables the user to focus on the quality of the analysis so that complete, reliable separations can be developed efficiently. The ACQUITY UPLC H-Class System combines UPLC separation principles with flexible instrument operation to provide the best possible results for bioseparations.
720003289, January 2010