This application note describes a rapid solution for the simultaneous analysis of 10 water soluble vitamin compounds commonly used to fortify food and beverage products, along with caffeine and six typically used foods dyes. This solution offers businesses a faster analysis and shorter sample turnaround time of 7.5 min, compared with traditional HPLC methods that can run up to 30 min for the analysis of a selection of food dyes, or an 18 min HPLC runtime for the analysis of vitamin B6 (pyridoxine) alone.
A rapid and simple method that enables the simultaneous analysis of 10 watersoluble vitamin compounds, along with caffeine, and six common food dyes, using a water/methanol gradient in one 7.5 min run.
Vitamin fortification of food and beverage products is an increasing trend, which has come about due to the poor levels of nutrients in many people’s diets. Manufacturers add vitamins to a wide range of products, and they use food dyes to make their products look more appealing to consumers. One of the fastest growing markets is the energy or health/nutritional beverages market, with data showing that energy drink manufacturers released one of every three new drinks developed in 2006.1 These range from off-the-shelf “sports drinks”, readily located in the refrigerated section of local markets, to specially formulated health and energy preparations, often pur-chased from specialty suppliers as powders that dissolve in water.
Food and beverage manufacturers are required to clearly label their products, indicating any vitamin compounds, colorings, and other additives that have been included. Manufacturers must comply with strict legal requirements, such as European Regulation (EC) No. 1925/20062 regarding the addition of vitamins and minerals to foods; or Title 21 of the U.S. Code of Federal Regulations (CFR) Part 101 – Food Labeling3 and Part 104 – Nutritional Quality Guidelines for Foods.4 Once a product has been formulated, food and beverage manufacturers require rapid, reliable, and cost-effective methods to analyze the nutritional content of their products to ensure that their label claims can be substantiated.
Currently water-soluble vitamin compounds and food dyes are analyzed individually, or in small groups, using a wide range of differ-ent analytical methods, such as microbiological assays, colorimetric analysis, titrimetric procedures, fluorimetric analysis, and HPLC methodologies.5,6 This means that analytical laboratories encounter a substantial financial outlay on several different types of instruments to facilitate multi-component analysis, as well as a significant invest-ment in personnel time and effort if a series of analyses are to be carried out. The ability to analyze water-soluble vitamin compounds and food dyes simultaneously using a single fast and easy solution provides businesses with the potential to improve productivity and increase revenue, resulting in enhanced efficiency, faster sample turnover, and reduced labor and training costs.
This application note describes a rapid solution for the simultaneous analysis of 10 water-soluble vitamin compounds commonly used to fortify food and beverage products, along with caffeine and six typically used foods dyes. This solution offers businesses a faster analysis and shorter sample turnaround time of 7.5 min, compared with traditional HPLC methods that can run up to 30 min for the analysis of a selection of food dyes,6 or an 18 min HPLC runtime for the analysis of vitamin B6 (pyridoxine) alone.7
Throughout the preparation and analyses, all solutions were protected from exposure to light and stored at < 5 °C.
The analysis of 10 water-soluble vitamin compounds, along with caf-feine and six common food dyes, was accomplished using ACQUITY UPLC with ACQUITY-extended λ PDA detection. The increased speed and enhanced resolution offered by ACQUITY UPLC enabled the elution of all compounds within 7 min. The resolution of all analyte peaks was sufficient to allow reliable quantitation, which would typi-cally not be possible with traditional HPLC in such a short runtime.
Figure 1 shows a chromatogram for solvent standards at concentra-tions of approximately 5 ng/µL. The accompanying information for Figure 1 is shown in Table 1, with compound names and expected retention times indicated.
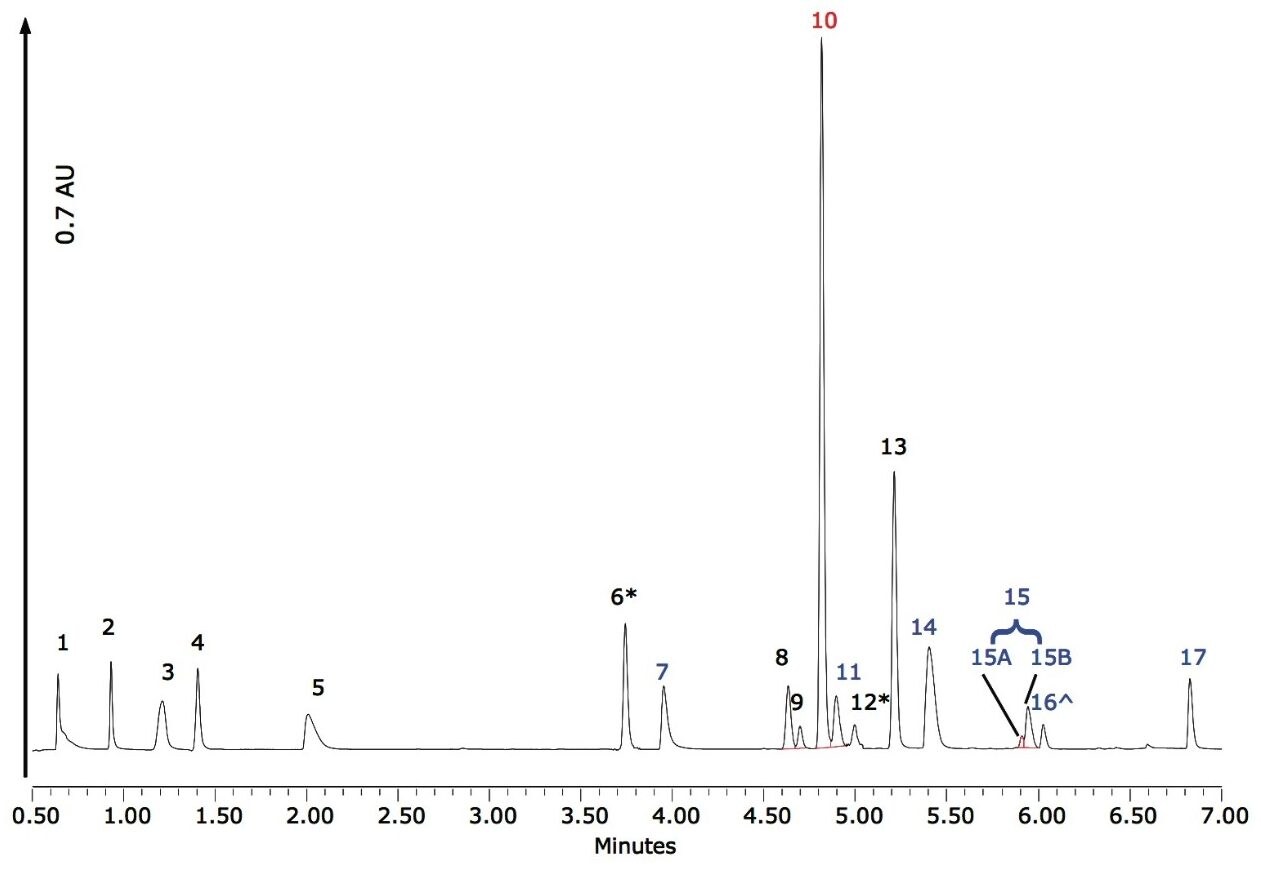
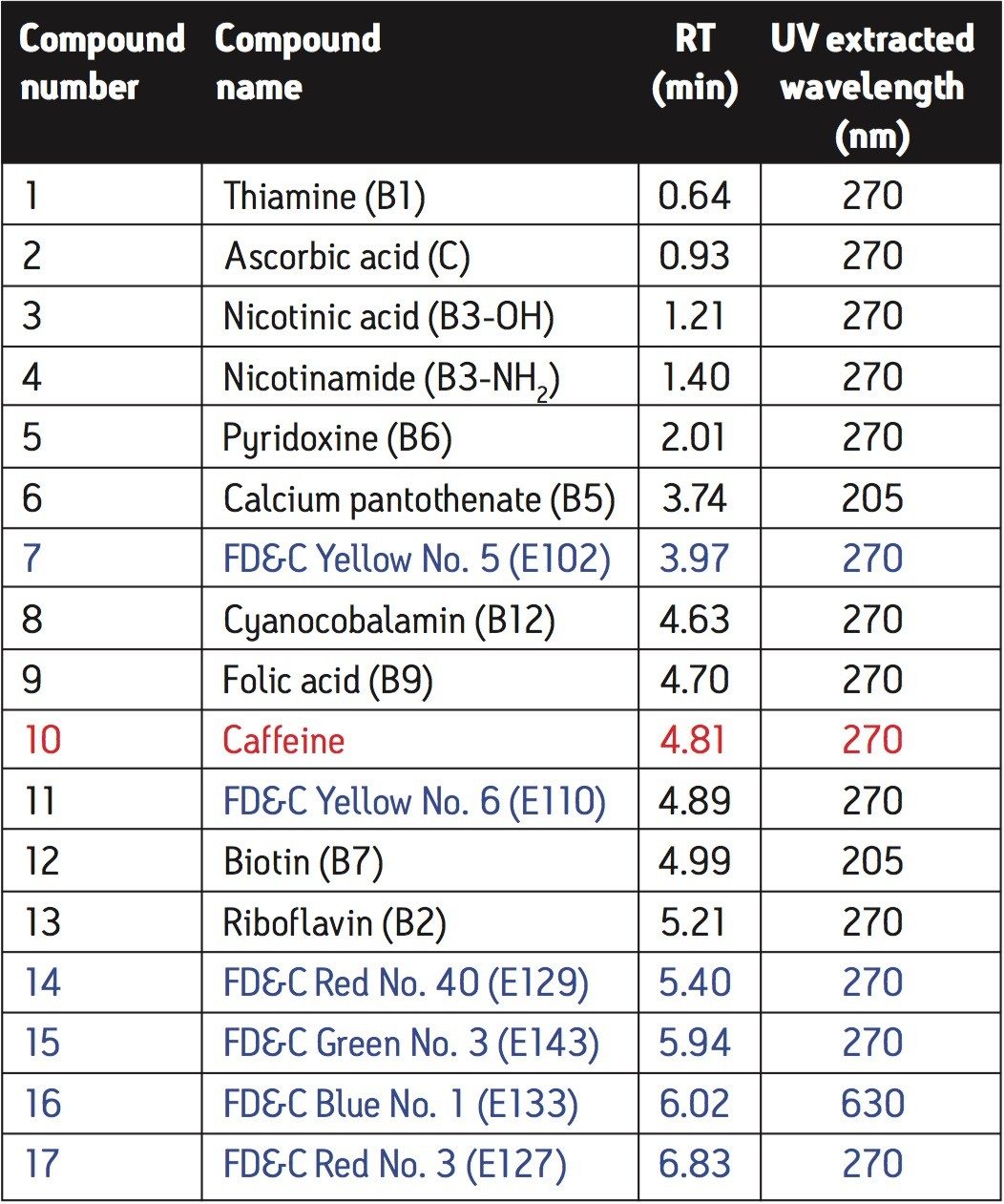
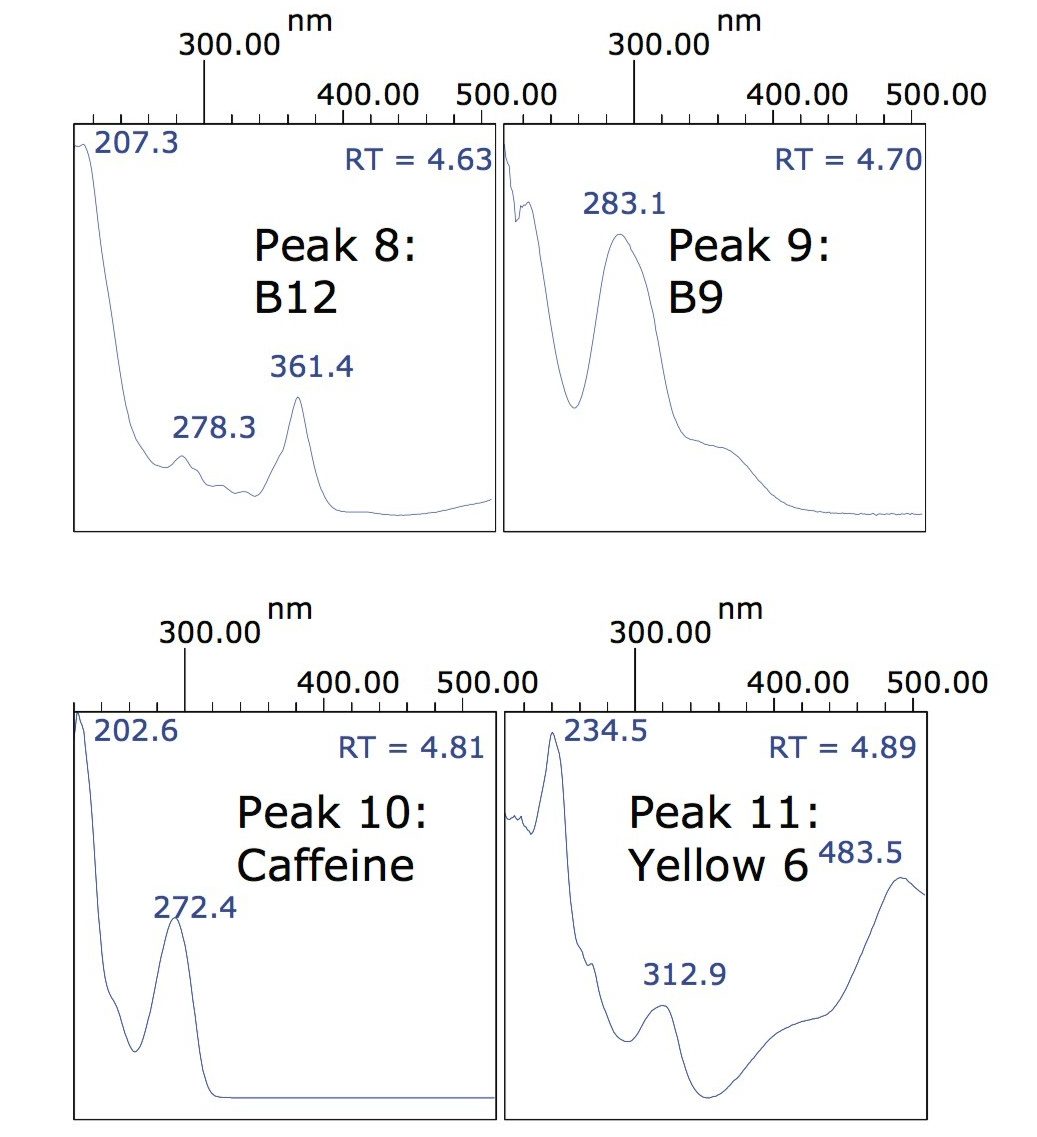
From Figure 1 it can be observed that peaks 8 and 9, as well as 10 and 11, elute close to each other; however, they are easily resolved on examination of their associated spectra. Figure 2 illustrates the differences between each pair of spectra.
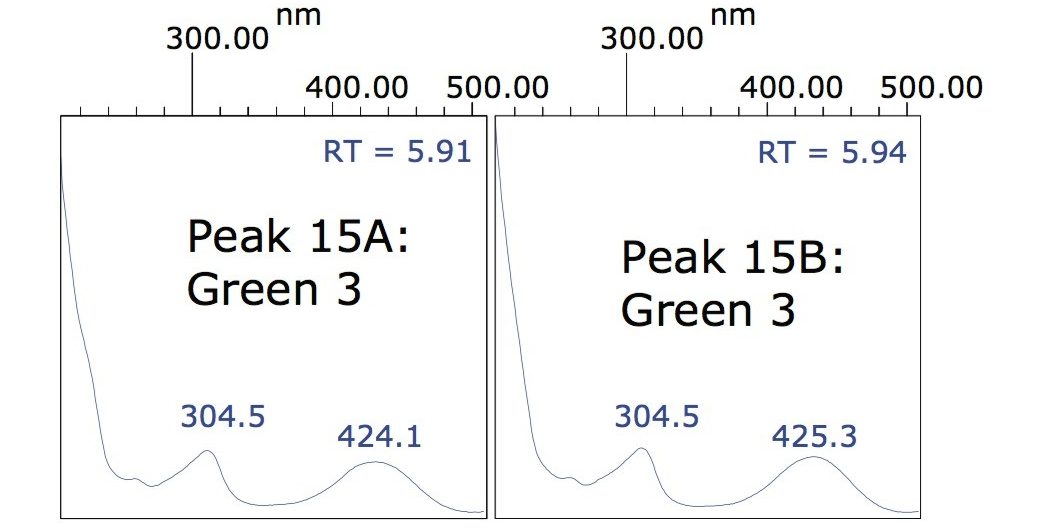
In addition, peak 15 was observed to have an accompanying shoulder (15A and 15B shown in Figure 1). In this case, review of the spectra indicated that these two components were identical, demonstrating that the main peak and the shoulder are isomers of the same dye. Empower 2 Software incorporates a powerful tool for the analysis of peaks, such as peak 15. With Empower, it is possible to label such peaks as Group Peaks enabling the software to treat multiple peaks, or peaks with shoulders, as one entity and integrate and quantify them accordingly. Figure 3 shows the spectra for peaks 15A and 15B.
These conditions were then used for the analysis of a vitamin-enriched, lemon-lime flavor fruit drink, purchased from a local grocery store. The drink was labeled as containing vitamins B3, B5, B6, and C, along with the food colors Yellow No. 5, and Blue No. 1.
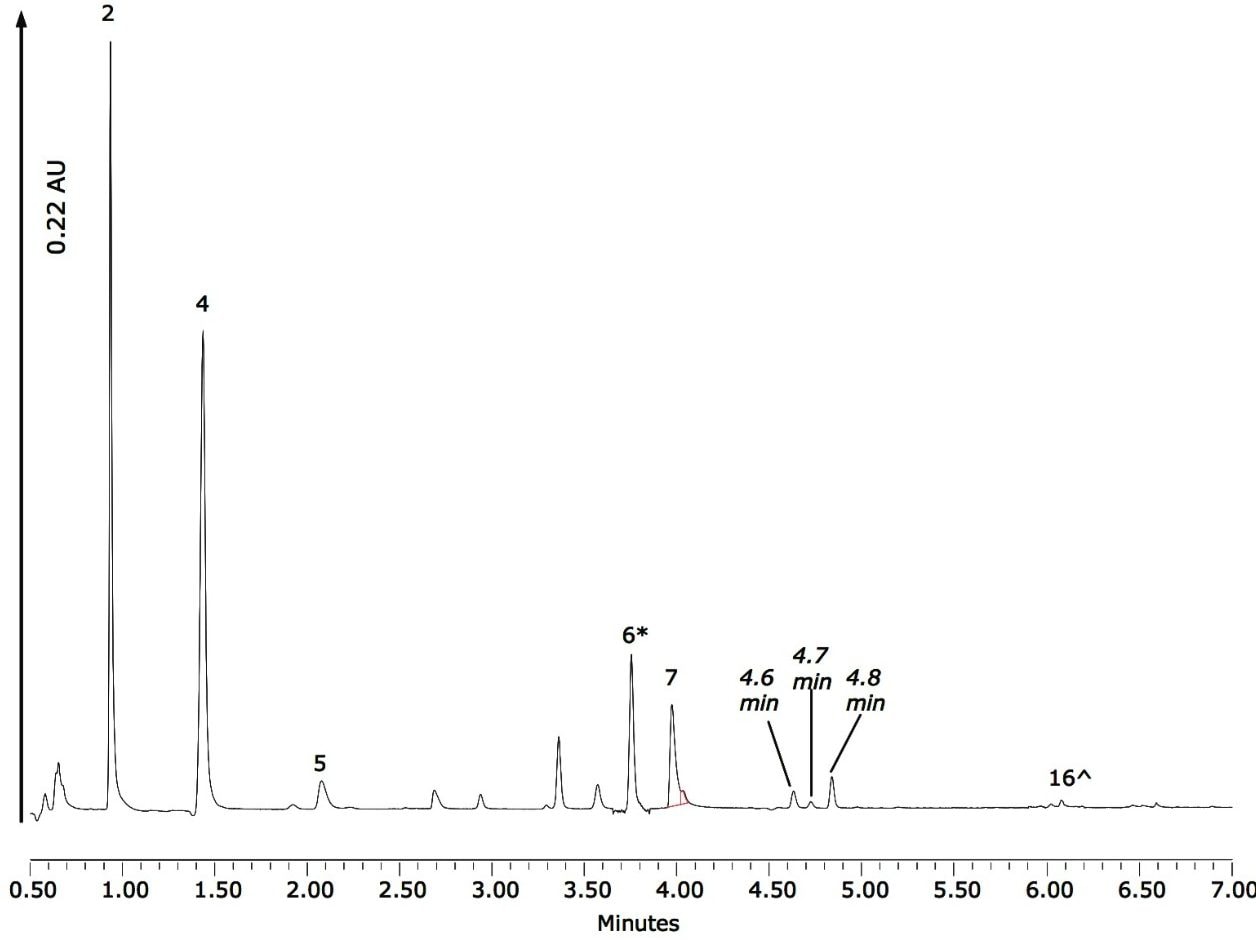
The drink was filtered using a 0.45 µm PVDF filter, then transferred to an amber Waters ACQUITY-certified vial with no further dilution. The chromatogram acquired from analysis of the fruit drink is shown in Figure 4, demonstrating that only minimal sample preparation is required prior to the analysis of this particular beverage matrix. The compound numbers in Figure 4 correspond to those given in Table 1, and it can be seen that all components stated on the label can be identified in the beverage matrix.
Figure 4 shows that peak 7 (the dye FD&C Yellow No. 5) has a shoulder that was not previously observed when the mix of standards was analyzed. This could indicate a co-eluting interference, or possibly the presence of different isomers of the dye. Inspection of the spectra, shown in Figure 5, suggests that the shoulder is a co-eluting species, which can be spectrally resolved from the dye molecule.
Also highlighted in Figure 4 are three peaks at 4.6, 4.7, and 4.8 min that could result from B12, B9, and caffeine respectively. Once again, examination of the spectra at those retention times provides information regarding the identity of these peaks.
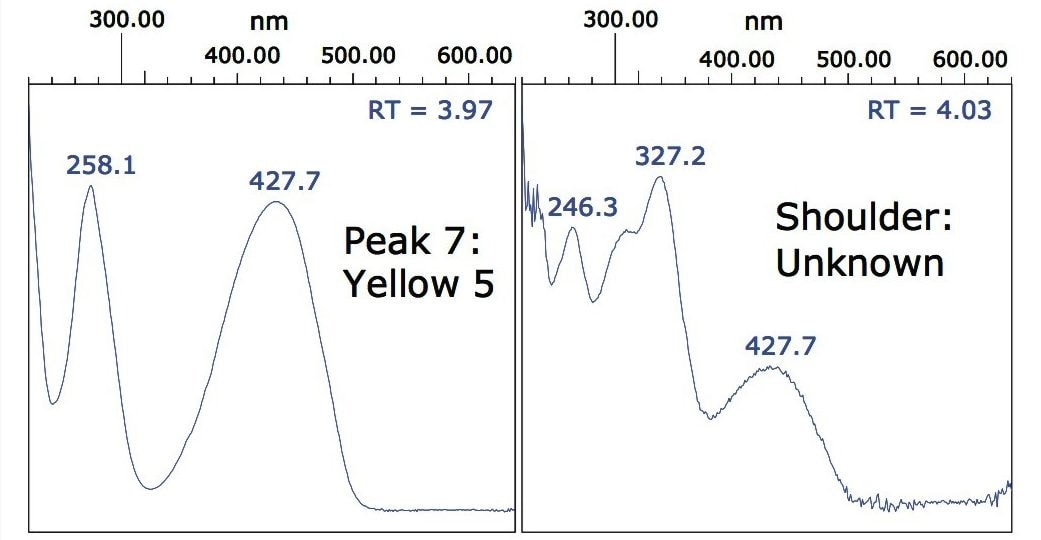
Figure 6 shows a comparison between the spectra for the B12, B9, and caffeine standards and the spectra acquired from the lemon-lime flavor fruit drink.
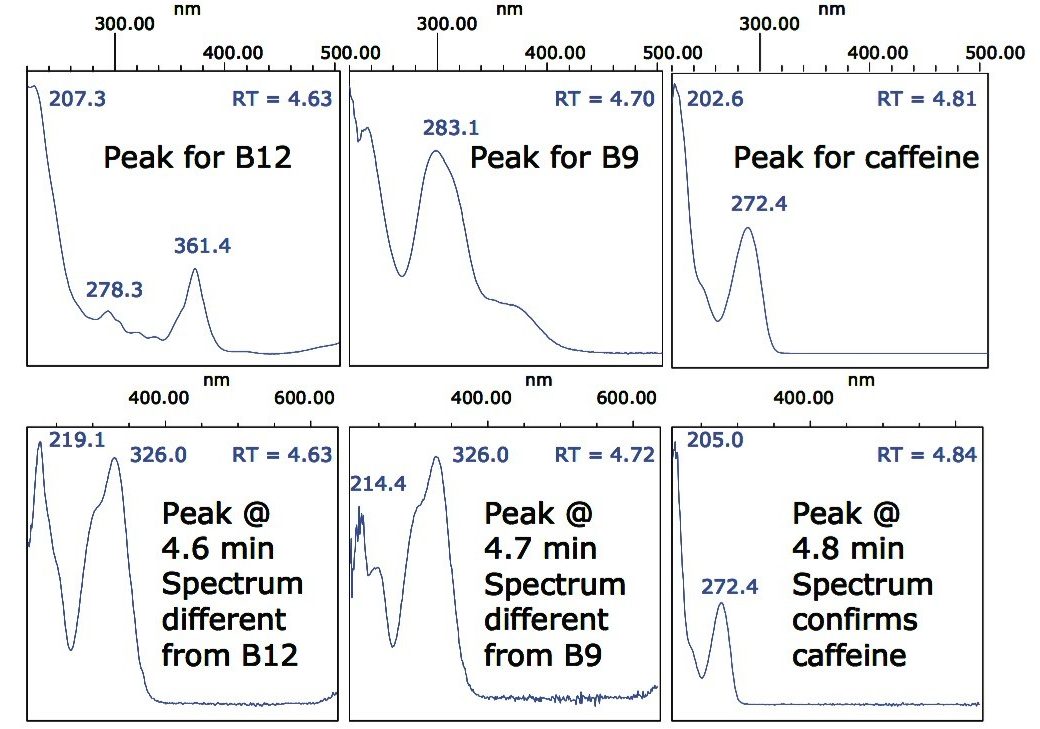
Figure 6 indicates that the peaks at 4.6 and 4.7 min are unknowns, and definitely not vitamins B12 or B9; however, the peak at 4.8 min does have a spectrum that suggests there is a small amount of caffeine present but undeclared on the beverage label.
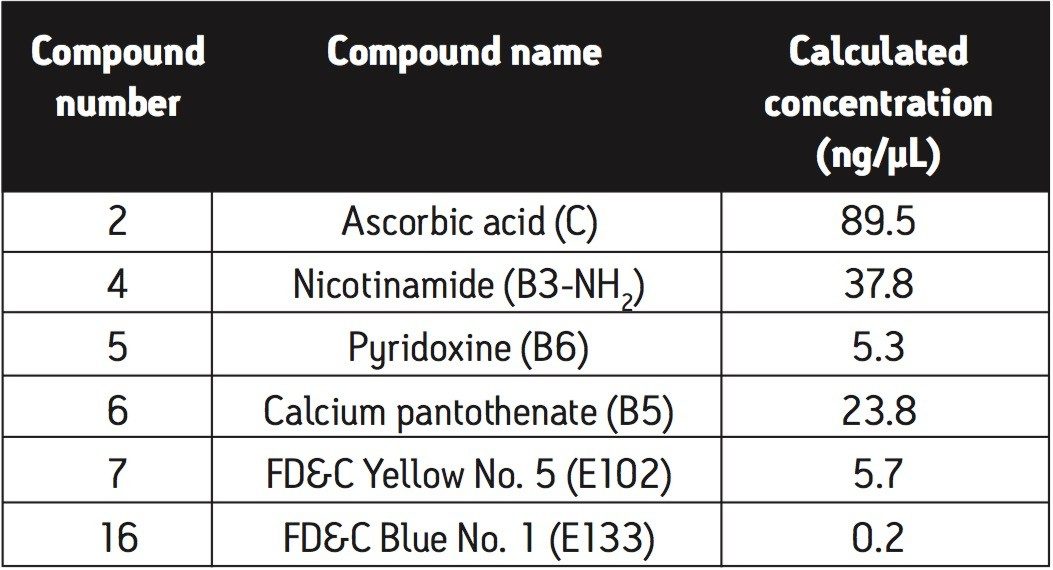
Having clearly identified the peaks of interest corresponding to the components identified on the beverage label, an average of three replicate injections was then used to calculate the concentrations of each of the identified compounds. This information is shown in Table 2.
A rapid and simple solution was developed for the simultaneous analysis of 10 water-soluble vitamin compounds, along with caffeine and six widely used food dyes in one 7.5 min run using ACQUITY UPLC with extended λ PDA, as shown in Figure 7. This system provides a reliable and cost-effective alternative to UPLC-MS/MS when the enhanced sensitivity and selectivity offered by MS detection is not required.

The ACQUITY UPLC System with PDA detection offers enhanced chromatographic resolution and shorter analysis times, compared with traditional HPLC methods. This provides the revenue-conscious laboratory with improved productivity and increased profitability due to faster sample turn-around times and reduced use of labora-tory consumables. In addition, the environmental impact is greatly minimized through decreased solvent usage.
720003188, September 2009