This application note uses the Waters AutoPurification system with automation and special software to focus on a specific portion of an analytical gradient to improve LC performance and fraction quality.
The primary goal of a purification laboratory is to efficiently isolate compounds, while maintaining purity and recovery. A successful purification meets both quantity and quality needs that equal to exceed the requirements of the next stage of the process.
Successful compound isolation may require the operator to optimize gradient parameters to improve the efficiency of the separation. This may be referred to as a focused, narrow, or shallow gradient. Focused gradients allow for optimal target separation from closely eluting impurities, thus improving the purity of the resulting fraction.
Fractions of insufficient purity can give ambiguous results in bioactivity screening or can be difficult to interpret in structural characterization. Fractions that do not meet minimum purity requirements may have to be reprocessed, which will decrease overall laboratory efficiency and subsequently increase costs.

A standard solution of pharmaceutical-like compounds was prepared to simulate the conditions under which many purification systems operate.
|
LC system: |
Waters AutoPurification system |
|
Column: |
Waters XBridge Prep OBD C18 5 μm, 19 x 50 mm XBridge C18, 5 μm, 4.6 x 50 mm |
|
Column temp.: |
Ambient |
|
Injection volume: |
Preparative, 1000 μL; Analytical, 25 μL |
|
Mobile phase A: |
0.05% Formic acid in acetonitrile |
|
Mobile phase B: |
0.05% Formic acid in water |
|
Flow rate: |
20 mL/min |
|
Gradient: |
5 to 95% B over 10 min |
|
MS system: |
Waters 3100 Mass Detector |
|
Ionization mode: |
Positive |
|
Switching time: |
0.05 sec |
|
Capillary voltage: |
3 Kv |
|
Cone voltage: |
60 V |
|
Desolvation temp.: |
350 °C |
|
Desolvation gas: |
500 °C |
|
Source temp.: |
300 °C |
|
Acquisition range: |
150 to 700 amu |
|
Acquisition rate: |
5000 amu/sec |
Screening samples before purification is usually carried out on either the analytical part of the purification system or on a separate LC. Analytical screening helps to determine if compound purification is required. Samples that do not have sufficient amounts of the compound of interest may not proceed for purification.1
The retention time of the compound can be used to indicate the approximate composition of the mobile phase conditions that are required to elute the compound off the analytical column.2 This composition can be used to determine what the preparative gradient conditions should be used to improve the separation.
The chromatogram in Figure 2 shows that the compound of interest, peak 1, is co-eluting with another compound, peak 2. Therefore this gradient, if scaled up to prep, would not provide sufficient separation to provide a pure fraction.
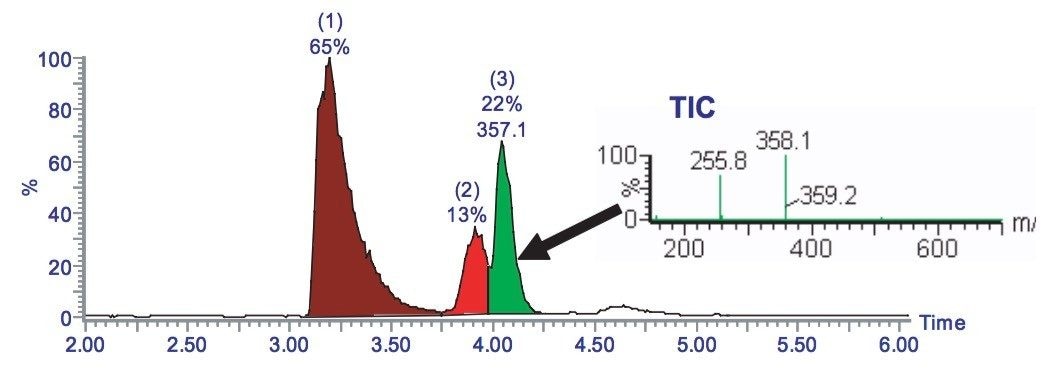
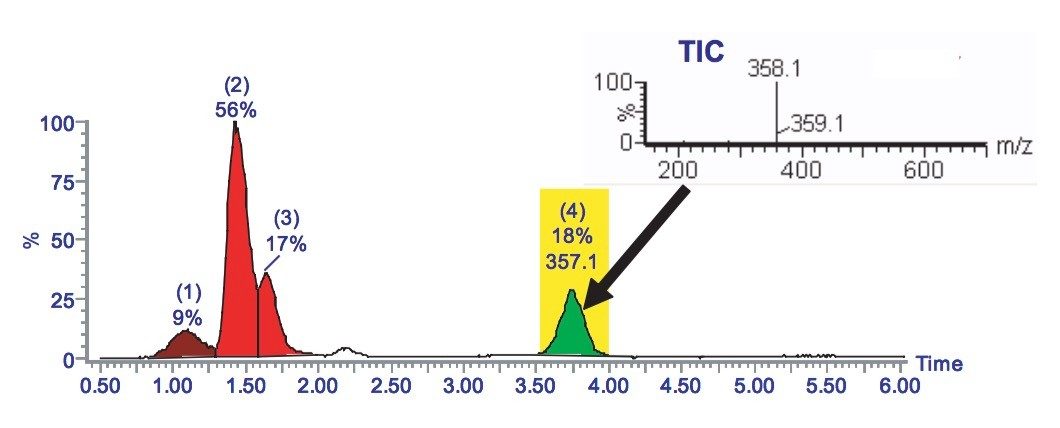
A shallow gradient can be used to improve upon the chromatographic resolution (Figure 3), which was achieved using the generic gradient. The pure fraction can be collected.
In a high throughput environment, it would not be efficient to manually create specific gradients for each sample to be purified. Automation of the process can be achieved with the use of AutoPurify, the application available within the FractionLynx Application Manager for MassLynx software. The software is capable of selecting one of six gradients, each of which focuses on a different portion of the generic analytical gradient.
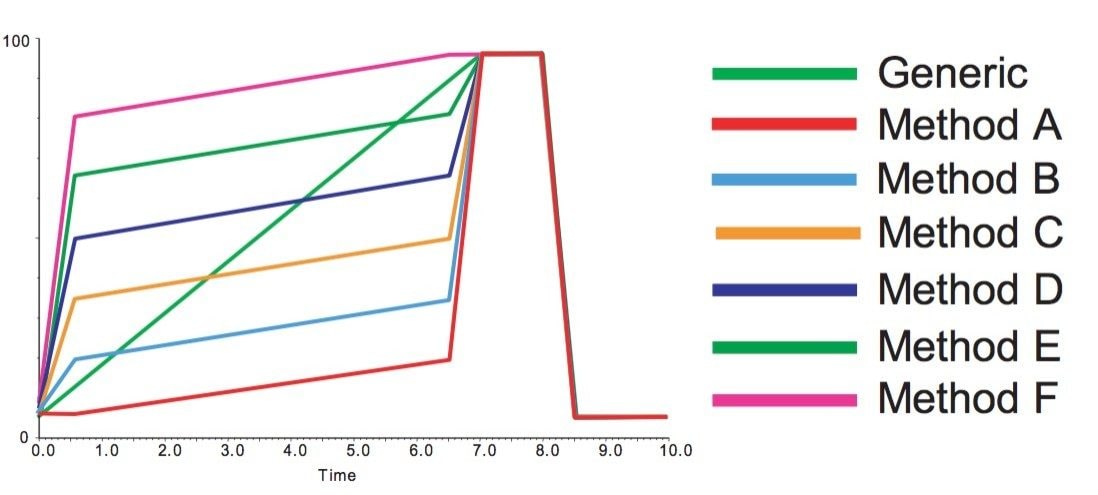
If the resolution is adequate in the analytical separation, even shorter focused preperative gradients can be used to isolate the compound of interest.
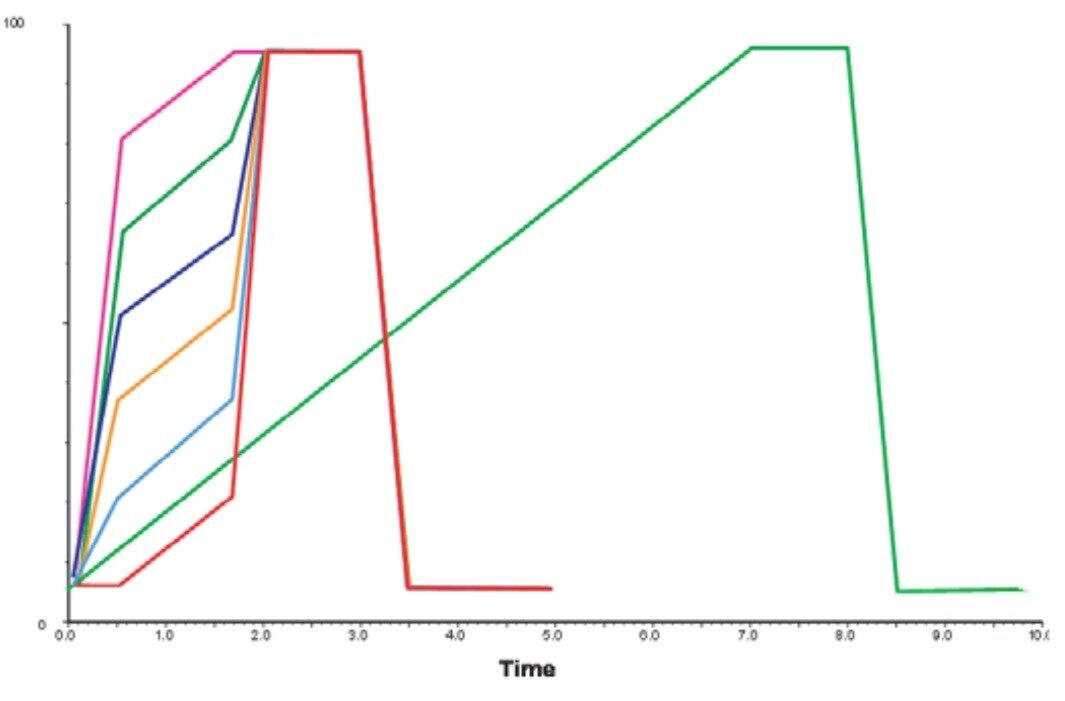
In the example in Figure 6, we can compare the isolation of the compound using either a long generic gradient or a short narrow gradient.
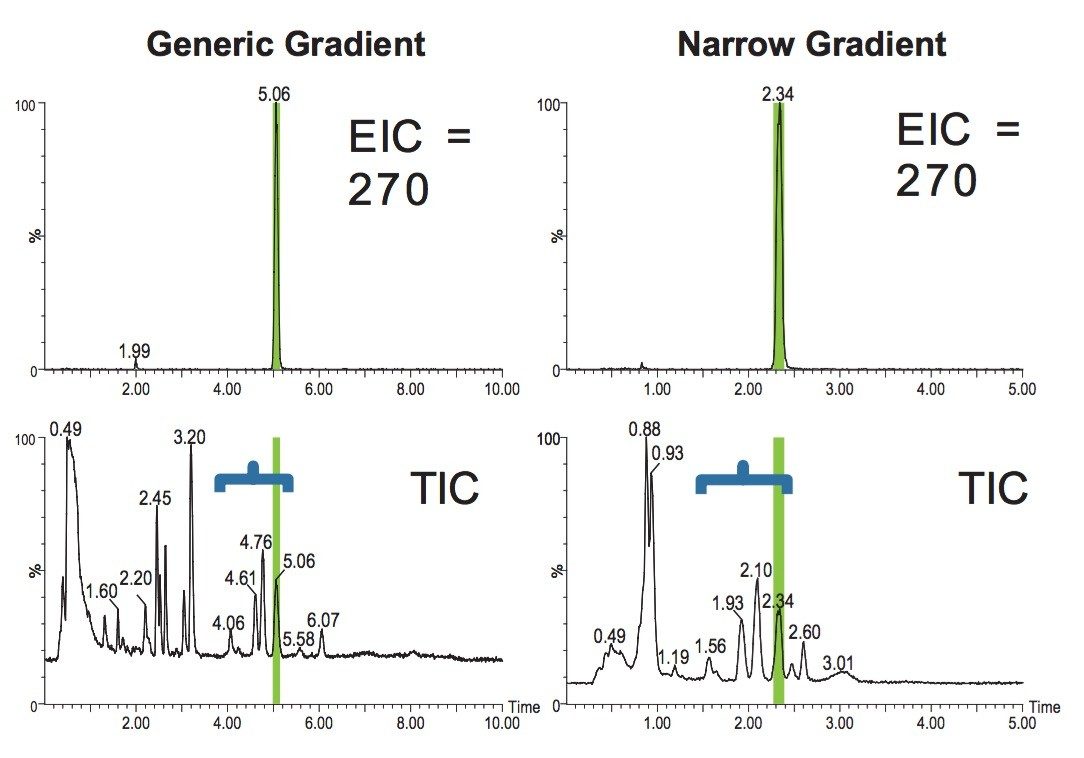
The result shows that the resolution is maintained over the focused section of the gradient (the blue bracket). Note that there is a loss in resolution, as expected, in the area of the gradient outside of the blue bracket.
720002284, June 2007