For forensic use only.
This is an Application Brief and does not contain a detailed Experimental section.
Testing for the library entries has been conducted at both 450 °C and 600 °C. No significant differences were observed in the results between the two temperatures. Therefore, either temperature may be used without impacting the integrity or applicability of the data. Moving forward, the 450 °C library will be used as the standard for consistency and efficiency.
The analysis of seized drugs plays a vital part in the effectiveness of national and international programs which aim to control the use, trafficking, and distribution of illegal drug substances. However, the sheer number of samples received for analysis, places a huge burden on the drug control laboratories and drug enforcement agencies.
Most forensic drug chemistry laboratories follow industry guidelines which stipulate that two independent analytical techniques should be used to analyze a drug sample.1,2 Typical workflows include colorimetric or TLC presumptive screening analysis followed up by a more selective method such as GC-MS. However, for many drugs, colorimetric tests are not available or may result in a high rate of false positives and TLC analysis can be time-consuming. This can lead to a greater number of samples requiring GC-MS thus leading to sample bottlenecks and sample backlogs. Consequently, methods that can facilitate a fast, but accurate, screening of drugs, are of interest.
This study explores the feasibility of using RADIAN ASAP – a new compact device based on Atmospheric Solids Analysis Probe-Mass Spectrometry (ASAP-MS), for rapid drug screening. Drug samples were analyzed following a simple dilution with methanol, and subsequent ‘dipping’ of a glass capillary into the sample. Analysis was performed using full scan MS over the range m/z 50–600. To further enhance specificity, the analysis was acquired simultaneously at four differing cone voltages i.e., 15, 25, 35, and 50V, which resulted in the generation of both precursor and product ions. Data was processed by LiveID 2.0 Software (Waters) which provided real-time matching of acquired data to a spectral library and calculated an average match factor.
Authorities worldwide constantly struggle in the fight against the proliferation of traditional and novel drug substances. The increase in number, diversity and potential toxicity of drugs is a major concern; it also presents significant challenges for the forensic laboratories who are involved in the analysis of seized substances and are under pressure to produce results quickly.
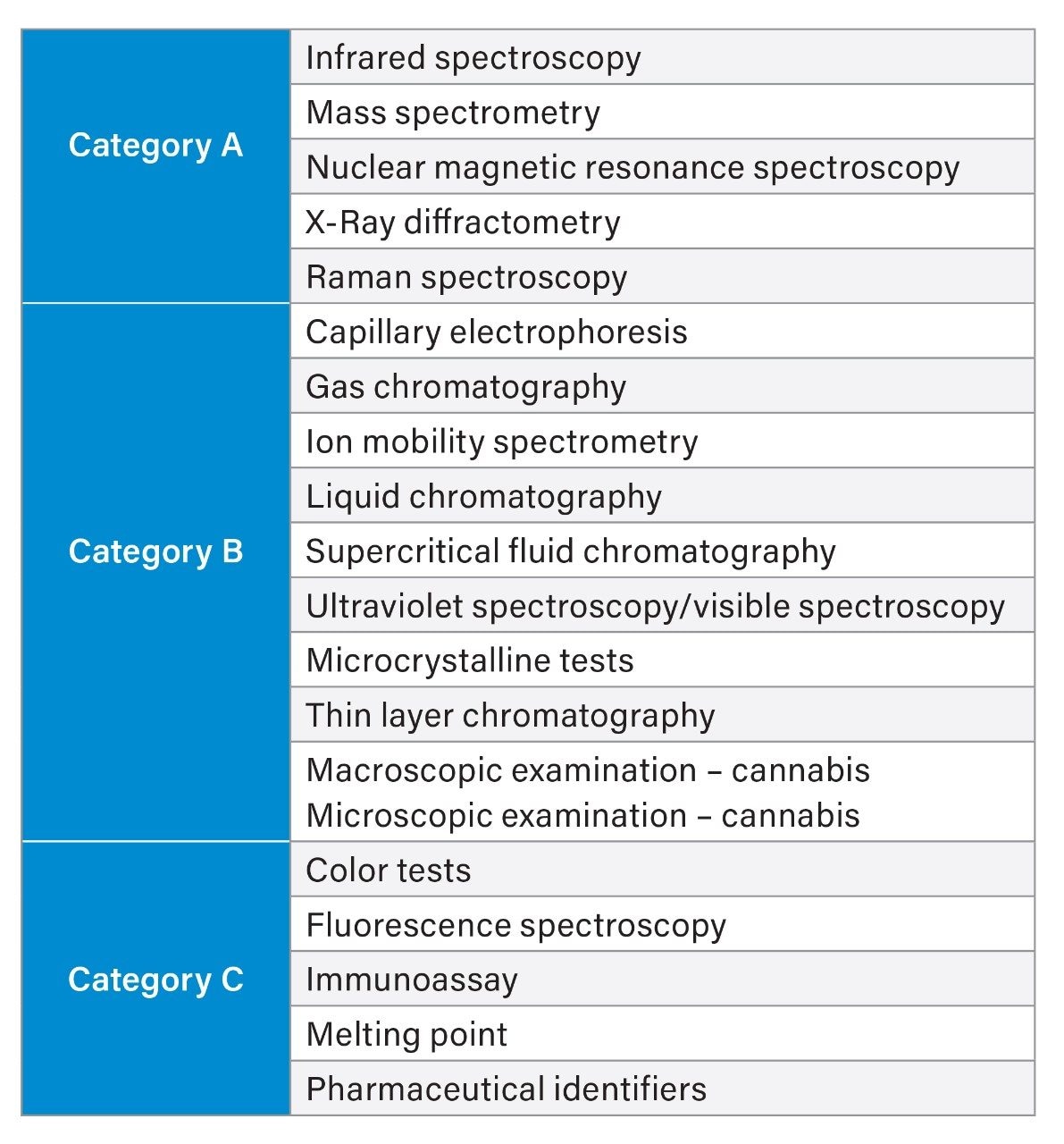
Industry guidelines, such as those provided by the Scientific Working Group for the Analysis of Seized Drugs (SWGDRUG) and the Drug Enforcement Administration (DEA) provide recommendations for the types, and minimum number of tests to provide a reliable scientifically supported identification of a drug or chemical.1,2 As a minimum it is recommended that at least two independent techniques are applied. The suite of analytical techniques are categorized by their discriminatory power (Figure 1) with Category A techniques representing the highest ranking in terms of selectivity, therefore when a Category A technique is applied, this must be supported by a minimum of one other technique (from Category A, B, or C). Commonly employed workflows include colorimetric tests, FTIR, or TLC presumptive screening analysis followed up by confirmatory analysis using gas chromatography in combination with mass spectrometry (GC-MS). However, for many drugs, colorimetric tests are not available or result in high rate of false positives, FTIR results of mixtures can be inconclusive and TLC analysis can be time-consuming. This can lead to too many samples requiring analysis by GC-MS leading to sample bottlenecks and sample backlogs. Consequently, analytical methods that can provide a fast but accurate, screen of drugs are of significant interest.
Mass spectrometry has long been considered to be a powerful analytical tool which offers high specificity, however, to date the adoption of MS as a screen, within drug control laboratories has been limited for a number of reasons, e.g., the cost of ownership, the perceived complexity of the equipment and requirement for expertise, and the relatively long run times often associated with chromatographic separation. However, these analytical systems have continued to evolve and more recent developments have resulted in robust ambient ionization MS techniques such as Atmospheric Solids Analysis Probe-MS (ASAP-MS) which permits direct analysis of samples with minimal sample preparation and without the need for lengthy chromatographic techniques.
RADIAN ASAP is a new small footprint system from Waters that combines the simplicity of ASAP with the specificity of MS.3 A series of samples including certified reference material (CRM), pharmaceuticals/over-the-counter (OTC) preparations and more than 60 unknown samples that had been confiscated at various music events/night-time venues by the police, were analyzed. Real-time results were provided by LiveID 2.0 Software, which matches the acquired data to a library and presents results as a match factor. An established High-Resolution Mass Spectrometry (HRMS) screening method was used for subsequent confirmatory analysis; this method incorporates a 15 min chromatographic separation and analysis by QTof Mass Spectrometry.4-5

Certified reference material for 67 drug substances were obtained from Merck Life Science (Dorset, UK). Pharmaceutical and OTC preparations were obtained from the local pharmacy. A series of unknown/suspect materials that had been confiscated at music events/night-time venues were supplied by the UK Police.
Certified reference material was typically supplied at a concentration of 1 mg/mL in methanol (or acetonitrile). Prior to analysis the individual standards were diluted with methanol to yield solutions at a concentration of 50 µg/mL.
Solid tablet pharmaceuticals, OTC and seized pills/material were simply added to a glass vial with 10 mL of methanol:water (50:50 v/v) and sonicated for 10 mins. Prior to analysis, a 25 µL aliquot was added to 475 µL methanol and vortex-mixed. If additional dilutions were required, these were made using methanol.
For capsular material, contents were emptied and 10 mgs of the material added to a glass vial with 10 mL methanol:water (50:50 v/v) and sonicated. Prior to analysis a 25 µL aliquot was added to 475 µL methanol and vortex-mixed. If additional dilutions were required, these were made using methanol.
For seized powders/crystalline material, 10 mgs of material were added to a glass vial with methanol:water (50:50 v/v) and sonicated. Prior to analysis a 25 µL aliquot was added to 475 µL methanol and vortex-mixed. If additional dilutions were required, these were made using methanol.
Sampling procedure – ‘dipping’ method
For each sample a new glass capillary was selected and cleaned using the automated bakeout procedure that is supplied. A ‘dipping’ method was used for each sample i.e., the cleaned capillary was held just below the surface of the liquid sample to a depth of approx. 1 cm for 5 sec, after which the capillary was placed into the holder and inserted into the RADIAN ASAP source. For these studies, each sample was analyzed in triplicate (same glass capillary used for three cycles of ‘dip and detect’).



Data was processed using the new LiveID 2.0 library matching software, which enables real-time library matching or post-acquisition processing of data files. LiveID was used in conjunction with the starter library which contained spectral data for approximately 50 common drug substances. For the spectral matching, LiveID calculates an average match factor (maximum is 1000) using a reverse-fit model. For these studies a match factor of 850 was used as the reporting cut-off, while match factors ≥900 were considered as high confidence detections.
Diluted samples were analyzed directly after dipping a cleaned glass capillary into the sample; data acquisition was automatically triggered on insertion of the sample holder into the device.
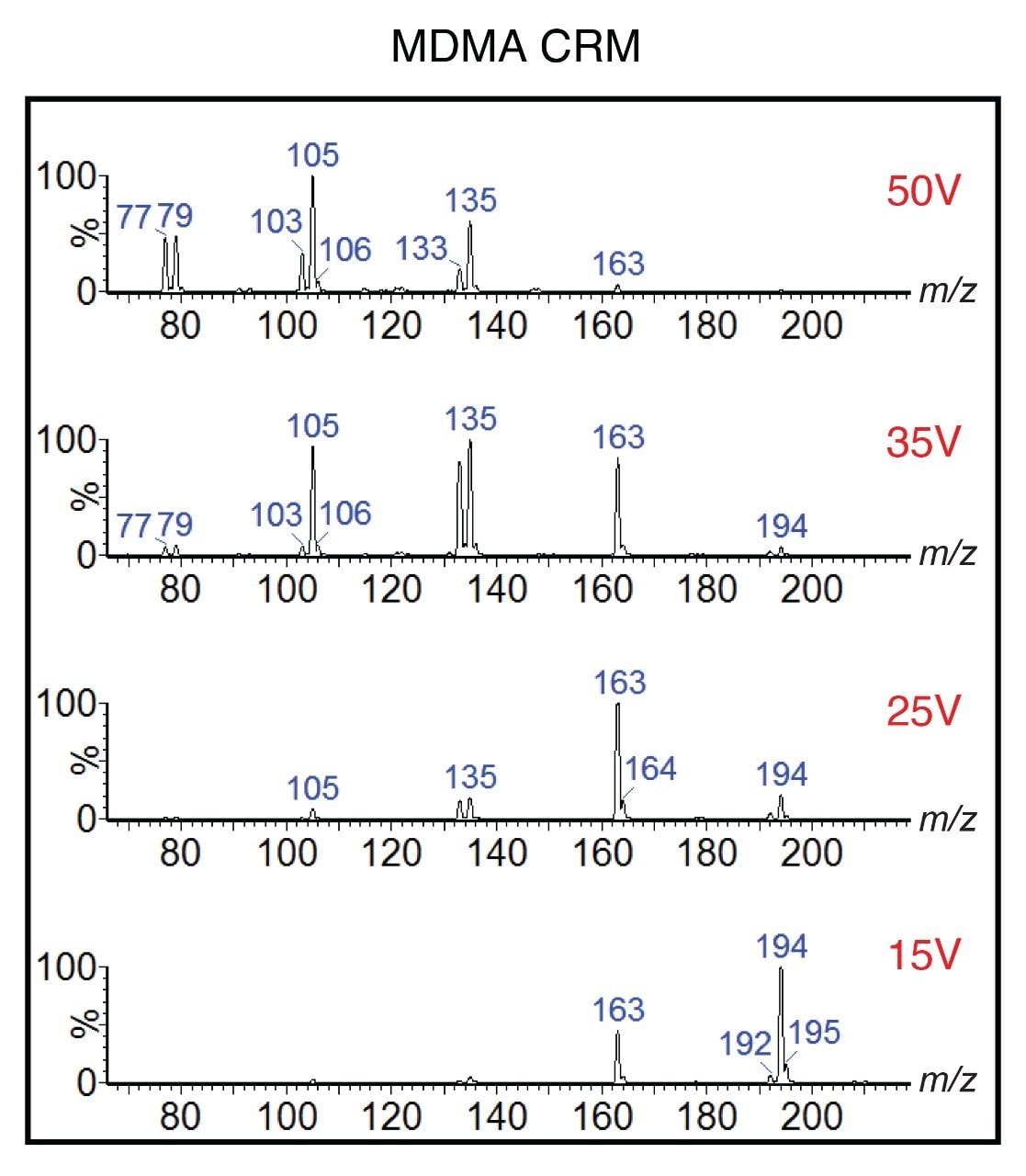
In ASAP ionization, samples are desorbed from the capillary using the heated nitrogen gas and subsequently ionized via a corona discharge. ASAP ionization is similar to the Atmospheric Pressure Chemical Ionization (APCI) process and typically results in [M+H]+ ions for most polar drugs. Mass detection was performed using full scan over the range m/z 50–600. However, to further enhance specificity for drug identification, analysis was acquired simultaneously at four differing cone voltages i.e., 15, 25, 35, and 50V, which resulted in the generation of both precursor and product ions. Figure 2 shows data for the CRM for MDMA and illustrates the wealth of information that can be generated using this approach.
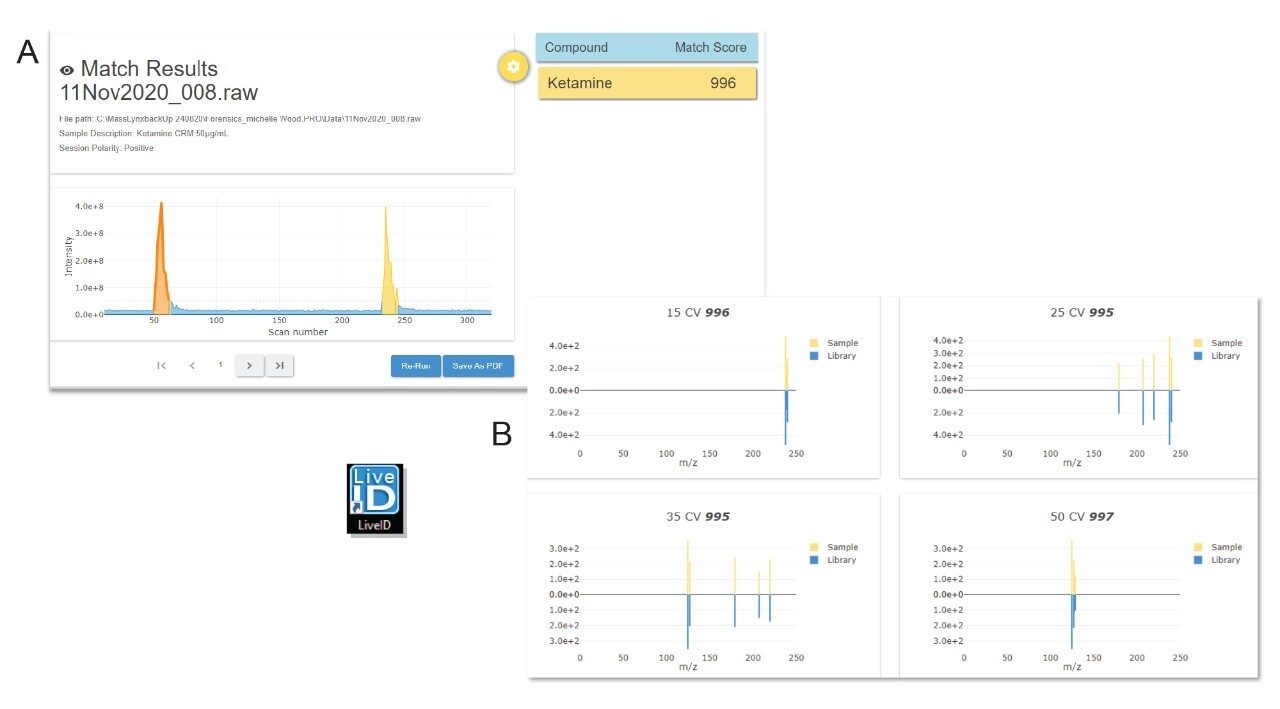
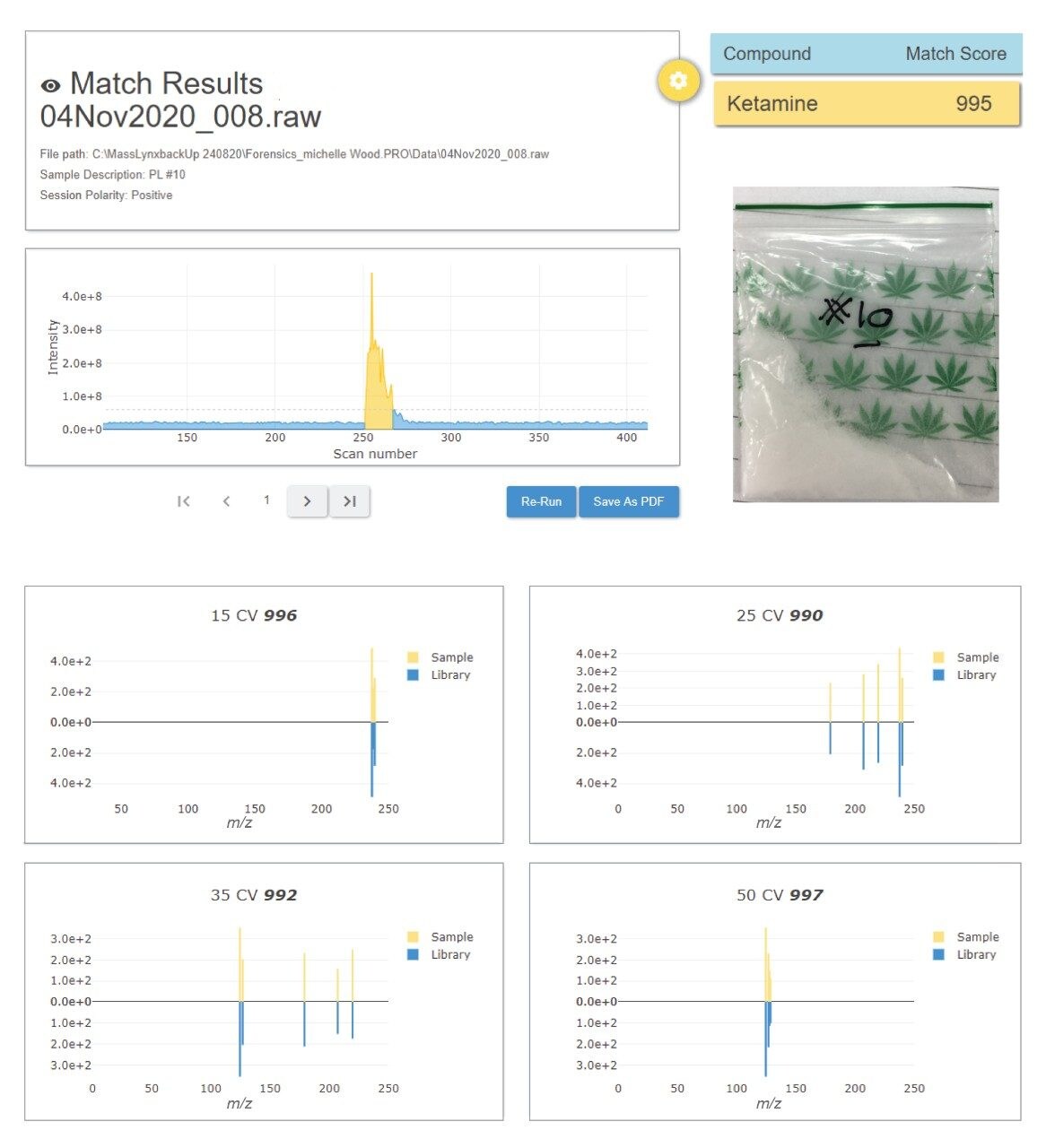
The seized drug library that is supplied with the RADIAN ASAP instrument has been acquired using the same conditions, therefore processing includes comparison of the spectral data acquired against spectra in the prepared library. This is achieved automatically, and in near-real time, using LiveID Software. An average match factor calculated. Figure 3 shows an example of the LiveID data obtained for the ketamine CRM.
In these preliminary studies, the effect of two desolvation temperatures i.e., 450 °C and 600 °C, were evaluated using CRM for 20 drug representative substances. Most analytes were detected with a mean (of the triplicate samplings) match factor >900 at both temperatures and the mean match factors were generally within 5% of each other at both temperatures. Three analytes, methamphetamine, MDEA and oxazepam showed slightly improved matches at the higher temperature e.g., methamphetamine gave a mean match factor of 918 at 450 °C, compared with 994 at 600 °C, consequently the higher temperature was applied in subsequent analyses.
The performance of the RADIAN ASAP was evaluated by analysis of CRM for 67 common drug substances at a concentration of 50 µg/mL. The overall results are summarized in Figure 4 and illustrates excellent sensitivity. Forty of the drugs analyzed were present in the library and 39/40 were correctly identified by LiveID above the minimum cut-off of 850. For these substances, match factors were all ≥877. Furthermore, in the majority (90%) of these analyses, only one compound was proposed. For the remaining analyses, a secondary compound was also proposed but at a lower match factor e.g., analysis of the CRM for hydrocodone, led to proposal of two substances hydrocodone and codeine – mean match factors were 970 and 885, respectively.
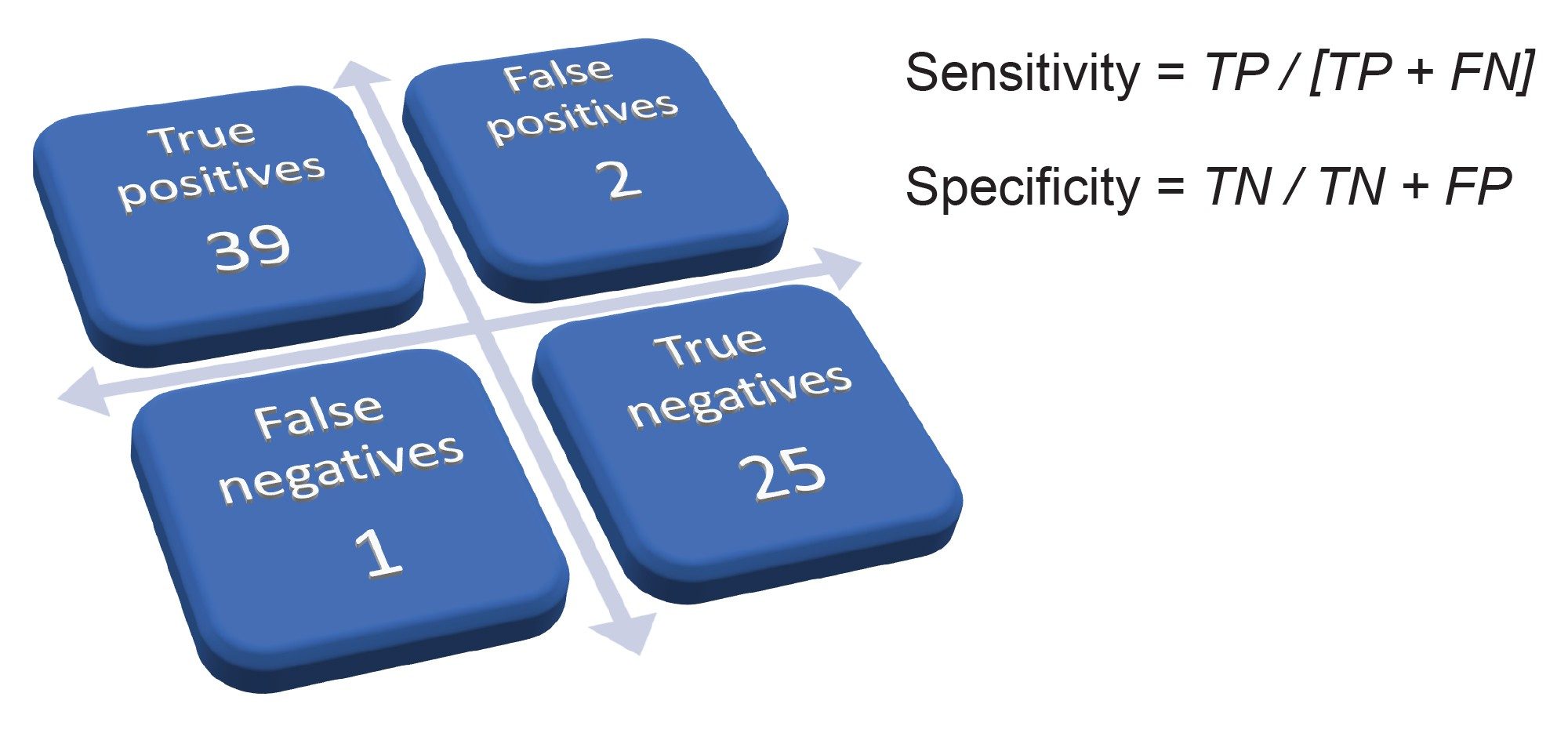
The specificity of the technique was also excellent at 92.6%. This parameter is a very important consideration for laboratories as an unacceptable level of false positive results from a preliminary screen will have a negative impact on the overall efficiency of the laboratory and will lead to more samples requiring additional analytical procedures.
In these particular analyses only two false positives were recorded. However, in both cases, it was recognised that the current library did not actually contain data for the reference material in question, but rather led to the proposal of related compounds from the same drug class e.g., analysis of para-methoxyamphetamine (PMA) CRM resulted in a proposal of methamphetamine, another substance of the amphetamine drug class. It is therefore anticipated that expanding/updating the library with the relevant data should improve accuracy of subsequent identification and result in proposal of the substance at a higher match factor than the related compound.
The study also included analysis of a number of pharmaceutical/OTC preparations and more than 60 unknown samples that had been confiscated at various music events/venues by the police. Overall, RADIAN ASAP with LiveID demonstrated very good qualitative agreement with an established High-Resolution Mass Spectrometry (HRMS) screening method.4-5 A high proportion of the seized drugs screened positive for the drug ecstasy (MDMA) and ketamine. The RADIAN ASAP data for one of the samples is shown in Figure 5, this result was obtained within 1 min.
RADIAN ASAP is a compact, robust, and easy-to-use system which enables rapid drug screening. The system combines the proven, gold standard technology of mass spectrometry for superior specificity, with the simplicity of direct analysis.
LiveID Software offers near-real time data processing with easy to interpret results typically obtained within 1 min.
RADIAN ASAP together with LiveID, demonstrates great promise for incorporation into the analytical workflow for seized drug analysis and offers forensic laboratories the potential for a rapid, accurate triage of samples that are not suited to colorimetric tests or produce inconclusive results with colorimetric tests or FTIR.
720007125, January 2021