This is an Application Brief and does not contain a detailed Experimental section.
For in vitro diagnostic use. Not available in all countries.
Vitamin K1 (phylloquinone) analysis using electrospray ionization mass spectrometry is challenging due to the hydrophobic nature of the molecule and lack of ionization sites. An additional issue is the low concentration of vitamin K1, which may be as low or even lower than 0.1 ng/mL in serum.
A new clinical research method for the analysis of vitamin K1 in serum has been developed using UPLC-MS/MS with electrospray ionization. 200 µL sample was processed with ethanol and centrifuged. The supernatants were loaded onto Oasis PRiME HLB µElution plates, washed, eluted, and a solvent exchange carried out prior to analysis.
The Waters ACQUITY UPLC I-Class/Xevo TQ-S micro IVD System enables the quantification of organic compounds in human biological liquid matrices.
This document describes a test of the analytical performance of the ACQUITY UPLC I-Class/Xevo TQ-S micro IVD System for the analysis of vitamin K1 in serum.
A chromatogram illustrating the chromatography of the vitamin K1 analysis is shown in Figure 1. Performance characteristics of vitamin K1 on the ACQUITY UPLC I-Class/Xevo TQ-S micro IVD System are shown in Table 1.

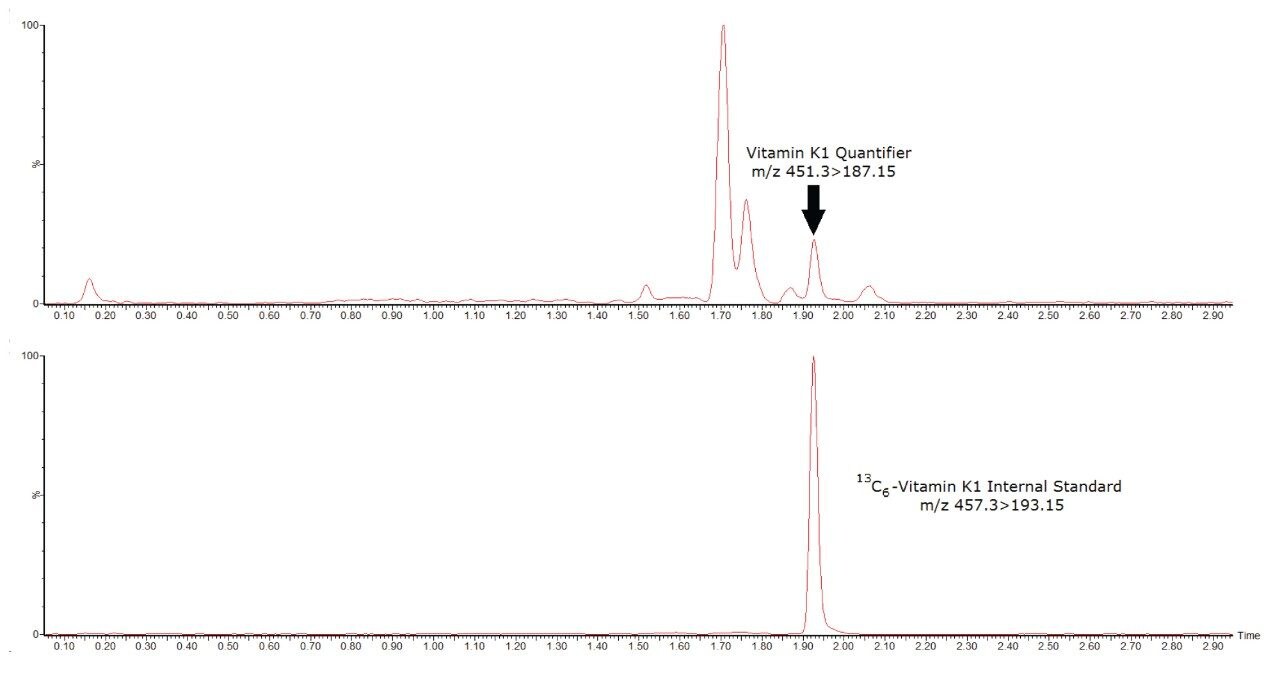
The Waters ACQUITY UPLC I-Class/Xevo TQ-S micro IVD System has demonstrated the capability to deliver analytically sensitive, accurate, and precise performance for vitamin K1 in serum.
The analytical performance data presented here is for illustrative purposes only. Waters does not recommend or suggest analysis of the analytes described herein. These data are intended solely to demonstrate the performance capabilities of the system for analytes representative of those commonly analyzed using liquid chromatography and tandem mass spectrometry. Performance in an individual laboratory may differ due to a number of factors, including laboratory methods, materials used, intra-operator technique, and system conditions. This document does not constitute a warranty of merchantability or fitness for any particular purpose, express or implied, including for the testing of the analytes in this analysis.
720007170, February 2021