This is an Application Brief and does not contain a detailed Experimental section.
The following work demonstrates the capabilities of the Andrew+ Pipetting Robot, an automated liquid handling device, for sample preparation and extraction. The Andrew+ was used to autonomously prepare and extract itaconic acid from human plasma using the Ostro Protein Precipitation and Phospholipid Removal 96-well Plate, with subsequent LC-MS/MS analysis and quantification using the ACQUITY Premier System and Xevo TQ-S micro Mass Spectrometer.
Sample preparation is a crucial step in an overall LC-MS bioanalysis workflow that is used to extract, purify, and concentrate samples from biomatrices prior to analysis. Common sample preparation techniques include sample dilution, protein precipitation (PPT), liquid-liquid extraction (LLE), and solid-phase extraction (SPE). A sample purification and cleanup step is often required in order to achieve optimal sensitivity, selectivity, and accurate quantification during LC-MS analysis. One of the simplest forms of sample cleanup is PPT. It involves taking a biological sample, diluting it with a protein disrupting solution (usually comprised of organic solvent), and extracting the analyte of interest from the matrix into the solution. This workflow may seem simple; however, it is common for errors to occur during this step due to the need for manual intervention.1 One such error that can occur is when the user is handling a pipette to transfer or dilute samples. The user can inadvertently pipette the wrong volume, dilute the wrong sample, or cause contamination by splashing during sample aspiration. Moreover, the need to reduce manual error is critical for the improvement of sample preparation workflows.
The Andrew+ Pipetting Robot, an automated liquid handling device, can effectively carry out pipetting and extraction steps for sample preparation workflows. Andrew+ runs on the cloud native, OneLab Software to create methods for liquid handling and extraction of biological samples as well as initiate flow through a vacuum apparatus, all on one deck. The easy-to-assemble dominoes can be placed in their appropriate spaces and connect via a magnet to ensure that nothing can be moved out of place during any steps in the method. The following work demonstrates how the Andrew+ autonomously prepared a standard curve and QCs and carried out a simple pass-thru extraction method of itaconic acid from human plasma using the Waters Ostro PPT and Phospholipid Removal Plate, for subsequent LC-MS analysis and quantification.
The Andrew+ prepared a calibration curve for itaconic acid in human plasma ranging from 0.5–100 ng/mL (N = 2), and quality control samples at 0.75, 7.5, and 75 ng/mL (N = 4), respectively. More information on standard curve preparation extraction using the Ostro plate and LC-MS analysis can be found in Waters application note 720006683.2 A recovery experiment was conducted for the low and high QC concentrations by the Andrew+ and by hand to compare the accuracy and precision of the fully automated workflow to a manual workflow. The Andrew+ loaded plasma samples and protein disruption solvent onto the Ostro PPT Plate and aspirated to mix. The vacuum was then applied by the Andrew+, with a constant pressure of -5 psi for five minutes. A user action notification was implemented on the software and it instructed that the precipitated samples be manually removed and transferred to a nitrogen evaporator, blown down to dryness, and placed back on the Andrew+ for reconstitution. The full Andrew+ deck layout can be seen in Figure 1. For analysis, the ACQUITY Premier System and Column was used to mitigate analyte metal chelation and ensure recovery of itaconic acid from the column and system. Percent recoveries for low and high QCs were better than manual recoveries at 100% and 99%, respectively. The comparison of recoveries for the automated workflow performed by the Andrew+ versus the manual workflow can be seen in Figure 2. Intra-day accuracy and precision of quality control samples with an N = 4 per concentration can be seen in Table 1. Accuracies were between 104.9–112.8% with RSDs between 1.3–3.6%. Excellent quantitative performance was achieved with an R2 = 0.997 over a 200-fold concentration range, seen in Figure 3.

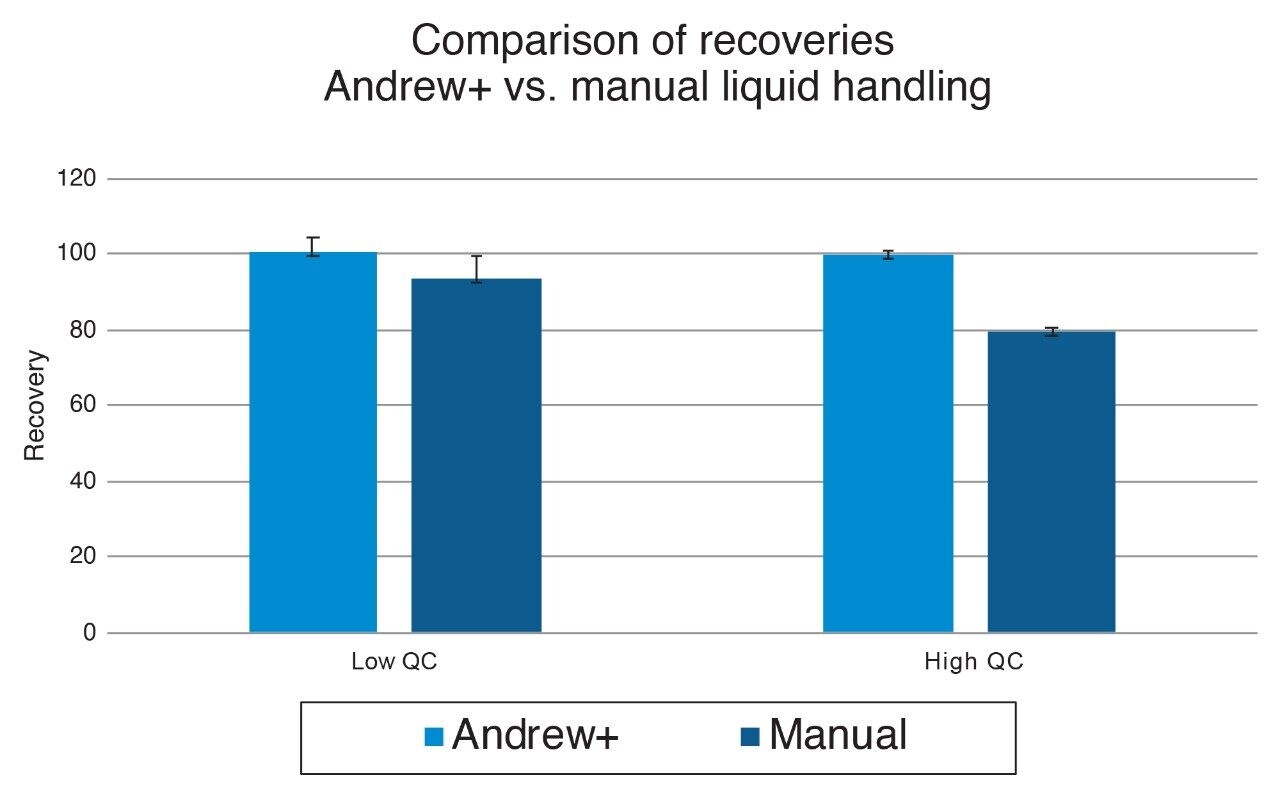
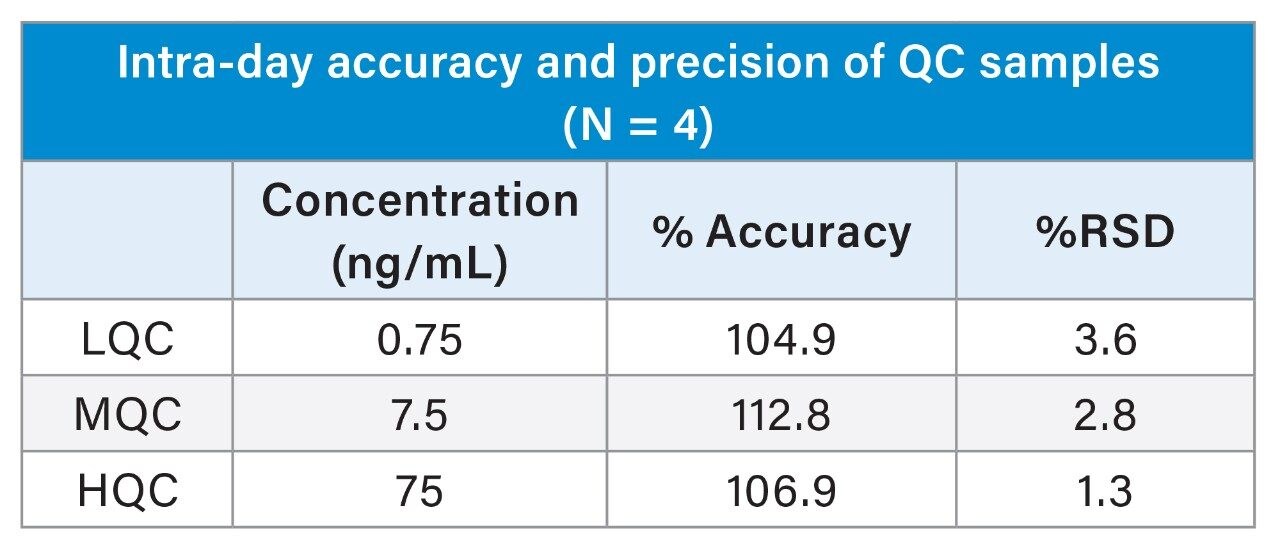
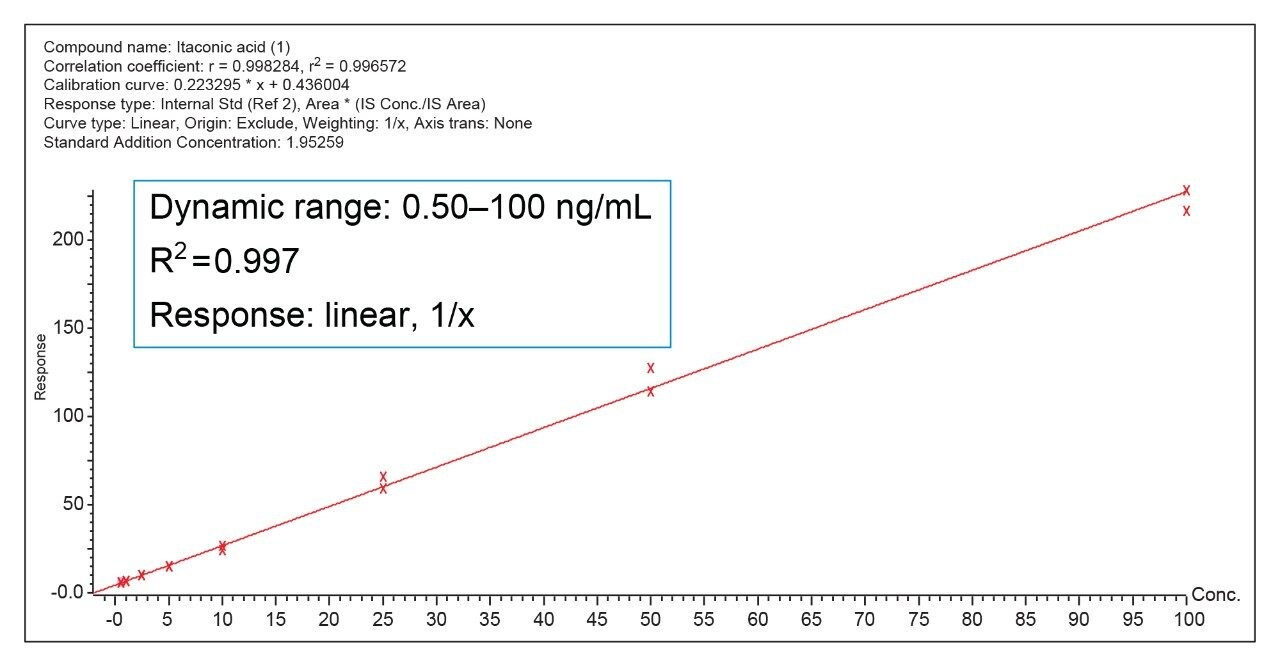
This work demonstrates successful automated sample preparation and PPT extraction using the Andrew+ Pipetting Robot with the Waters Ostro 96-well Plate. When compared to a manual workflow, the Andrew+ had better recoveries and improved RSDs, with great intra-day QC accuracy and precision, meeting recommended performance criteria for bioanalytical quantitation assays.3 The Andrew+ method demonstrated the ability to autonomously pipette and extract a full standard curve, and give excellent quantitative performance. Overall, the Andrew+ Pipetting Robot, and broadly applicable bioanalytical workflow, offers an easy-to-use automated liquid handling and extraction solution for Ostro PPT and phospholipid removal.
720007176, February 2021