Many of the methods currently employed within the food manufacturing industry for quality control purposes are based on spectroscopic techniques. By comparison, direct Mass Spectrometry (MS) is a relatively new technique which has proven to offer comparable speed and ease-of-use, alongside the advantages of higher selectivity, sensitivity, and diagnostic chemical information. Numerous MS based methods have been explored for food authenticity analysis based either on the target detection of adulteration markers or the development of multivariate classification models. Within this study, we evaluated the performance of the RADIAN ASAP in combination with LiveID for chemometric modelling and subsequent real-time quality control testing. The gross addition of bulking material to dried Mediterranean oregano was used as a case study. A sample set, consisting of authentic dried oregano, olive leaves (a frequently reported adulterant), and admixtures thereof were used. The chemical profile of each sample was generated using the RADIAN ASAP and the species diagnostic region of the mass spectral profile was used to generate a multivariate model using the PCA/LDA algorithm in LiveID. The predictive accuracy of the binary model was shown to be 100% via independent validation. The model was then used for the real-time classification of a set of challenge samples. Representative samples of four different herb species (marjoram, thyme, mint, and cistus) not included in the model were classified as “outliers” and an estimated screening threshold of ≤30% adulteration of oregano with olive and cistus leaves (w/w) was calculated.
The RADIAN ASAP, in combination with the chemometrics package LiveID (v2.0), was evaluated as a tool for the rapid screening of raw food ingredients for quality control testing purposes. The gross addition of extraneous bulking material to dried herbs as an economically motivated food fraud was used as a case study. Mediterranean oregano is one of the most appreciated culinary herbs and is also used as a functional ingredient in food preparations, perfumes and cosmetic products. Its substitution with lower economical value plant leaves having similar visual properties (e.g. sumac, cistus, myrtle, olive leaves) has been previously reported in scientific literature, along with use of non-mass spectrometric detection methods.1-4 Currently, most screening methods employed by the food manufacturing sector for this type of testing are based on vibrational spectroscopic or DNA-based techniques.5 Direct-Mass Spectrometry has proven to offer comparable speed and ease-of-use, alongside the advantages of higher selectivity, sensitivity and provision of additional and valuable chemical information relating to diagnostic marker compounds or the presence of unexpected contaminants, for example.
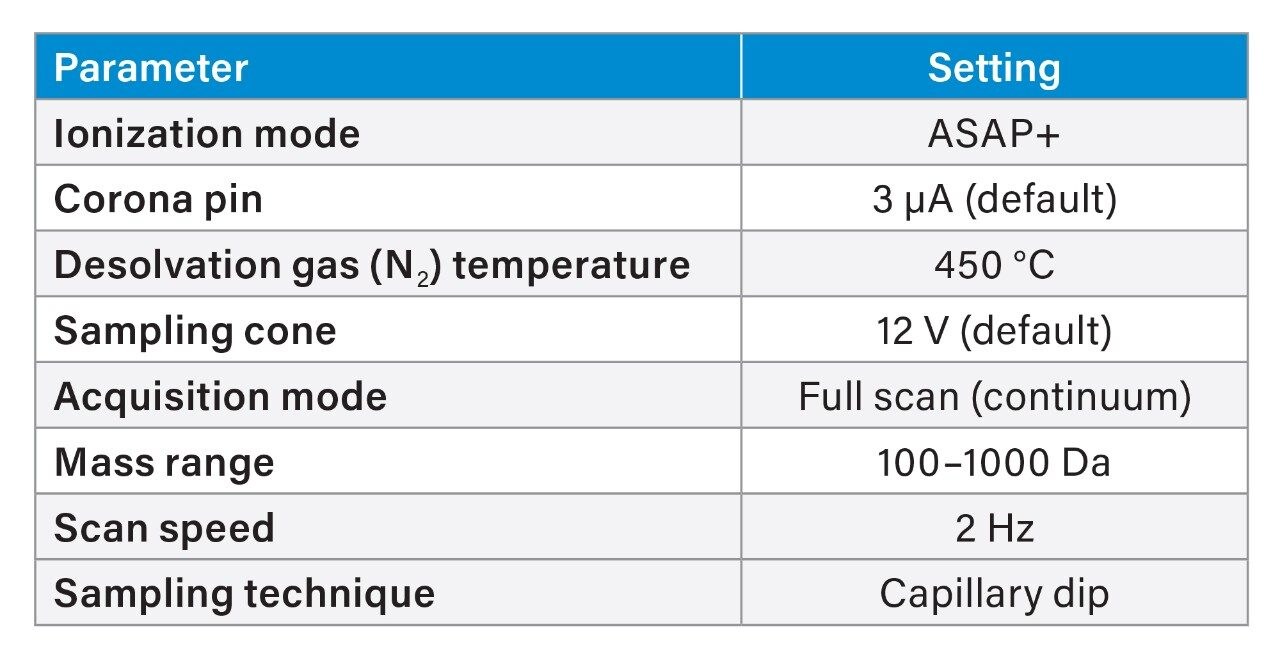
The RADIAN ASAP is a new, small footprint instrument based on the established ACQUITY QDa Mass Detector and the Atmospheric Solids Analysis Probe (ASAP) technique for direct sample introduction. The ASAP ionization mechanism in ASAP is Atmospheric Pressure Chemical Ionization (APCI) which is applicable for a wide range of thermally stable, lower molecular weight, and volatile and semi-volatile compounds in the polar to mid-polar range.
The sample introduction is performed using disposable glass capillaries either via a simple dipping technique or dispensing a fixed volume of sample extract onto the end of the capillary, according to the needs of the analysis. In the dipping technique, the capillary is dipped into, or wiped across the sample surface depending on the form of the sample. Prior to sampling, the glass capillary is loaded into the instrument, an automated cleaning step (termed the bakeout function) is performed to remove any contamination present on the glass surface.
RADIAN ASAP utilizes a fixed ionization source geometry incorporating a horizontal sample loading mechanism. This fixed source geometry serves to minimize the method development phase as the optimum positioning of the sampling capillary in proximity to the APCI pin and ionization region are fixed. The region of ionization and sampling cone are off axis to the mass detector. This design serves to improve assay robustness via reduction of detector contamination from crude samples by avoiding some of the neutral species and ion current entering the ion guide region.
For the purpose of model training and validation, a characterized set of authentic samples from the following species were sourced directly from commercial producers or collected from growing plants of known species, country of origin and harvest year; Mediterranean oregano (Origanum vulgare & O. vulgare spp. heracleoticum) n=35, olive (Olea europaea) n=20, marjoram (Origanum majorana) cistus (Cistus incanus), thyme (Thymus vulgaris), and mint (Mentha spicata).
The selection of representative likely substitutes was based on the phylogenetic proximity to oregano. Three of the species selected belong to the same family (Lamiaceae) with two of these also belonging to the same genus (Origanum) as oregano. An additional two species belonging to different families (Oleaceae and Cistaceae) which have previously been reported as bulking agents in herb fraud were also included in this study.1
Blends containing authentic oregano and the other herb species were prepared based on a weight to weight basis (in the range of 0–30%) to mimic a substitution fraud scenario.
Homogenous, dried plant material was weighed (0.2 g) and extracted with 10 mL of methanol. The mixture was shaken for 10 mins using a rotary shaker at 1,300 rpm. The extract was centrifuged at 1000 rpm for 4 minutes and an aliquot of the resulting supernatant (400 µL) was dispensed into a 1mL autosampler vial.

A pre-cleaned glass capillary was dipped into the autosampler vial containing the extract to full depth and held in contact with the solution for c. 5 sec. The glass capillary was immediately placed in the RADIAN ASAP sample loader and introduced into the ionization source region. The analysis was made whilst the extract was still wet to promote the proton transfer ionization mechanism.
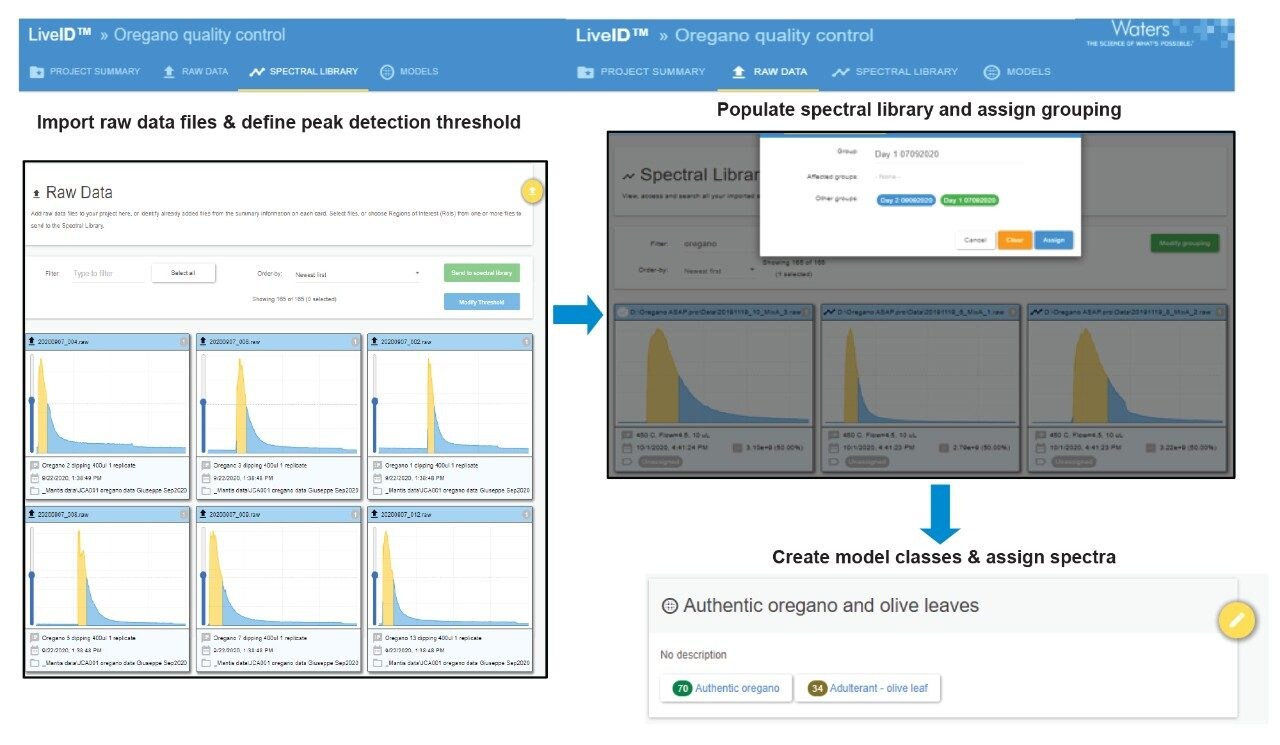
A training set of samples containing 35 different authentic oregano and 18 different olive leaf samples was selected to create the LiveID authenticity model. The samples were analyzed in a randomized order by a single analyst on a single instrument over two different days to generate two technical replicates per sample giving a population of 70 oregano and 36 olive leaf samples. The RADIAN ASAP spectra generated for the authentic oregano and other herb samples showed repeatable (from technical replicates), reproducible (from biological replicates), feature rich profiles. LiveID (v.2.0) was employed to create and validate chemometric models for discrimination between the authentic and adulterated oregano samples.
The MassLynx raw data was imported into LiveID and the pre-processing steps; normalization according to the Total Ion Current (TIC) and applied mean centering for scaling were performed to account for any instrumental sensitivity fluctuations. The LiveID workflow steps for the spectral library population are summarized in Figure 2.
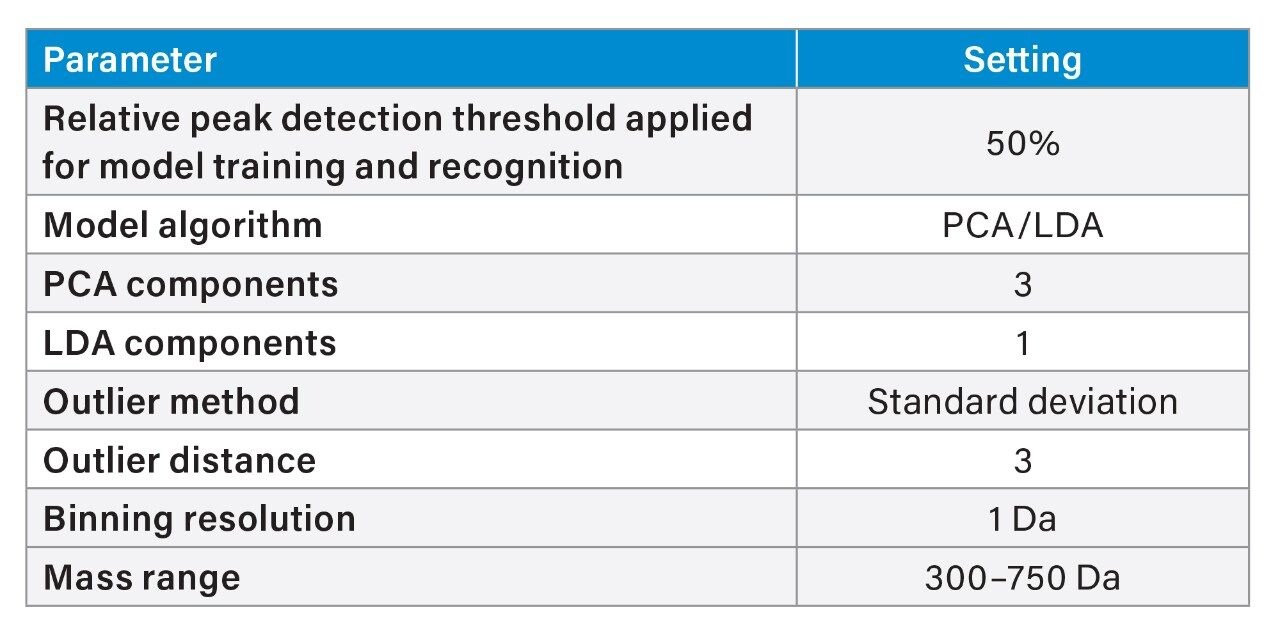
During the model optimization phase an unsupervised, Principle Component Analysis (PCA) model was built using the full spectral range (m/z 100–1000) acquired and five principal components. A binning resolution of 1 Da units was applied (equivalent to the mass resolution of the RADIAN ASAP detector). Interrogation of the LiveID PCA single component loadings plots revealed the statistically relevant m/z features responsible for the clustering according to herb species.
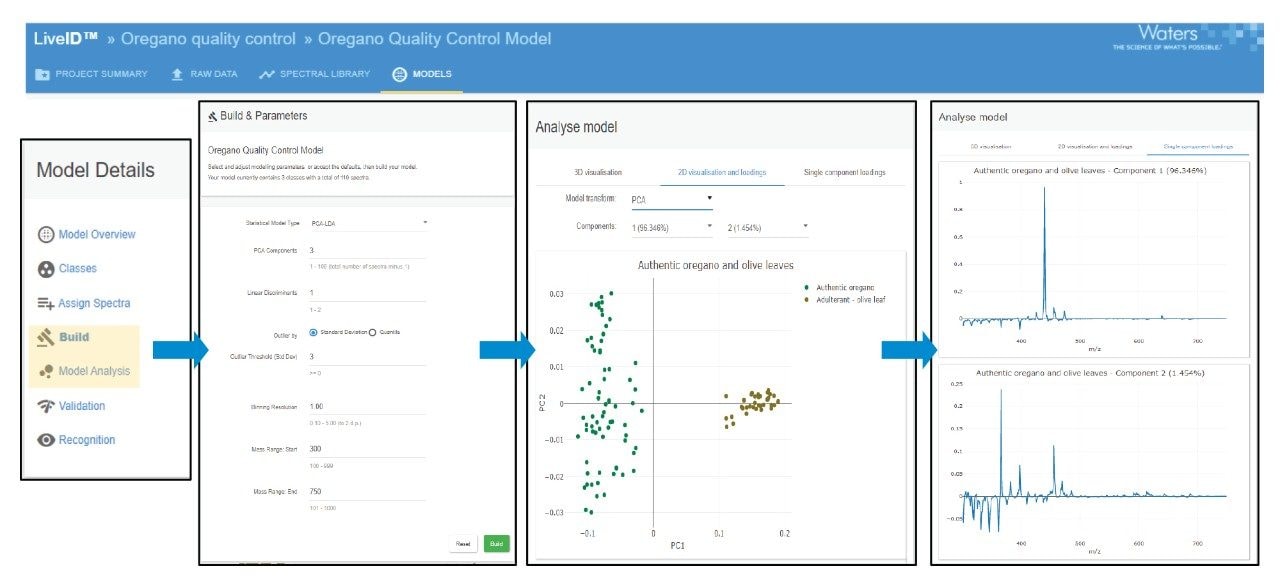
As the most diagnostic spectral range was found to be between m/z 300–750 this range was used for the subsequent model training and validation phases. A PCA/LDA model was generated whereby the supervised algorithm, Linear Discriminant Analysis, was subsequently applied to the PCA model. Three principal components were retained explaining more than 96% of the total variance in the data. One linear discriminant was applied to the reduced data set. An outlier distance of three standard deviations (97% confidence interval) was defined around the classes combining both scores and residual distances from the class centroid.
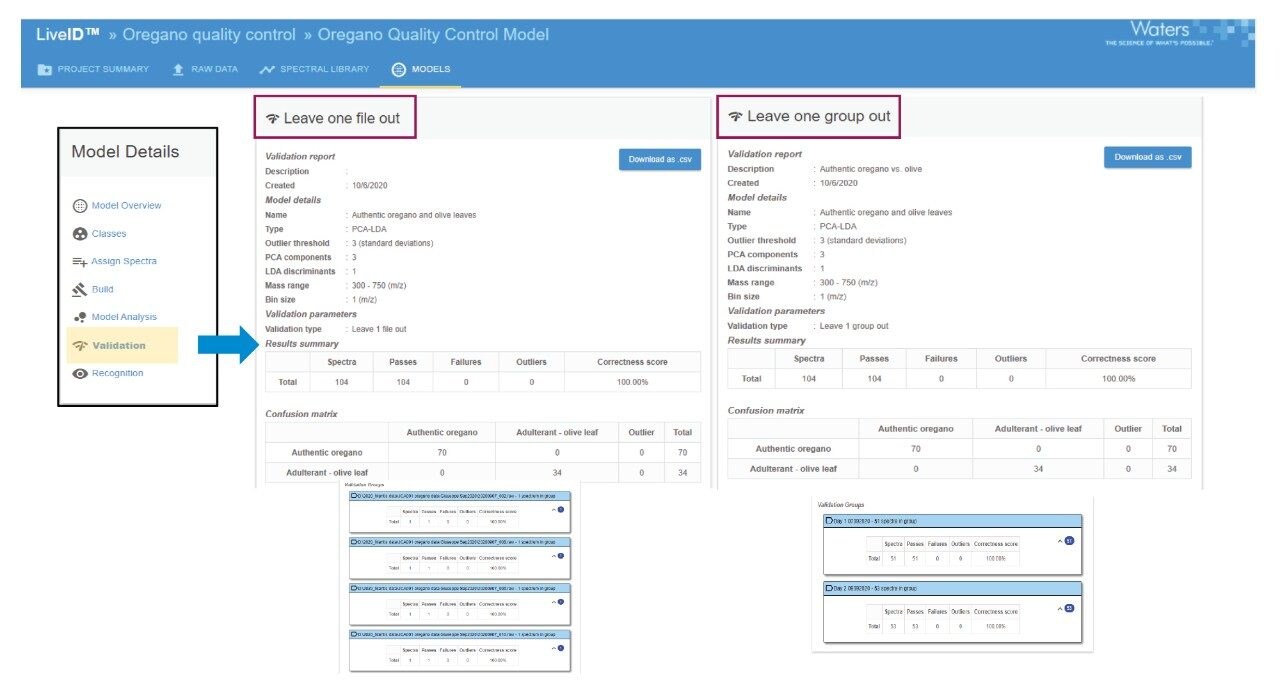
Automated in-silico cross validation was performed using two of the available validation methods in LiveID. The “leave one file out” method was used to determine % predictive accuracy for the adulteration discrimination and “leave one group out” method was used to assess the inter-day model stability.
In the case of “leave one group out”, sample replicates acquired on days 1 and 2 were assigned to a group in the LiveID spectral library. Models were created according to the analysis day grouping and subsequently challenged with the replicates acquired on the alternate day and vice versa.
Using the model parameters described above an overall correctness score (predictive accuracy) of 100% was obtained in both validation modes indicating the model reliably discriminates between the two species of herb leaf included in the model based on chemical profile differences. The inter-day model stability is robust indicating that the technical variance between analysis days is negligible.
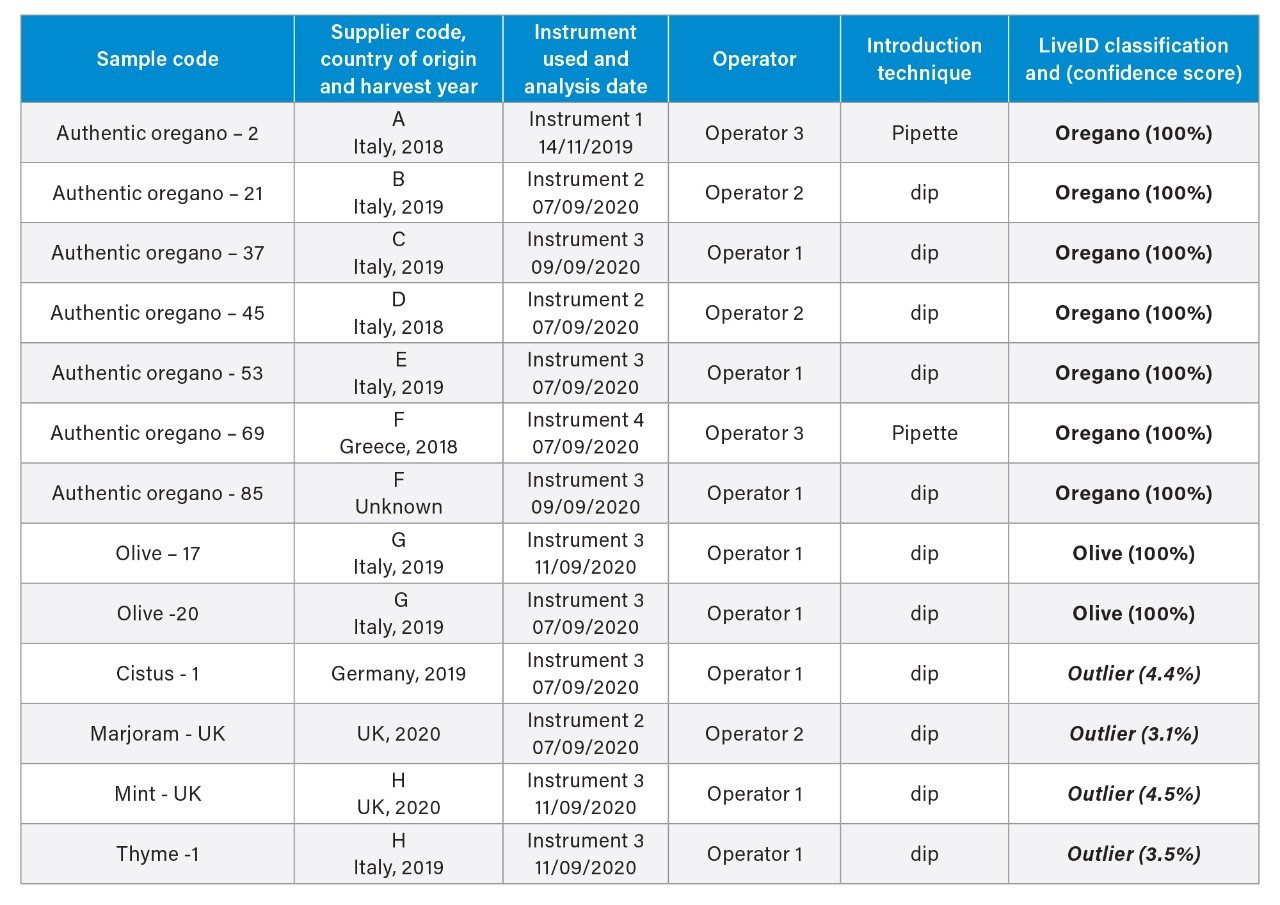
To further evaluate the reproducibility, robustness, and predictive accuracy, the model was challenged with various adulterants to simulate anticipated fraud scenarios (as 100% adulterant) as well as a selection of authentic oregano and olive leaf samples not used for model training.
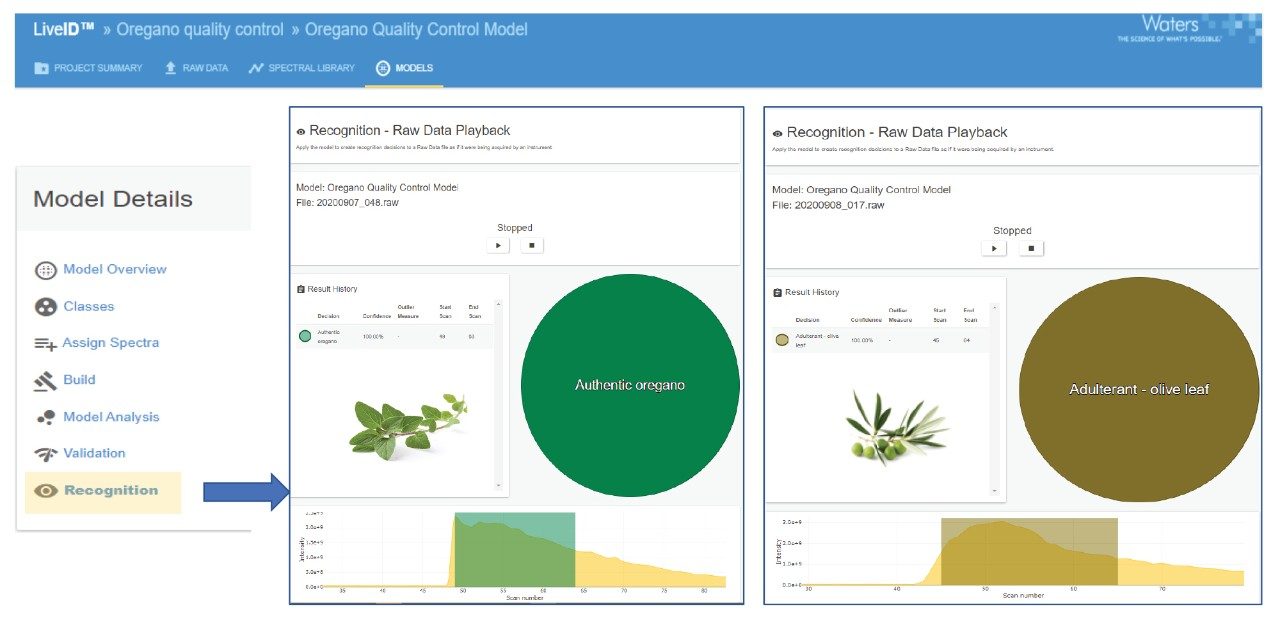
A random selection of seven authentic oregano and two olive leaf samples (representing 20% of the model training sets) were tested on one of four different instruments by one of three analysts using either the dipping or pipetting sample introduction technique. In all cases the LiveID playback recognition returned the correct classification result with a 100% confidence score. Representative samples of four different herb species not present in the model (marjoram, thyme, cistus, and mint) were also included in the validation study. The model returned an “outlier” classification indicating the chemical profile was not recognized as matching either authentic oregano or the single adulterant class (olive leaf) included in this model within the defined outlier distance.
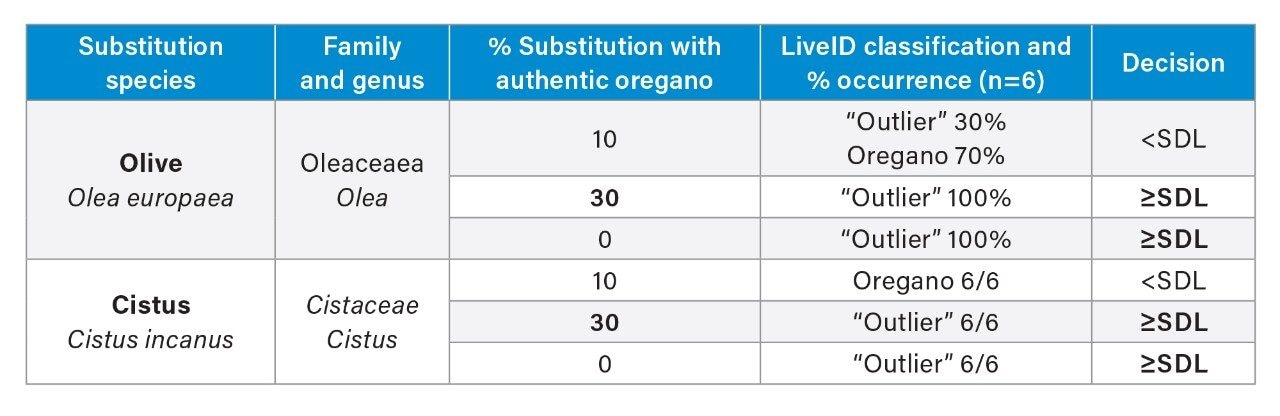
To estimate the assay Screening Detection Limit (SDL), blends containing an individual adulterant species (olive and cistus) in authentic oregano at 10 and 30% (w/w) were used. The blends were analyzed on the same instrument on two different days (n=6) and classified via LiveID real-time recognition. The SDLs were estimated as less than or equal to 30% for both olive and cistus indicating that a robust screening threshold of 30% can be established for both species. Further work is required to define the cut off levels for other potential adulterant species or admixtures thereof.
Sara Stead (Waters Corporation), Tito Damiani (University of Parma, Department of Food and Drug, Parco Area delle Scienze), Nicola Dreolin (Waters Corporation), Giuseppe Sammarco (University of Parma, Department of Food and Drug, Parco Area delle Scienze & Barilla G.R. F.lli SpA Research, Development & Quality), Michele Suma (Barilla G.R. F.lli SpA Research, Development & Quality) and Chiara Dall’Asta (University of Parma, Department of Food and Drug, Parco Area delle Scienze).
720007045, October 2020