This application note illustrates sources of oligonucleotide loss due to adsorption on metallic column hardware (frits). Solutions, columns, and best practices for improved oligonucleotide analysis are provided.
Nucleic acid-based modalities are novel types of therapeutic compounds. Among these are antisense oligonucleotides (ASO),1 silencing RNA duplexes (siRNA),2 micro RNA (miRNA),3 and larger biopolymers such as messenger RNA (mRNA) or plasmids that can serve as vaccines.4 In this application note we will focus primarily on therapeutic oligonucleotides.
Therapeutic oligonucleotides, typically 15-35 mers, are prepared by chemical synthesis. Quality control and characterization are performed using Polyacrylamide Gel Electrophoresis (PAGE), Capillary Gel Electrophoresis (CGE), or Anion-Exchange Chromatography (AEC).5 Recently Ion-Pair Reversed-Phase Liquid Chromatography (IP RP LC) has become the method of choice for LC and LC-MS analysis of oligonucleotides.1,6 IP RP LC-MS is applicable for sensitive analysis of ASO and siRNA therapeutic compounds.
IP RP LC of oligonucleotides is performed with ion-pairing mobile phases consisting of a hydrophobic amine and an acid.7-9 An example of a useful ion-pairing mobile phase is 25 mM hexylamine adjusted to pH 7-9 with acetic acid (hexylammonium acetate; HAA). Elution of oligonucleotides from reversed-phase columns is accomplished with gradients of acetonitrile or methanol. Column temperature is typically set to 60 °C to eliminate oligonucleotide secondary structure and its impact on LC separations.10 Both the alkaline pH and elevated temperature used for analysis are detrimental to the lifetime of silica-based chromatographic columns. Therefore, hybrid organic/inorganic stationary phases such as those used in ethylene bridged hybrid (BEH) columns, are recommended for IP RP LC separations of oligonucleotides.11 BEH columns are significantly more stable than conventional silica columns when using alkaline mobile phases.12
Ultra-performance UPLC columns can resolve target oligonucleotides from their shorter synthetic impurities and products of oligonucleotide metabolism. N/N-1 resolution is routinely achieved for 21/20 mer oligonucleotides.13 The separation becomes progressively more difficult for longer nucleic acids.14,15 Columns packed with sub-2-µm particles can resolve oligonucleotides up to 60 mer using shallow gradients.16 This is an adequate separation power for the majority of ASO and siRNA applications.
The principles of both AEC and IP RP LC oligonucleotide analysis are well understood. However, experts in the field of nucleic acid analysis are aware of non-specific adsorption problems that compromise oligonucleotide analysis and quantitation.17 Negatively charged nucleic acids are known to interact (chelate) with metallic surfaces such as stainless-steel or titanium, or more accurately with the oxide layer present on the metal surfaces.18,19 The adsorption on metallic surfaces may contribute to peak tailing, recovery loss, and sample carryover.20 This behavior is most apparent when a new column, previously unexposed to oligonucleotide samples, is used, and it gradually diminishes as the column is exposed to multiple sample injections.
Some laboratories have developed protocols for LC system and column conditioning to mitigate the above described effects. LC system conditioning is often performed with an instrument wash using diluted phosphoric acid. Chromatographic column conditioning is generally carried out with repetitive injections of sacrificial oligonucleotide sample prior to the analysis of a target oligonucleotide. The problem of sample loss due to adsorption on metallic surfaces is most apparent in trace analysis when sub pmol amounts of samples are analyzed.
Waters MaxPeak High Performance Surfaces (HPS) were developed for column hardware with the goal of minimizing non-specific metal interactions with analytes such as oligonucleotides, phosphopeptides, small molecule organophosphates, and other analytes that have historically shown strong affinity towards metal surfaces. MaxPeak Premier Column hardware built with MaxPeak HPS creates a barrier between the metal surfaces of the fluid path and sample thus minimizing interaction between analytes and column hardware. This application note illustrates improvements in the analysis of oligonucleotides that are afforded using ACQUITY Premier Oligonucleotide BEH C18 Columns. Considerations for LC system optimization will be discussed in separate application notes.
A 25 mer phosphorothioate (PS) oligonucleotide CTC TCG CAC CCA TCT CTC TCC TTC T, MW 7776 Da (GEM 91) was dissolved in the mobile phase aqueous buffer at a concentration of 2 pmol/µL. 1 µL (2 pmol) of sample was injected 20-90 times.
|
Instrument: |
ACQUITY UPLC H-Class Bio conditioned by 500 pmol of 39 mer oligodeoxythymidine |
|
|
Data management: |
Empower 3.0 Software, MISER experiment is enabled by FTN Sample manager firmware 1.65.356(MISER_HT_V13). Specific MISER commands such as number of injections, injection frequency, and gap timing were specified in FTN instrument method comment window. |
|
|
Method Conditions |
||
|
Columns: |
No column was used for MISER experiment. Instead, 2.1 mm UPLC column frit in holder was placed in the flow path after the injector and before the detector. In a control experiment the holder with the frit was replaced with a PEEK union (metal free flow path). |
|
|
Mobile phase 1: |
Aqueous 10 mM hexylammonium acetate (HAA) buffer, pH 7 |
|
|
Mobile phase 2: |
Aqueous 10 mM ammonium acetate (AmAc) buffer, pH 4.5 |
|
|
Mobile phase 3: |
Aqueous 10 mM ammonium acetate (AmAc) buffer, pH 5 |
|
|
Mobile phase 4: |
Aqueous 10 mM ammonium acetate (AmAc) buffer, pH 6 |
|
|
Mobile phase 5: |
Aqueous 10 mM ammonium acetate (AmAc) buffer, pH 7 |
|
|
Mobile phase 6: |
Aqueous 10 mM ammonium acetate (AmAc) buffer, pH 8 |
|
|
Isocratic conditions: |
0.2 mL/min of specified mobile phase |
|
|
Experimental temp.: |
25 °C |
|
|
Detection (UV): |
260 nm PDA, titanium 5 µL detector cell |
|
|
Injection volume: |
1 µL |
Sample Preparation:
The MassPREP Oligonucleotide Standard (p/n: 186004135) contains 1 nmol of 15, 20, 25, 30, and 35 mer oligodeoxythymidines. The vial contents were dissolved in 200 µL of deionized water, giving a final concentration of 5 pmol/µL. 2 µL of this sample was injected (10 pmol) on column.
|
Instrument: |
ACQUITY UPLC H-Class Bio conditioned by 500 pmol of 39 mer oligodeoxythymidine |
|
Data management: |
Empower v3.0 Software |
|
Method Conditions |
|
|
Columns: |
ACQUITY Premier Oligonucleotide C18, 130 Å, 1.7 µm, 2.1 x 50 mm (p/n: 186009484) |
|
Mobile phase A: |
25 mM HAA in water, adjusted with acetic acid to pH 6, 7 or 8.5 |
|
Mobile phase B: |
50% of mobile phase A and 50% acetonitrile (v:v) |
|
Column temp.: |
60 °C |
|
Detection (UV): |
260 nm PDA, titanium 5 µL volume |
|
Injection volume: |
2 µL |
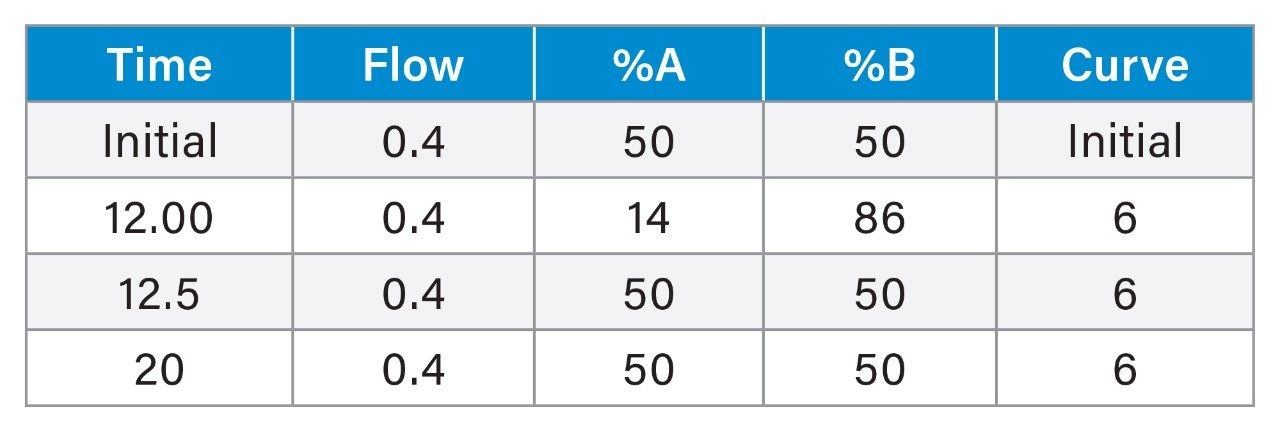
Relative recoveries in MISER experiment were estimated from the observed oligonucleotide peak areas. The peak area representing 100% recovery was obtained either with a PEEK union or after conditioning an ACQUITY Premier Oligonucleotide BEH C18 Column with an excess of oligonucleotide sample.
For the OST MassPREP standard, 100% recovery was considered to be the peak signal obtained after column conditioning with single or several 500 nmol injections of 39 mer oligonucleotide.
In our initial experiments, we evaluated the adsorptive loss of a 25 mer phosphorothioate (PS) oligonucleotide using a conditioned ACQUITY UPLC H-Class Bio System. The goal of the experiment was to estimate analyte loss due to single 2.1 mm stainless frit, the same frit used in UPLC column hardware. The frit housing outlet was connected to a PDA detector cell using 75 µm I.D. x 40 cm PEEK tubing. No chromatographic column was utilized in this experiment.
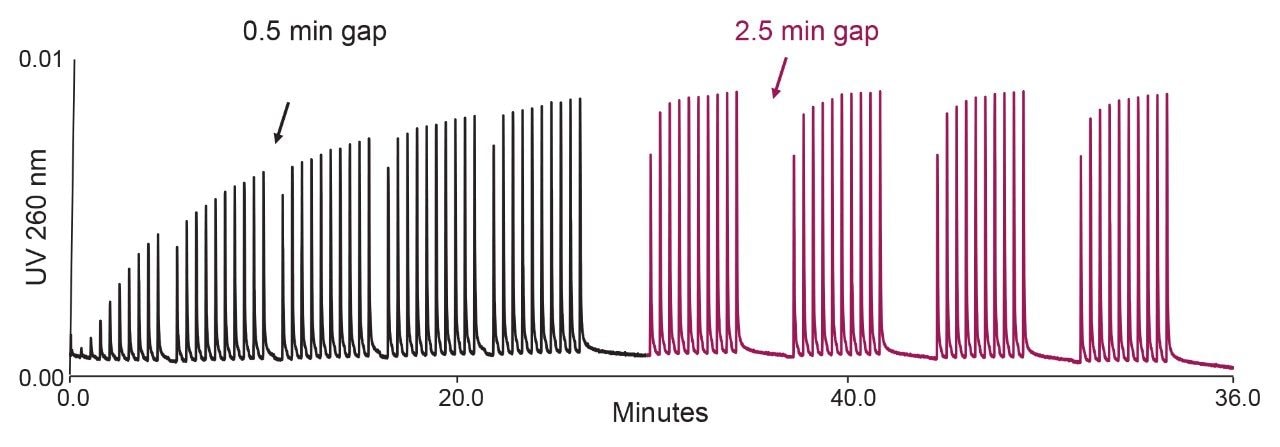
We employed a Multiple Injection in a Single Experimental Run (MISER) data acquisition approach.21 Up to 90 injections of the 25 mer oligonucleotide were performed in a single run. The injection interval was thirty seconds, and after every 10 injections a 30 second gap was inserted to improve the visualization of the results (Figure 1). In the initial test, we used a pH 7 aqueous 10 mM hexylammonium acetate (HAA) mobile phase. Red chromatogram in Figure 1A represents 20 injections of oligonucleotide sample on PEEK union. Consistent peak heights were observed. The peaks area in Figure 1A represent 100% oligonucleotide recovery. Next we replaced PEEK union with a holder containing 2.1 mm I.D. stainless-steel frit and performed 50 injections of the same sample. Chromatogram in Figure 1B illustrates the oligonucleotide sample loss due to adsorption on a frit surface. The initial peaks are low, but they increase in subsequent injections. This is because the active adsorption sites on the metal frit are gradually saturated with an excess of sample. This effect is known as LC system/column conditioning. The average peak area from Figure 1A experiment was used to quantify the analyte loss on a stainless-steel frit in Figure 1B. The recovery in the 50th oligonucleotide injection was 71%. Peak signals plateau after 50 injections; we observed only a marginal peak area improvement in subsequent injections (data are not shown).
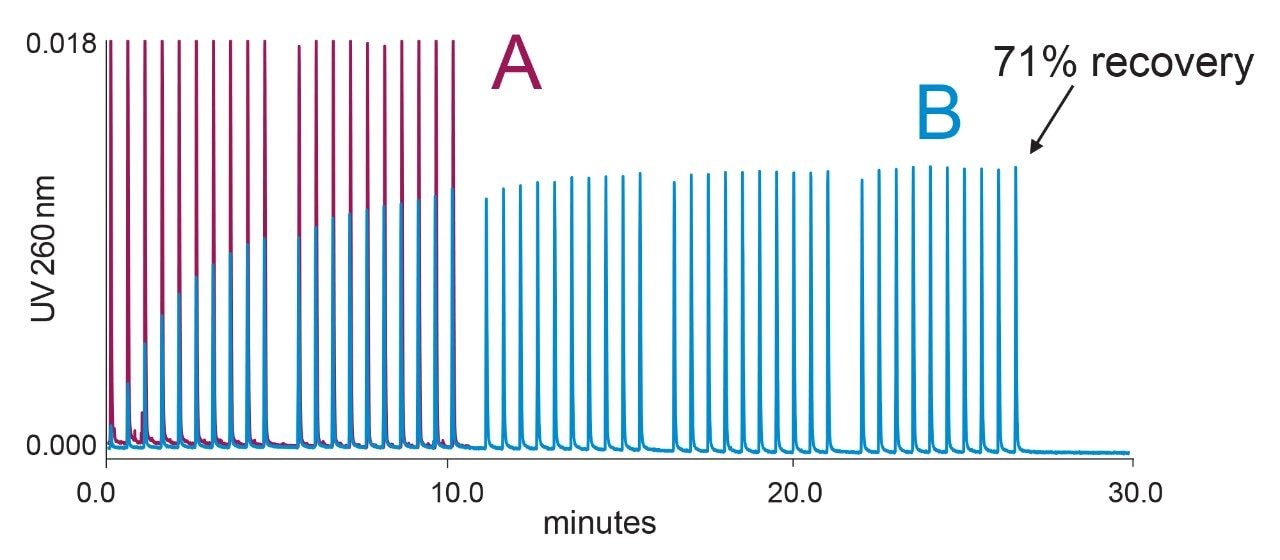
In a second study, we investigated oligonucleotide adsorption as a function of mobile phase pH. The mobile phases were aqueous 10 mM ammonium acetate adjusted to different pH values with acetic acid or ammonium hydroxide. Observations were again made on a 2.1 mm I.D. stainless-steel frit, using a new frit for each pH experiment. The results shown in Figure 2 demonstrate that oligonucleotide losses are more pronounced at acidic pH. For pH 7–8, conditions typically used for oligonucleotide separations, the sample loss is less apparent, however, challenges persist with the initial 10–20 injections for any given experimental condition. The data acquired for Figure 2 were used to estimate the cumulative oligonucleotide loss for the first 50 injections in each pH experiment. The loss was calculated from the peak areas for each injection on the stainless-steel frit (Afrit) and area APEEK obtained in the control experiment with a PEEK union.
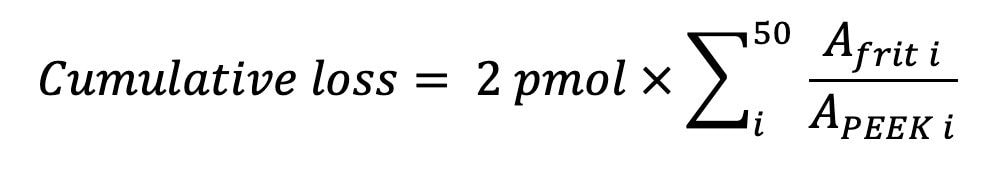
The cumulative oligonucleotide loss was 39.9, 24.1, 13.8, and 7.4 pmol in the pH 5, pH 6, pH 7, and pH 8 experiments, respectively. This estimate provides an important guideline for LC system/column conditioning. Analysts skilled in the practice of oligonucleotide analysis often perform a series of injections with a sacrificial oligonucleotide prior to the analysis of target sample. From the cumulative recovery results we conclude that UPLC systems and columns should be conditioned with a sample mass exceeding 10 pmol, preferably exceeding ~ 100 pmol of sample, in order to achieve efficient conditioning with pH 7–8 mobile phases. Experiments performed with 10 mM HAA buffers or 10 mM triethylammonium acetate (TEAA) buffers showed a similar magnitude of oligonucleotide adsorption (data are not shown).
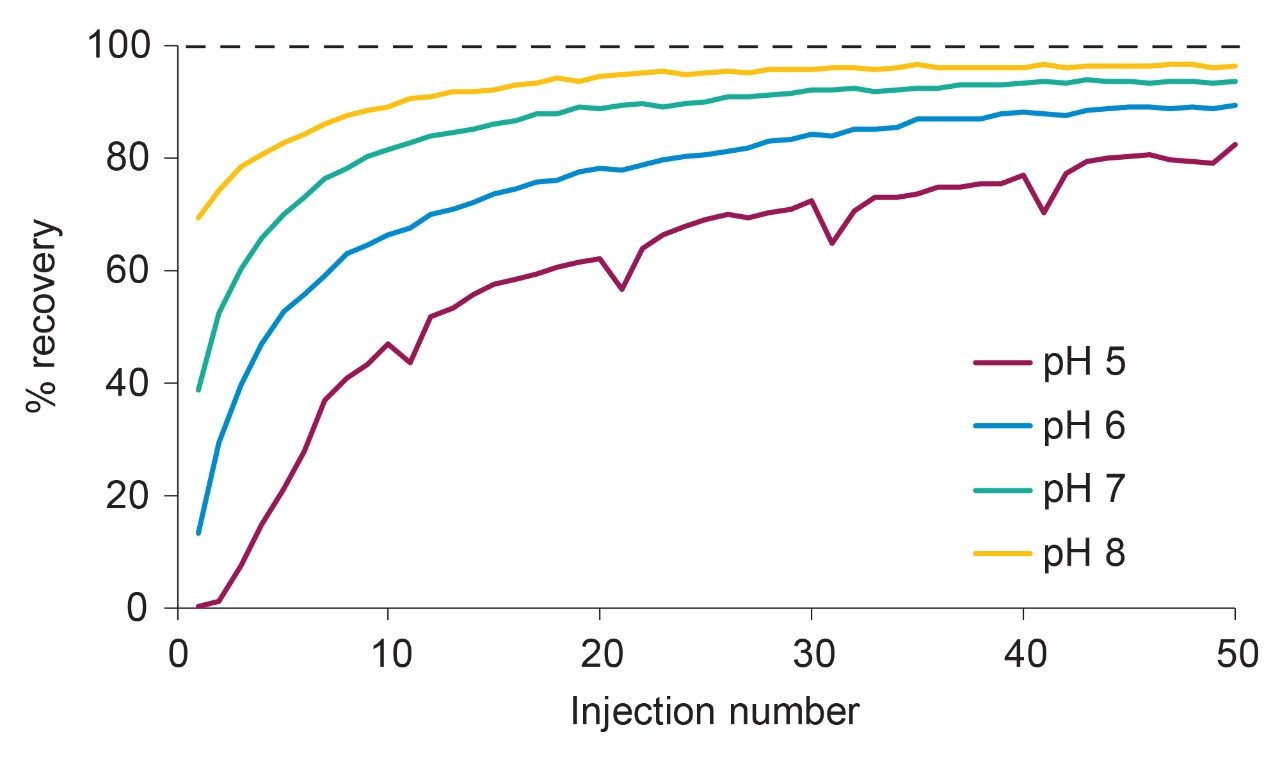
Inspection of Figures 1 and 2 reveals important insights that should be considered when dealing with sample conditioning of an LC instrument and column. First, the analyte signal does not reach full 100% recovery despite many repeat injections. Second, the “dip” in the recovery during the pH 5 test sequence (Figure 2) highlights the fact that conditioning is not permanent, but rather a transient effect. The dip occurs after the time gap inserted after each 10 injections. During the extended wash, a portion of the analyte adsorbed on the frit surface is washed away with an excess of the mobile phase. This “de-conditioning” phenomenon occurs in all experiments in Figure 2; however, it is most apparent at pH 5. The phenomenon of de-conditioning is illustrated in more detail in Figure 3 for results obtained using a pH 4.5 10 mM ammonium acetate mobile phase. Figure 3 reveals that more extensive washing leads to further de-conditioning of the frit. Gradual removal of the analyte from the metallic frit surface is also evident as peak tailing.
The analyte bleed from the metal surface is the reason why it is difficult to completely condition the stainless-steel hardware and achieve 100% recovery even after 50 sample injections (see Figure 1B). The analyte adsorbed on the frit is continually washed away during the experiment. Sample bleed likely contributes to unexpectedly high levels of sample carryover in oligonucleotide analyses. The column conditioning explains why sudden decreases in sample signal may occur after installation of a new column or after thoroughly washing an LC system. It is advisable to repeat the conditioning protocol after such events to achieve the results consistent with a previously established calibration curve.
The data presented in Figures 1–3 confirm that oligonucleotide LC analyses may be complicated by non-specific sample adsorption on metallic LC components. This phenomenon interferes with quantitative analyses, especially in sensitive LC-MS analyses. We observed that non-specific adsorption is not limited to stainless-steel surfaces; various degrees of sample loss were observed on titanium and non-ferric metal alloys (data not shown). To address these sample losses, Waters has developed MaxPeak High Performance Surfaces, used in MaxPeak Premier Columns. This technology utilizes hardware that has a barrier layer consisting of a highly crosslinked composition of hybrid-silica that is covalently immobilized onto the metal surface. [Lauber et al, Waters White Paper, 720006930EN, 2020] HPS technology is used to permanently deactivate column hardware, including the frits. MaxPeak Premier Columns offer a superior solution to the transient column conditioning protocols practiced in many laboratories.
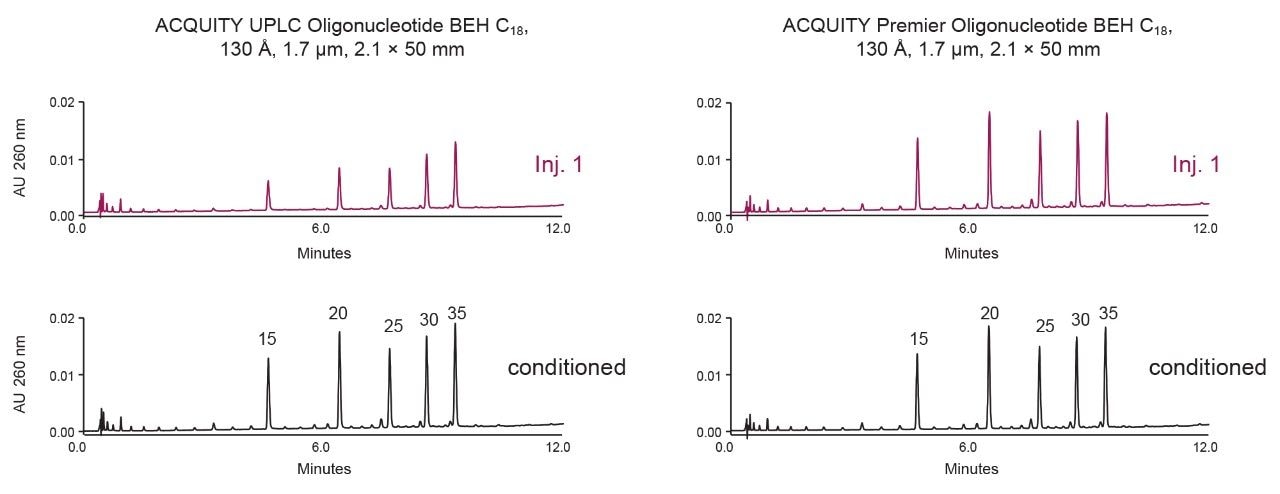
An ACQUITY UPLC Oligonucleotide BEH C18 Column was selected for high-resolution oligonucleotide analysis. This column is packed with 1.7 µm hybrid-silica particles that are well suited to separations performed at neutral to moderately basic pH and high temperatures. ACQUITY Oligonucleotide BEH C18 Columns are packed in stainless-steel hardware, so some sample losses should be expected due to adsorption on metallic surfaces. In contrast, ACQUITY Premier Oligonucleotide BEH C18 Columns are constructed with the same particles but with MaxPeak HPS column hardware. These two column types have been compared in the following work.
Figure 4 shows a comparison of the performance of the conventional ACQUITY UPLC stainless-steel versus MaxPeak Premier hardware columns (both are packed with identical chromatographic particles). As expected, minor loss of sample is observed for several initial injections, until the stainless-steel column is transiently conditioned. No apparent loss of sample was observed with the MaxPeak Premier Column; nearly complete oligonucleotide recovery was observed from the first sample injection.
As discussed above, the choice of pH is an important experimental consideration. Elevated pH enables improved recovery of oligonucleotides even with conventional column hardware. However, the choice of pH is limited by two factors. First, column stability is reduced at basic pH and shorter column lifetimes can be expected at higher pH. Second, the ion-pairing retention mechanism relies on the amine mobile phase additive being protonated to effectively form ion-pairs. The ion-pairing of oligonucleotides with the protonated amine is necessary for retention on reversed-phase columns. Because most of the amines suitable as ion-pairing agents have pKa values ~ 10.5, they become progressively less charged in mobile phases with pH values above 9.5, which leads to loss of oligonucleotide retention and resolution. We do not recommend ion-pair reversed-phase separations of oligonucleotides using mobile phases with pH values above 8.5.
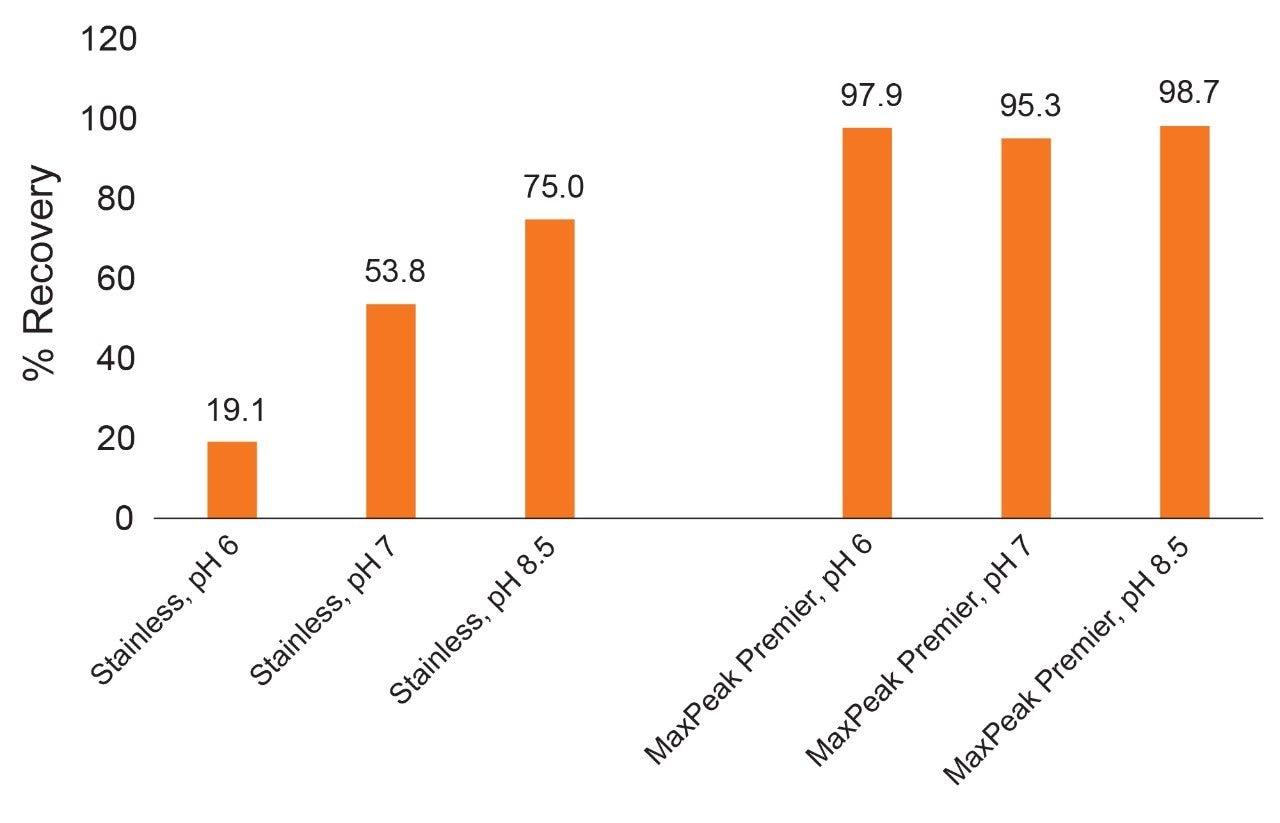
Recovery determinations for a 30 mer oligodeoxythymidine were made at three different pH values for six previously unused columns. A 25 mM HAA buffer system adjusted to pH 6, 7, and 8.5 was used to compare conventional stainless-steel columns with ACQUITY Premier Columns. It is important to consider that the LC system can also contribute to analyte loss due to non-specific adsorption on hardware components. The experiment shown in Figure 5 was carried out with a conditioned LC system to minimize these confounding effects. The recoveries observed in this experiment are shown in Figure 5. As predicted, elevated pH helps to mitigate analyte loss. However, the recovery remains incomplete even at pH 8.5 for stainless-steel columns. In comparison, MaxPeak Premier Columns give nearly complete recovery within the investigated pH range of 6–8.5. The minor analyte losses observed using the MaxPeak Premier Columns may be caused by incompletely conditioned LC system hardware.
This application note suggests best practices for achieving sensitive oligonucleotide analyses. We demonstrate that stainless-steel column hardware contributes significantly to non-specific oligonucleotide adsorption. Repeat sample injections gradually conditions metal surfaces in the column and LC instrument. However, we have observed that this conditioning is only transient; the sample is gradually desorbed from metal surfaces of the LC system (and column) leading to partial hardware de-conditioning with mobile phase flushing.
The pH of the mobile phase is an important experimental parameter. Elevated pH improves oligonucleotide sample recovery; however, this solution is not as effective as desired.
The recently introduced MaxPeak HPS Technology is a promising approach to minimize the undesirable interaction of oligonucleotides with metallic surfaces. ACQUITY Premier Columns incorporating this new technology are now available for the improved analysis of oligonucleotides. By employing this new technology, analysts can improve the quality and repeatability of oligonucleotide analyses, for both conventional LC-UV applications and, in particular, for sensitive LC-MS analyses.
720007012, Revised August 2023