This is an Application Brief and does not contain a detailed Experimental section.
Monitoring of N-nitrosamines in various matrices is of high interest, due to their potential carcinogenicity and detection in a variety of commodities and pharmaceuticals consumed by humans. These impurities require sensitive and selective detection in the sub-ng/mL range. The following method is capable of simultaneous detection and quantitation of six widely analyzed N-nitrosamines (NDMA, NDEA, NMBA, NDBA, NEIPA, and NDIPA) at or less than 0.1 ng/mL, using a highly sensitive and selective QTof acquisition mode. Referred to as Tof MRM, this acquisition mode provides selectivity through isolation of the analyte precursor m/z in the quadrupole, followed by a targeted increase in signal. Presented here are the UPLC chromatographic separation and LLODs, LLOQs, and linearity for the six N-nitrosamines analyzed using the Tof MRM method. Quantitative performance of this method is reported for NDMA and NDEA, demonstrating precise, accurate and repeatable measurement at both 1.25 and 12.5 ng/mL quality control (QC) levels. Furthermore, the use of the UNIFI Scientific Information System for acquisition and data processing offers a modern platform for GxP-compliant HRMS analysis of the investigated N-nitrosamines.
N-nitrosamines are small molecule compounds generally formed as reaction products between nitrate and amines1. These compounds are known potential carcinogens2, therefore warranting active monitoring to mitigate human exposure. They can be found occurring in natural water systems2, tobacco products2, and as impurities in pharmaceutical products1,3,4. Particular attention has been drawn to the 2019 detection of N-nitrosodimethylamine (NDMA) in ranitidine, a histamine-2 blocker available over the counter for the treatment of gastroesophageal reflux disease and stomach/intestinal ulcers5. NDMA, N-nitrosodiethylamine (NDEA) and N-nitroso-N-methyl-4-aminobutyric acid (NMBA) have also been found as impurities in angiotensin II receptor blocker (ARB) drug products (the “-sartan” class of drugs) which are used to treat heart failure and high blood pressure4. It is suspected in the ARB drugs that three other N-nitrosamines, N-nitrosoethylisopropylamine (NEIPA), N-nitrosodiisopropylamine (NDIPA) and N-nitrosodibutylamine (NDBA) could also be present4.
Active monitoring of these harmful compounds at relatively low concentrations (ng/mL or lower) is of high priority and relies on highly sensitive and selective analytical methods. Use of liquid chromatography-mass spectrometry (LC-MS) has been successfully applied for the confident identification and quantification of N-nitrosamines in a range of matrices1,2. Specifically, use of targeted acquisitions such as multiple reaction monitoring (MRM) on tandem quadrupole MS has proven to attain lower limits of quantification (LLOQ’s) at or below 0.1 ng/mL for N-nitrosamines6,7. Methods using high resolution MS (HRMS) platforms for N-nitrosamines analysis have also been explored4,8 and afford additional specificity through the use of accurate mass measurement.
Here, we demonstrate the ability to attain both sensitive and selective detection of six N-nitrosamines (NDMA, NDEA, NDIPA, NEIPA, NMBA, and NDBA) using Tof-MRM acquisition on the Xevo G2-XS QTof. This methodology isolates the known precursor molecule in the quadrupole, followed by targeted enhancement of a specified precursor or product ion(s). This combination produced increased selectivity and sensitivity of the N-nitrosamines over typical full scan MS, with the added benefit of specificity of accurate mass measurement inherent to HRMS.
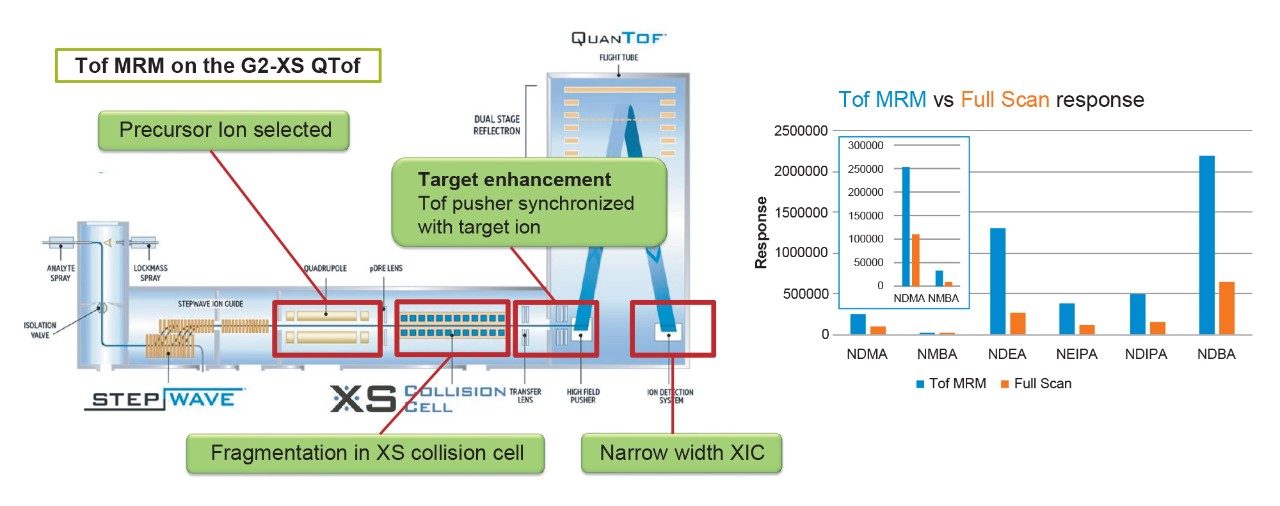
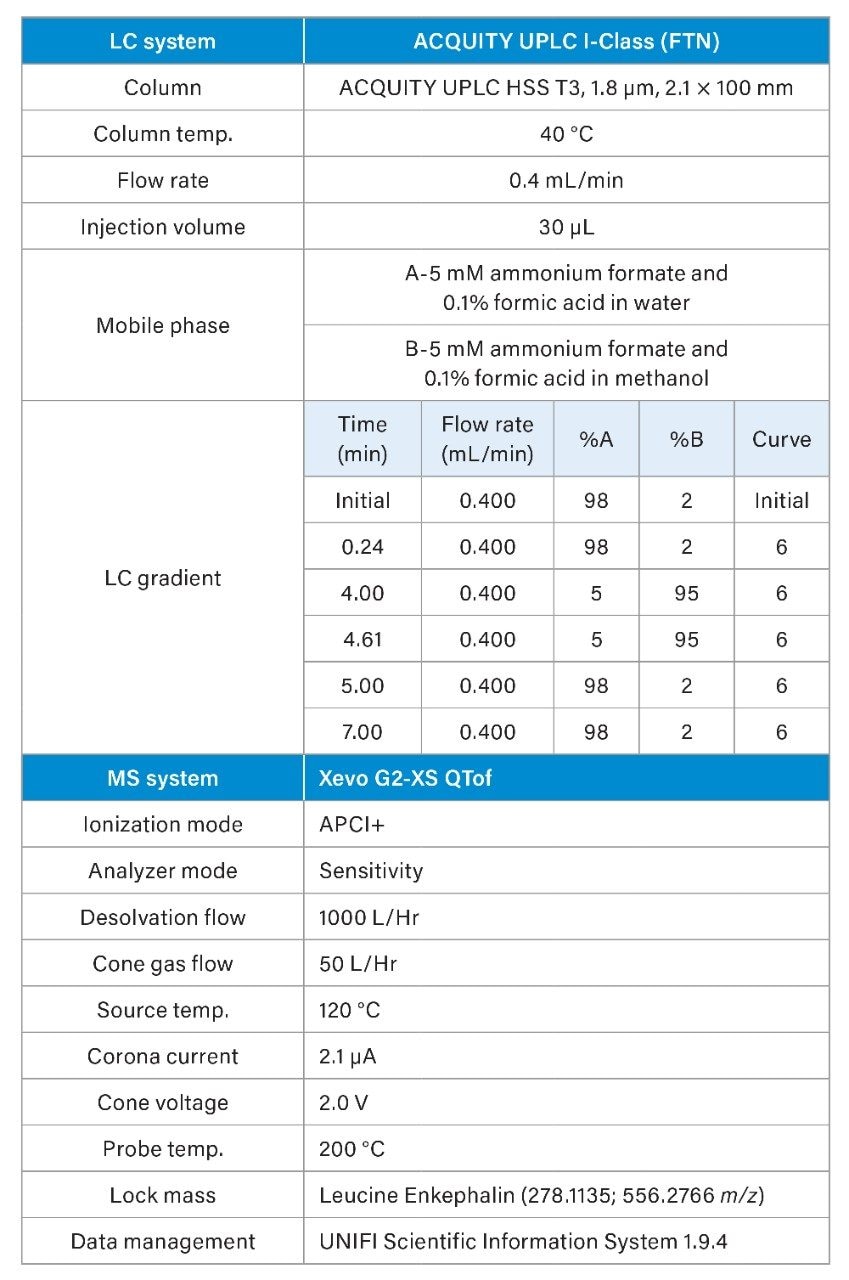
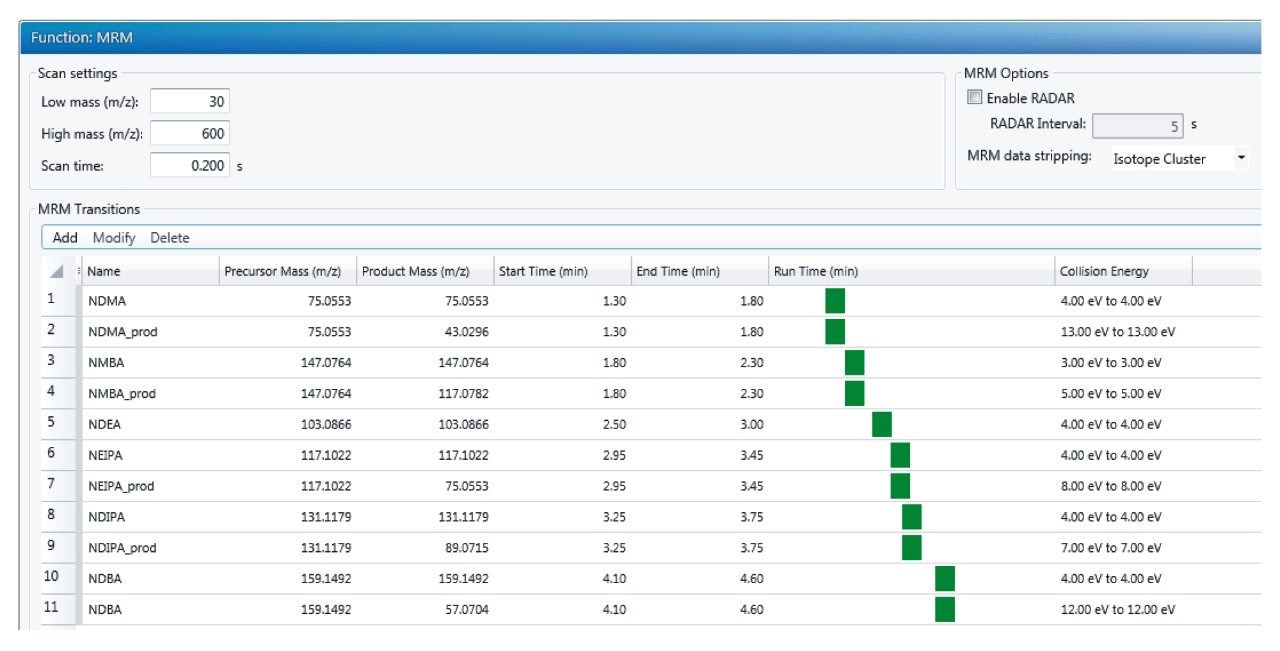
Analysis of NDMA, NMBA, NDEA, NDIPA, NEIPA, and NDBA was carried out using the ACQUITY UPLC I-Class and Xevo G2-XS QTof MS with the Ion Sabre II APCI probe. Data acquisition, processing and review was carried out using the UNIFI Scientific Information System, a comprehensive informatics platform which contains features for GxP compliance such as data traceability and advanced security9. Separation of the 6 nitrosamines (shown in Figure 1) was achieved using a previously described6 LC method using the ACQUITY UPLC HSS T3 Column (p/n 186003539). Table 1 shows the LC-MS conditions employed for this analysis. Following retention time and optimal ion transmission settings determination, the development of a targeted QTof method was performed using Tof MRM acquisition. Tof MRM operates by time-segmented selection of the specified precursor mass, which is isolated in the quadrupole and then sent to the collision cell where either ion transmission or collision induced dissociation (CID) occurs. Then, user specified m/z value(s) are subjected to targeted signal enhancement through pusher synchronization in the Tof region10. Accurate mass measurement is then achieved for all ions, and an extracted ion chromatogram is generated by the UNIFI Software for the specified m/z values. For the N-nitrosamine compounds in this study, targeted enhancement was performed for NDMA, NMBA, NDIPA, NEIPA, and NDBA of both precursor and selected product ions. For NDEA, targeted enhancement was performed only for the precursor ion, owing to an abundance of signal. Figure 2 shows the final Tof MRM acquisition method, for which optimized collision energies were determined experimentally as part of method development.
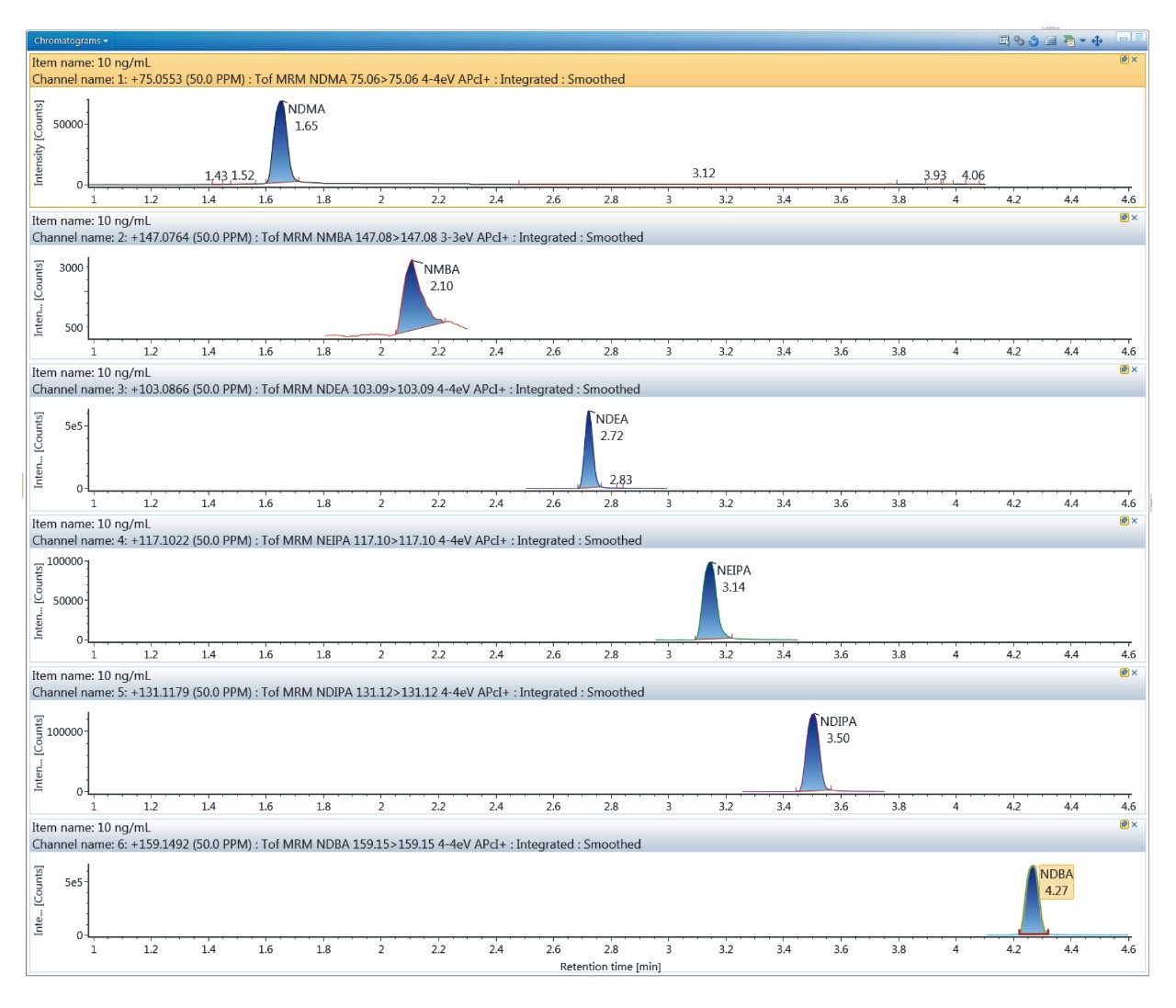
Quantitative performance using the developed method was assessed using two dilution series ranging from 0.01-100 ng/mL, with triplicate injections at each point (6 injections total). The resulting lower limits of detection and quantification (LOD/Q), linear range and R2 values are described in Table 2, affording sub-ng/mL detection capability for the 6 N-nitrosamines studied here. Further measurement of two QC levels, 1.25 ng/mL and 12.5 ng/mL, are shown in Figure 3 for NDMA and NDEA. Here, both compounds are shown to have accurate and precise measurement at both levels. Calculated concentrations were averaged across the 5 replicate injections, and for both compounds are within 15% of the actual concentration (Figure 3a and b). Additionally, %RSDs were <7% for NDMA and NDEA at both QC levels, as based on calculated concentration. As this data demonstrates, reliable quantitation for these two high-profile N-nitrosamines is achieved using the developed UPLC-Tof MRM MS method.
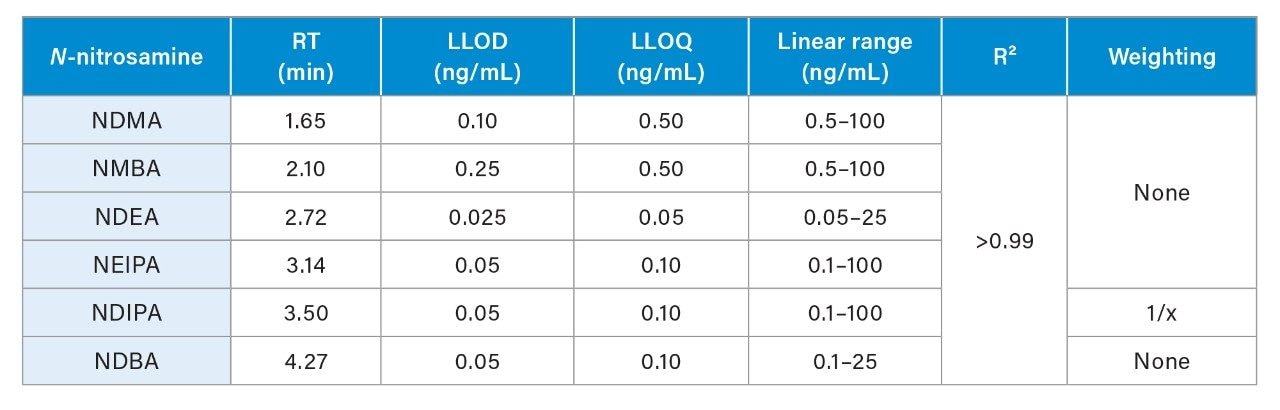
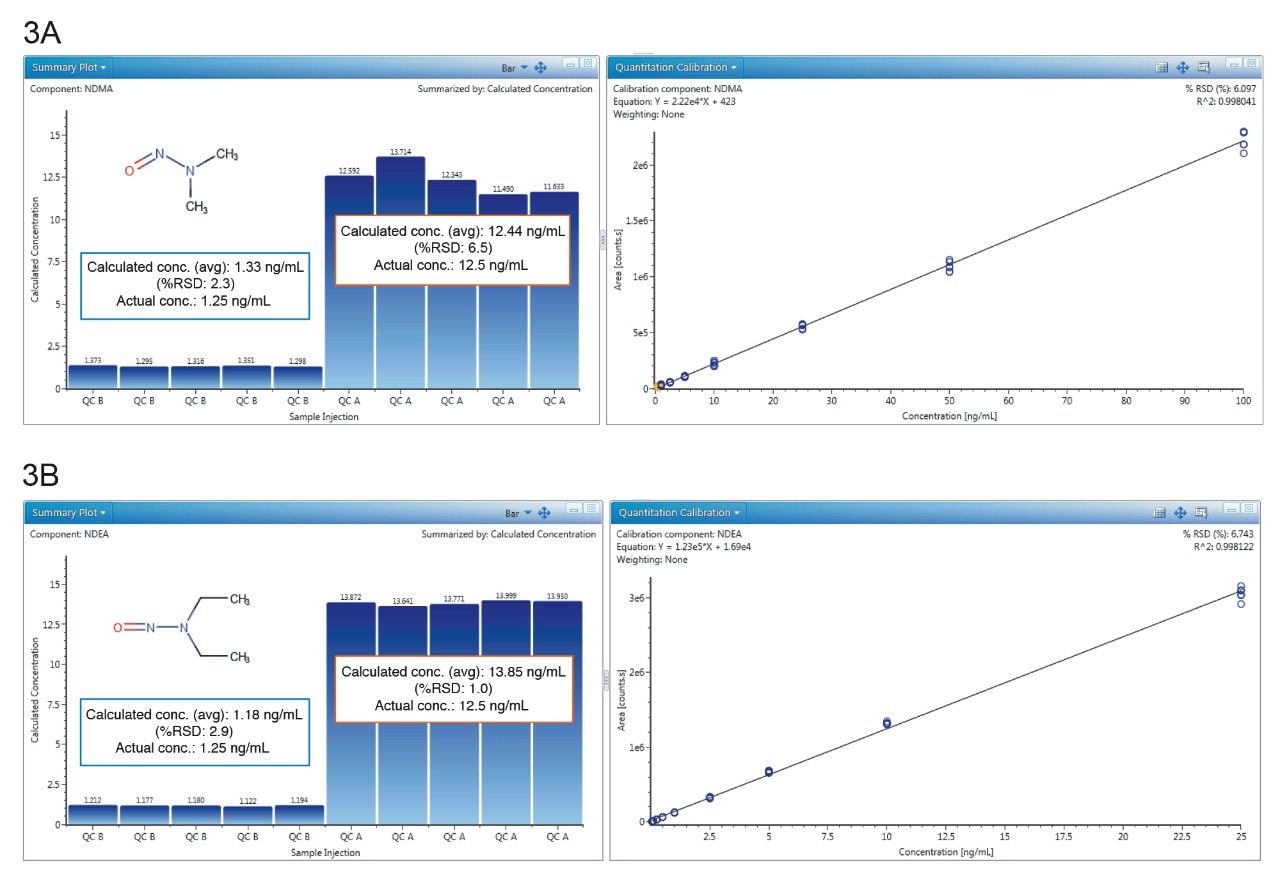
Figure 3a. NDMA summary plot of calculated concentrations (mean accuracy) at QC level A (12.5 ng/mL spike) and B (1.25 ng/mL spike), and calibration curve (0.1–100 ng/mL).
Figure 3b. NDEA summary plot of calculated concentrations (mean accuracy) at QC level A (12.5 ng/mL spike) and B (1.25 ng/mL spike), and calibration curve (0.025–25 ng/mL).
Methods for the analysis of potentially carcinogenic N-nitrosamines require sufficient sensitivity to accurately measure at sub-ng/mL concentrations. Here, we have demonstrated that using Tof MRM on the Xevo G2-XS QTof MS coupled with UPLC chromatographic separation, LLOQ levels at or below 0.5 ng/mL for NDMA, NDEA, NMBA, NDIPA, NEIPA, and NDBA were achieved. Quantitative testing of the method also demonstrated reliable and accurate measurement (within 15%) of two QC levels in the low ng/mL range for NDMA and NDEA. Overall, the ACQUITY UPLC coupled to Xevo G2-XS QTof MS with the UNIFI Scientific Information System offers a modern platform for GxP-compliant, sensitive, selective, and specific HRMS quantitation of N-nitrosamines.
720006951, July 2020