This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the Waters BioAccord system-to-system reproducibility for intact mAb glycoprofiling.
In 2017, the FDA published a paper entitled “A Retrospective Evaluation of the Use of Mass Spectrometry in FDA Biologics License Applications”¹ that highlights the increased use of mass spectrometry for the characterization of protein-based biotherapeutics since 2000. Most recently, there has been a concerted effort from the pharmaceutical industry to expand the use of liquid chromatography and high-resolution mass spectrometry (LC-MS) in late stage development and QC environments. However, the complexity of LC-MS operation requires highly skilled scientists to generate and collect meaningful data. In addition, LC-MS data variability from instrument to instrument poses challenges in developing MS-based assays (such as the Multi-Attribute-Method) in process development and QC laboratories. There is a pressing need for ease-of-use and robust analytical systems for routine analysis of biotherapeutic drug molecules.
The launch of the BioAccord System is the first step to address the industry’s needs. As illustrated in previously published application notes and technology briefs,2-9 the BioAccord System meets many of these requirements. In this study, we focused on the reproducibility of the BioAccord System using well-designed experiments. This two-part study includes intact mass analysis and peptide mapping/monitoring. This technology brief focuses on the first part: intact mass analysis.

The BioAccord System is physically comprised of an ACQUITY UPLC I-Class PLUS System configured with an optical detector (TUV or FLR) coupled in-line with an ACQUITY RDa Detector (a compact time-of-flight mass detector). The system operates on the UNIFI Scientific Information System platform, providing streamlined workflow solutions with automated data acquisition, processing, and reporting (including automating relative abundance calculations). This system is purposely designed for routine biopharmaceutical analysis in both regulated and non-regulated laboratories.
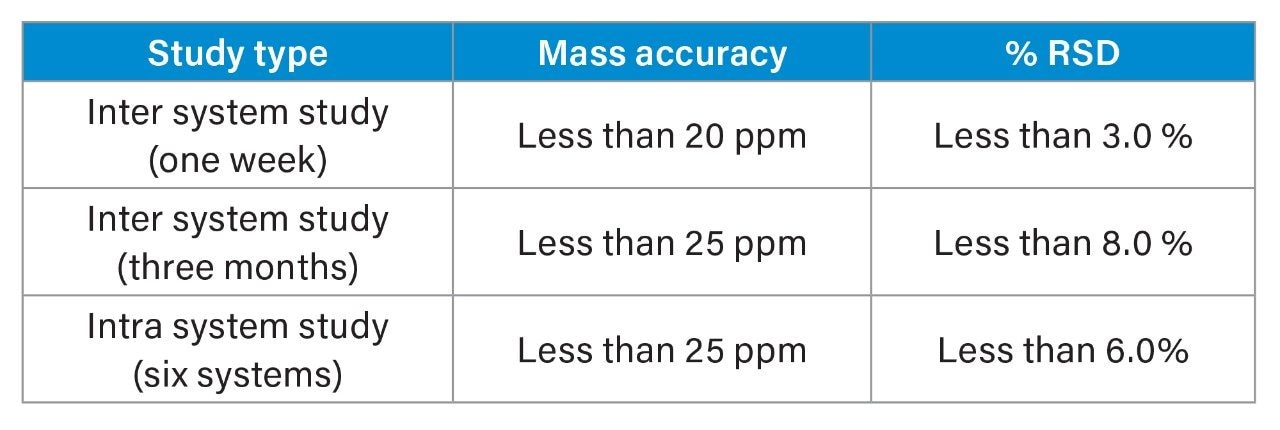
Intact mass analysis by RPLC-MS is a simple and fast way to obtain molecular weight and relative abundance of major glycoforms for mAb products. All the intact mass analyses in this study were carried out using the Waters Humanized mAb Mass Check Standard, originated from NIST mAb reference material (p/n: 186009152). Milli-Q Water (400 µl) was added to 80 µg of intact NIST mAb to produce a solution of 0.2 µg/µL. 0.8 µg was injected for analysis. An ACQUITY UPLC BEH C4, 1.7 µm, 2.1 mm x 50 mm (p/n: 186004495) was used at 80 °C. Mobile phase A was Milli-Q water with 0.1% formic acid and mobile phase B was acetonitrile with 0.1% formic acid. A linear gradient of 95% A to 15% A from 1 min to 3.7 min was run at 0.4 mL/min, with a total run time of 7 min. The RDa detector was operated in positive full-scan mode (at 2 Hz) with a mass range of 400 to 7,000 m/z. The cone voltage was set at 70 V, capillary voltage at 1.5 KV and the desolvation temperature was kept at 550 °C. A threshold of ≤25 ppm mass error was used to confirm the glycosylation forms of the intact monoclonal antibody for all the experiments (Table 1 summarizes mass accuracy).
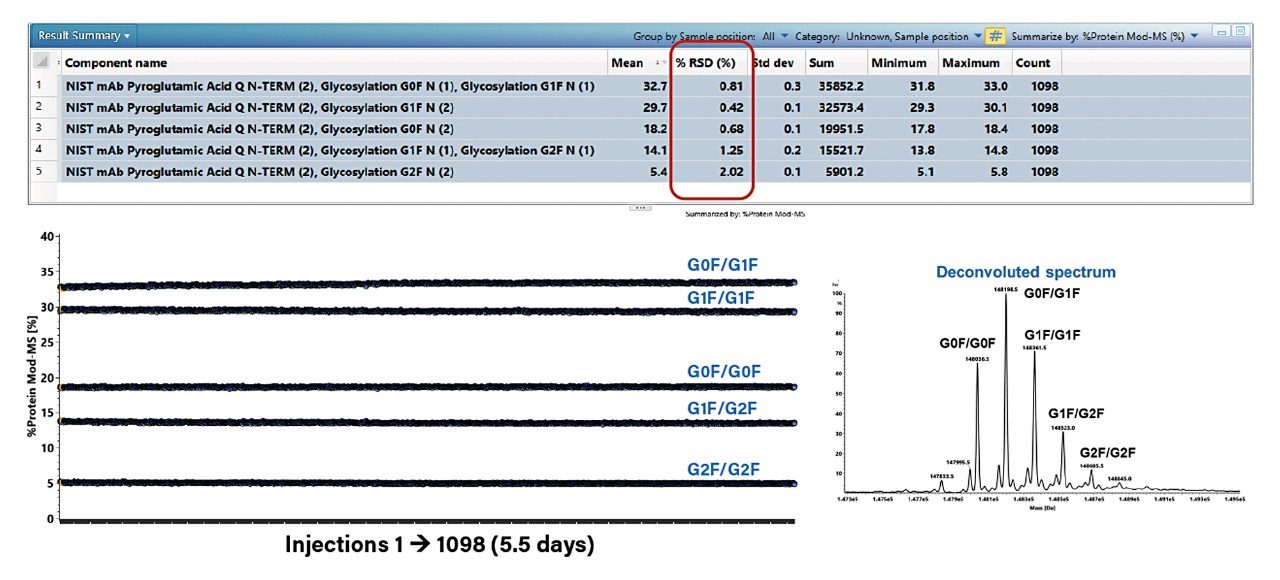
Figure 1 displays inter-system reproducibility for intact mass, focusing on %RSD of relative percent of major glycoforms. The experiment was run continuously for 5.5 days (1098 injections) on the BioAccord System. Relative abundances of the five major glycosylation forms (G0F/G0F, G0F/G1F, G1F/G1F, G1F/G2F, and G2F/G2F) were calculated via automated data processing in UNIFI using MaxEnt 1 charge deconvolution. The charge deconvoluted spectrum is displayed along with the %RSD trending plot for the relative abundance of the five glycoforms shown in Figure 1. The result summary table shows that the %RSD of the five major components is about 2%, demonstrating that the BioAccord System can operate continuously with stable and consistent ion signal for a week without the need of recalibration or tuning.
A second experiment was performed to show the reproducibility of the intact mass analysis on one of the systems over a three-month time period. In this work, instead of conducting the experiment continuously on the system, 10 injections were run each day on day 1, 2, 3, 5, 8, 14, and 90. Then the relative abundance of the same five glycosylation forms was plotted in the summary trend plot. The experiment results (data not displayed) show that the %RSD of the relative abundance of the five major glycoforms is less than 8.0%, demonstrating that the BioAccord System is highly stable and robust over a long period of time (three months in this case).
After evaluating the inter-system reproducibility for intact mass analysis, we conducted an intra-system reproducibility study. In this work, we ran the same experiment on six different BioAccord Systems for 10 injections on each system (eight separate experiments in total). Results are shown in Figure 2 (systems one and six had duplicated data sets). The experimental results show that the %RSD of the relative abundance of the five major glycoforms was found to be less than 6.0%, highlighted in red in the result summary table, demonstrating that these six BioAccord Systems have excellent intra-system reproducibility for intact mass analysis.
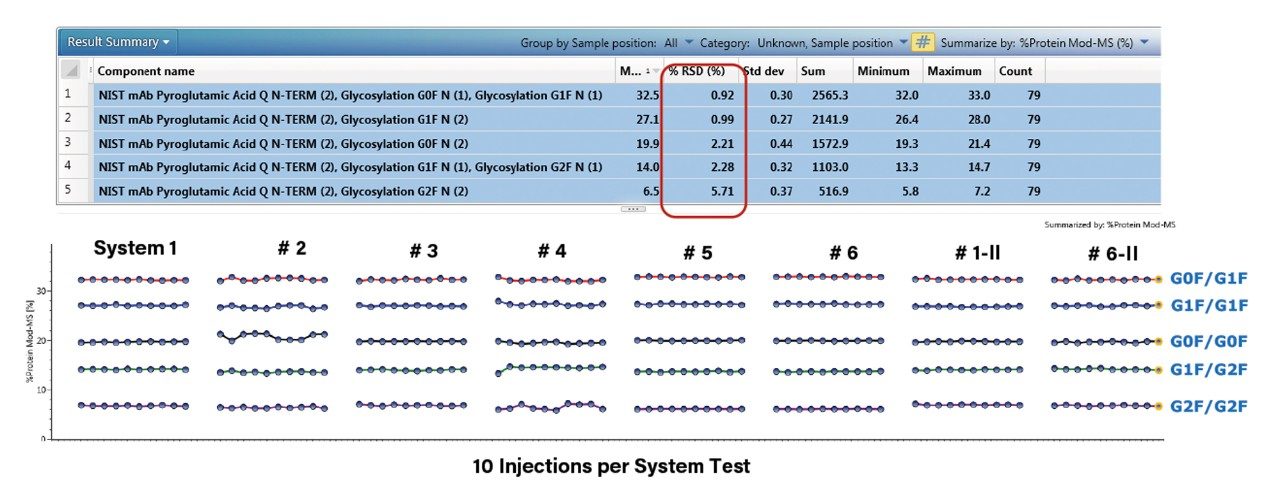
Table 1 summarizes the experiment results of the major glycoform mass accuracies and the %RSD of the relative abundance, as described in this technology brief.
The inter-system studies showed that the BioAccord System had excellent reproducibility in glycoform profiling, not only for five and half days continuously run, but also for a 90-day time course study. For the intra-system study, the experiment results showed that the BioAccord System had a high degree of consistency across six different systems. The relative abundance of the top five major glycoforms were found to be less than 6.0% RSD.
Both the inter- and intra-system reproducibility data generated from BioAccord Systems adds confidence in the deployment of MS-based assay in late stage development and QC labs.
720006722, January 2020