This application note shows the separation and quantification of empty and full capsids on a Waters Protein-Pak Hi Res Q AEX Column.
Recombinant adeno-associated virus (AAV) vector has been increasingly used as a vehicle in gene therapy to deliver therapeutic genes.1 During the AAV biomanufacturing process, high percentage of capsids that do not contain the desired DNA (empty capsids) are also produced and are therefore unable to provide therapeutic benefits. It has been reported that empty capsids can reduce transduction efficiency and induce unnecessary immune response.2 On the other hand, recent data show that empty AAV capsids can enhance gene transfer.3 Whether beneficial or detrimental, full versus empty capsid formulations need to be monitored to ensure consistent product quality and efficacy.
Multiple techniques have been used to quantify empty capsids in AAV samples. Analytical ultracentrifugation (AUC),4 UV absorbance spectrophotometry,5 and electron microscopy6,7 are extensively used in measuring empty/full ratio of AAV particles. AUC, which measures the sedimentation rate of the particles, is the gold standard in this application area. However, hours are required for skilled AUC proficient analysts to interpret the data and AUC deployment can consume hundreds of microliters of highly valued sample. In addition, an assay based on AUC analysis is challenging to validate. Static spectrophotometry that measures at 260 nm versus 280 nm has also been used to estimate empty from full capsids. Although a quicker method, the accuracy of the results can be compromised by sample matrix interferences. In addition, sizeable amount of sample is required to obtain adequate absorbance values. Transmission electron microscopy (TEM) and cryogenic electron microscopy (Cryo-EM) provide direct visualization and counting of the empty and full particles, but the quantification heavily relies on image quality and field selection. The most recently used technique for this analysis is charge detection mass spectrometry (CDMS).8 This MS method directly measures the molecular weight of the particles and has been shown capable of accurately measuring empty, partial, and full AAV particles. Currently, no commercial CDMS instrument is available.
Anion-exchange chromatography (AEX) was reported to have the potential of separating empty and full capsids and it has been suggested that this separation is driven by differences in their relative surface charges.9,10 This technique consumes small amounts of sample, and can yield robust and reproducible results using standard LC instrumentation. In this application note, we show the separation and quantification of empty and full capsids on a Waters Protein-Pak Hi Res Q AEX Column.
The AAV8 samples containing either filled capsids (full) or null capsid (empty, without Green Fluorescent Protein [GFP] genes) were injected directly onto the anion-exchange column without dilution. The concentration of the full AAV8 capsid is 8.5 E12 vg/mL, while that of the empty capsid is 1.67 E12 capsids/mL. The samples were supplied by BioReliance Corporation, Rockville, MD, USA.
In a separate set of samples, AAV1, AAV2, AAV5, AAV6, AAV8, AAV9 are all provided as full capsids with GFP genes inside (Vigene Biosciences, Rockville, MD, USA). All samples were injected directly onto the anion-exchange column without dilution. The concentrations of the samples range from 1.3 × 1013 to 6.7 × 1013 GC/mL.
|
System: |
ACQUITY UPLC H-Class Bio |
|
Sample temp.: |
10 °C |
|
Analytical column temp.: |
30 °C |
|
Flow rate: |
0.35 mL/min or 0.4 mL/min |
|
Injection volume: |
0.2–6 μL |
|
Column: |
Protein-Pak Hi Res Q, 5 μm, 4.6 × 100 mm (p/n: 186004931) |
|
Detection: |
Fluorescence detector: excitation: 280 nm, emission: 350 nm; ACQUITY UPLC TUV Detector with 5 mm titanium flow cell, Dual wavelength: 280 nm and 260 nm |
|
Sample vials: |
Polypropylene 12 × 32 mm Screw Neck, with Cap and Pre-slit PTFE/Silicone Septum, 300 μL volume (p/n: 186002639) |
|
Mobile phase A: |
100 mM Tris-HCl |
|
Mobile phase B: |
100 mM Tris base |
|
Mobile phase C: |
1 M salt (please see text and figures) |
|
Mobile phase D: |
Water |
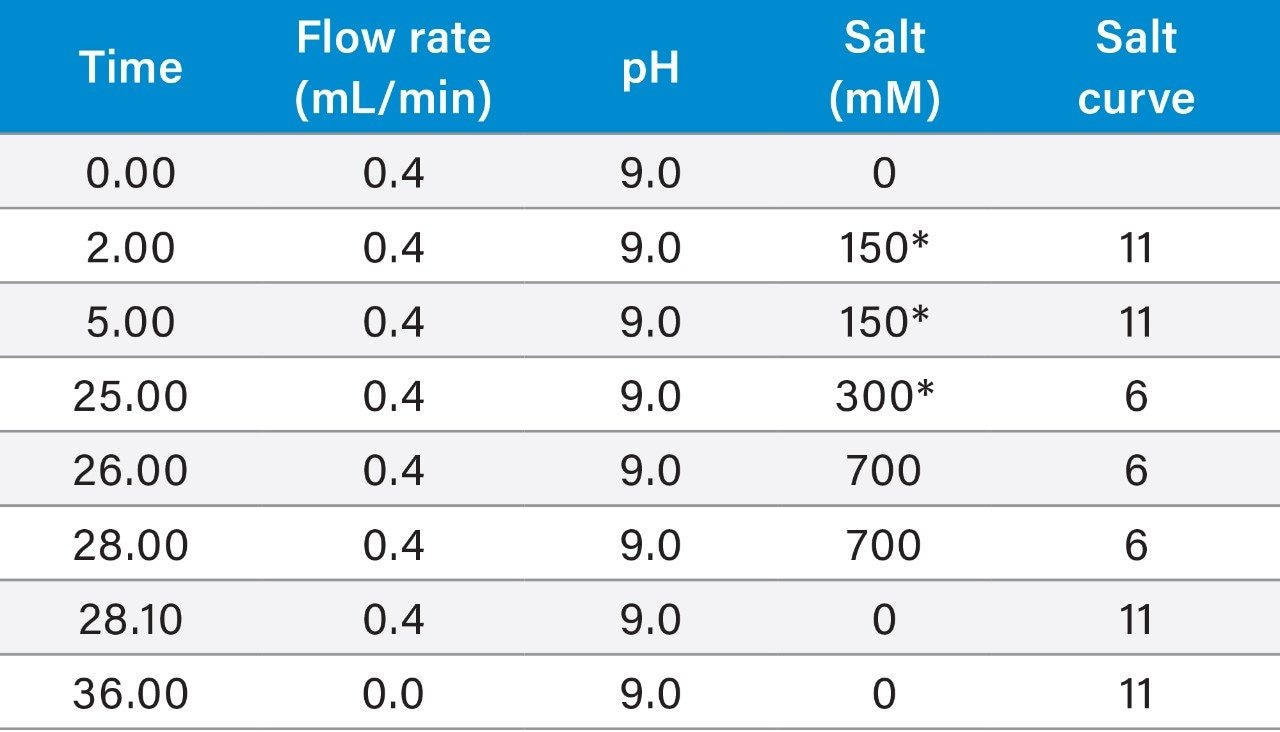
|
Mobile phase A: |
250 mM Bis-tris propane, pH 9 or 10 |
|
Mobile phase B: |
20 mM MgCl2 |
|
Mobile phase C: |
1 M tetramethylammonium chloride |
|
Mobile phase D: |
water |
|
Data management: |
Empower 3 (FR 4) |
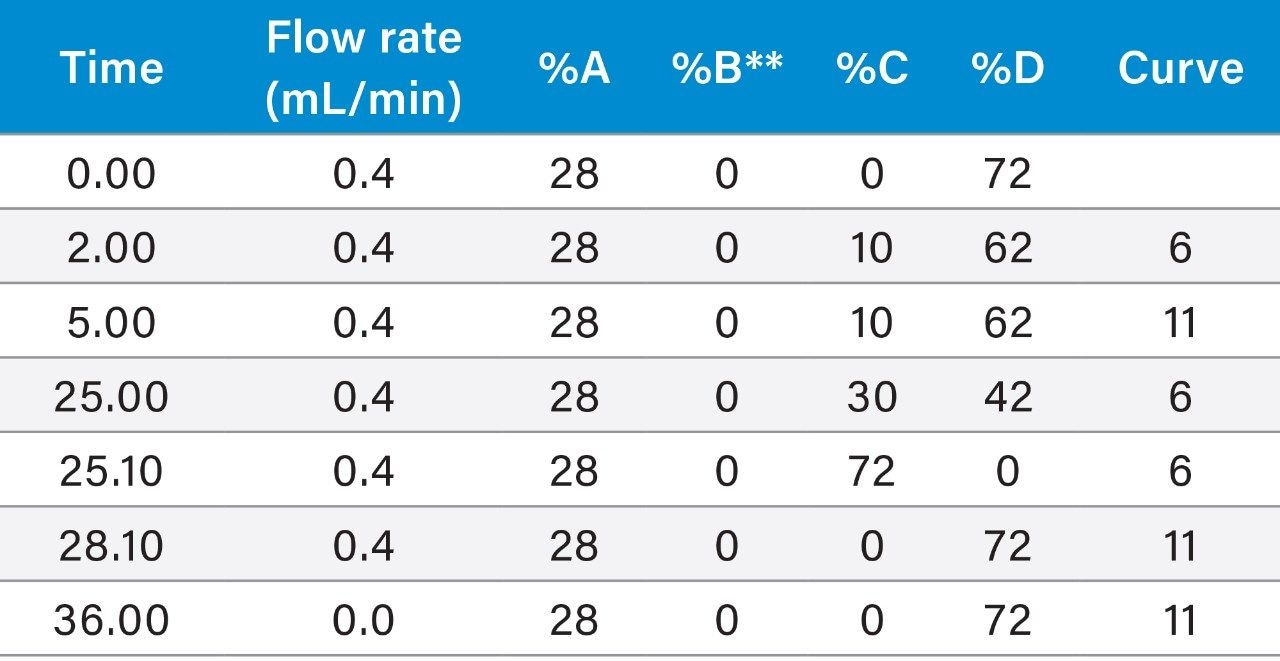
AAV8 empty and full capsid mixtures were separated on a Protein-Pak Hi Res Q Column using gradient methods as described above (Figure 1). In agreement with the previously reported results,9,10 the full capsids eluted later than the empty capsids on the strong anion-exchange column, thereby, indicating that the full capsids have more anionic charge than the empty capsids, likely due to their encapsulated DNA.
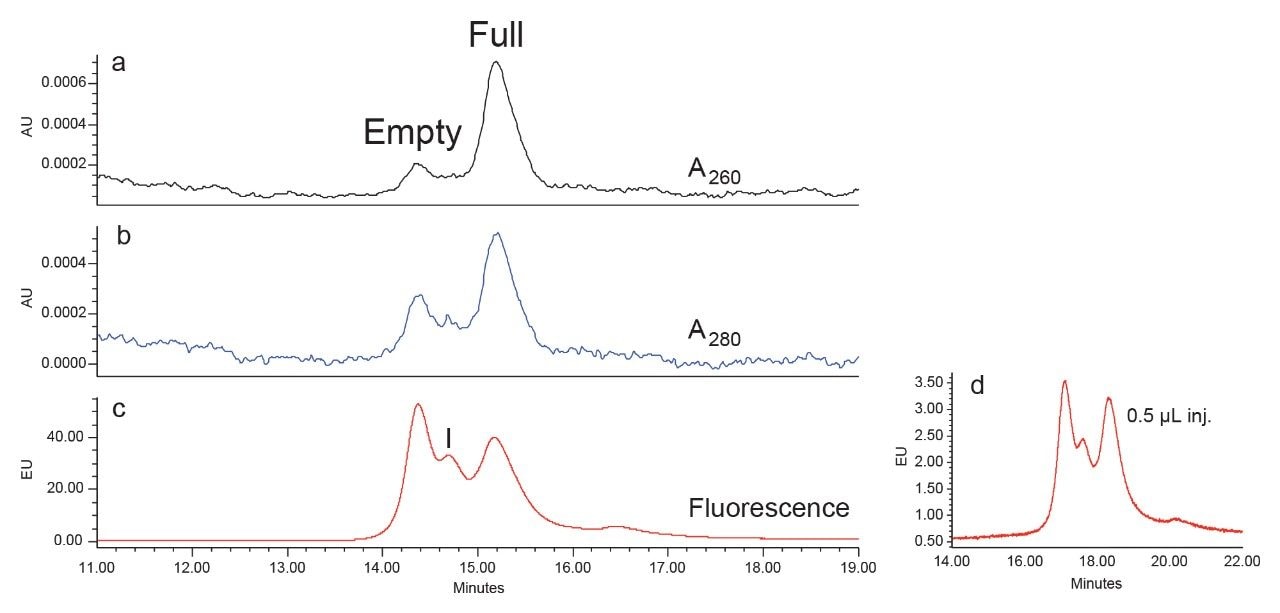
Figures 1a and 1b showed the UV detection at 260 nm and 280 nm, respectively. Consistent with the literature,9 for the full capsid, the peak area is greater at 260 nm than that at 280 nm; while for the empty capsid, the peak area is less at 260 nm than that at 280 nm. This is because at 280 nm, the capsid proteins have stronger absorbance than DNA, while at 260 nm, viral DNA absorbs stronger than the capsid proteins.
Shown in Figure 1c is the signal from the fluorescence detection with the same separation method. Notice that the fluorescence signal was much higher than those from the UV detection, which has been shown in previous reports.9 Even with a 0.5 μL injection of the sample (~4*109 vg on column), the signal is well above the detection limit (Figure 1d). For this reason, only the fluorescence detection data are shown in the rest of this application note.
The intermediate peak eluting between the empty and the full capsid (Figure 1c, Peak I) appears to be present in the empty capsid sample and is presumed also to be predominantly empty based on the electron microscopy data which measured a full capsid content of 0.5% for the empty sample. Additionally, the A260/A280 UV ratio of the intermediate peak is consistent with the value observed for the empty peak (data not shown). Based on these results there is no substantial evidence that the intermediate peak represents capsid that contains partial DNA content. It is assumed that the protein charge heterogeneity contributed by either primary structure or conformational changes in the AAV8 capsids has nearly the same degree of impact to the AEX separation as the ssDNA content. It is also possible that higher concentration of NaCl may cause conformational or structural changes of the empty capsids, resulting in changes in surface charge. These results are consistent with previously published AAV-AEX purification results, which shows that empty, full, and partially full AAV are observed throughout the AEX elution of several serotypes.11
While the initial conditions showed that Protein-Pak Hi Res Q Column was able to effectively separate the empty and the full capsids, method development was carried out to further optimize and evaluate the robustness of the separation.
The first parameter of the optimization is usually the mobile phase pH. The initial testing showed that at pH 9 a better resolution between full/empty capsids was achieved than at pH 8.5 using Tris buffer (data not shown).
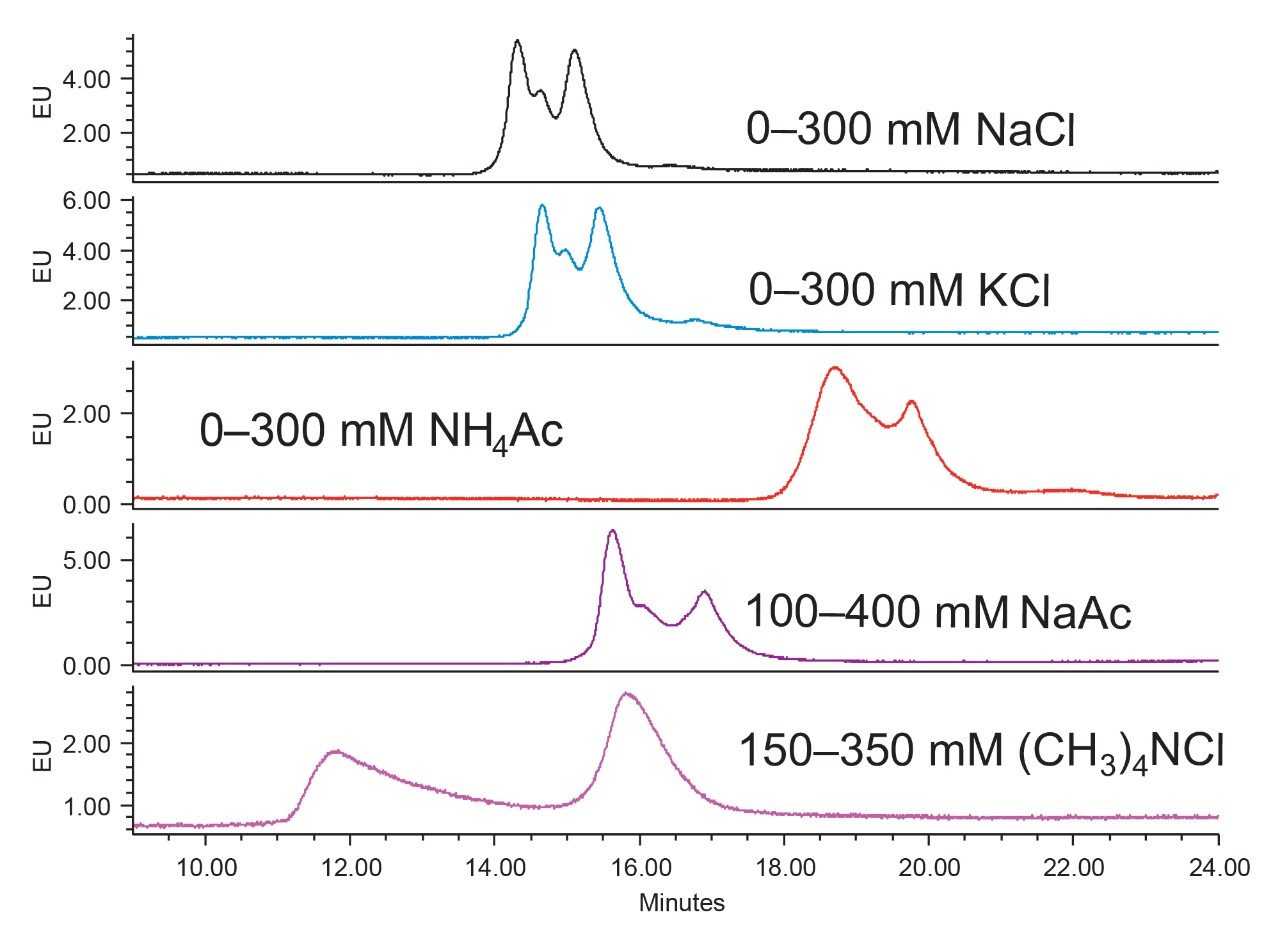
Figure 2 shows the chromatograms of using different types of salt in a Tris buffer (pH 9) for separating the empty and the full capsids. Differences in retention time and resolution between the empty and the full capsids were obtained using different types of salt. The empty/full ratio also varied, likely due to different extent of resolution between the empty and the full capsids. Among all the salts tested, tetramethylammonium chloride delivers the best resolution between the empty and the full capsids.
Both Tris buffer and Bis-tris propane buffer were tested for their impact on the separation at pH 9. Both conditions provided an effective resolution between the empty and the full capsid, with Bis-tris propane providing a slightly better resolution and a better baseline stability (data not shown).
When the separation in pH 9 and pH 10 is compared using the Bis-tris propane buffer, pH 9 yields a better separation result (Figure 3).
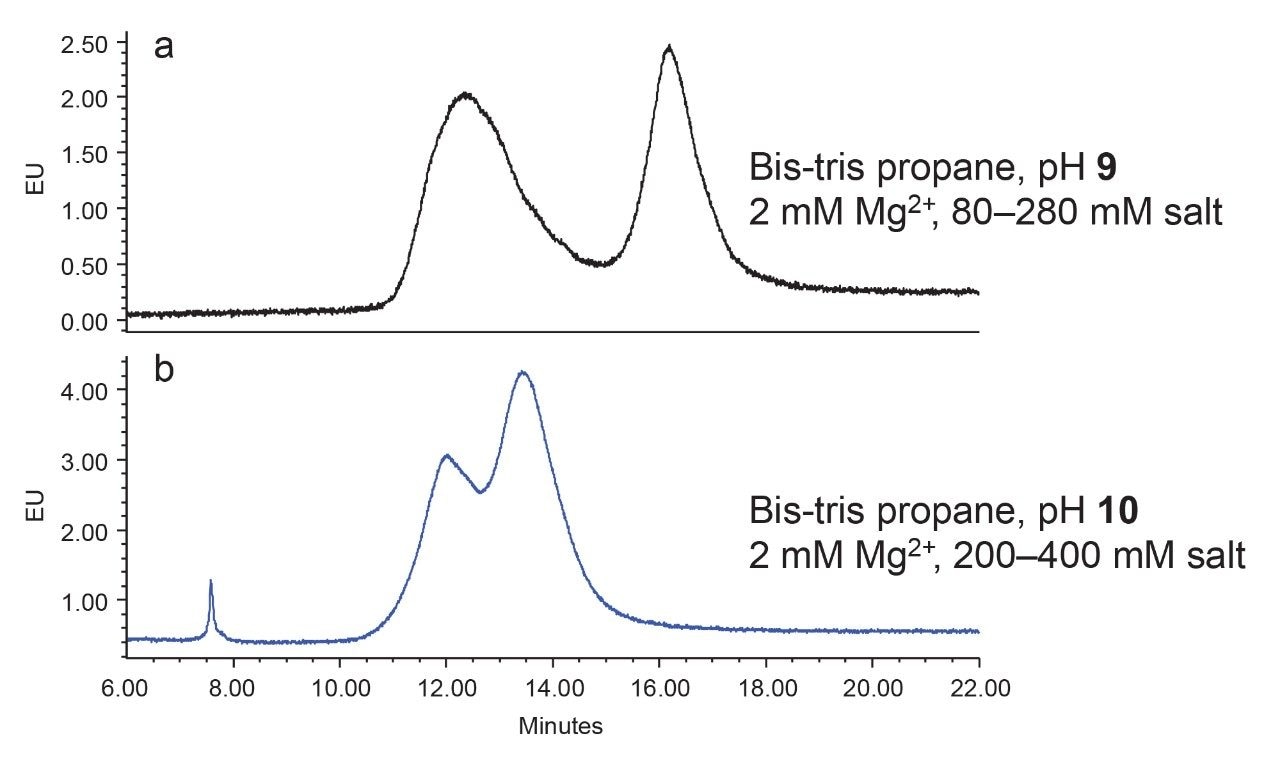
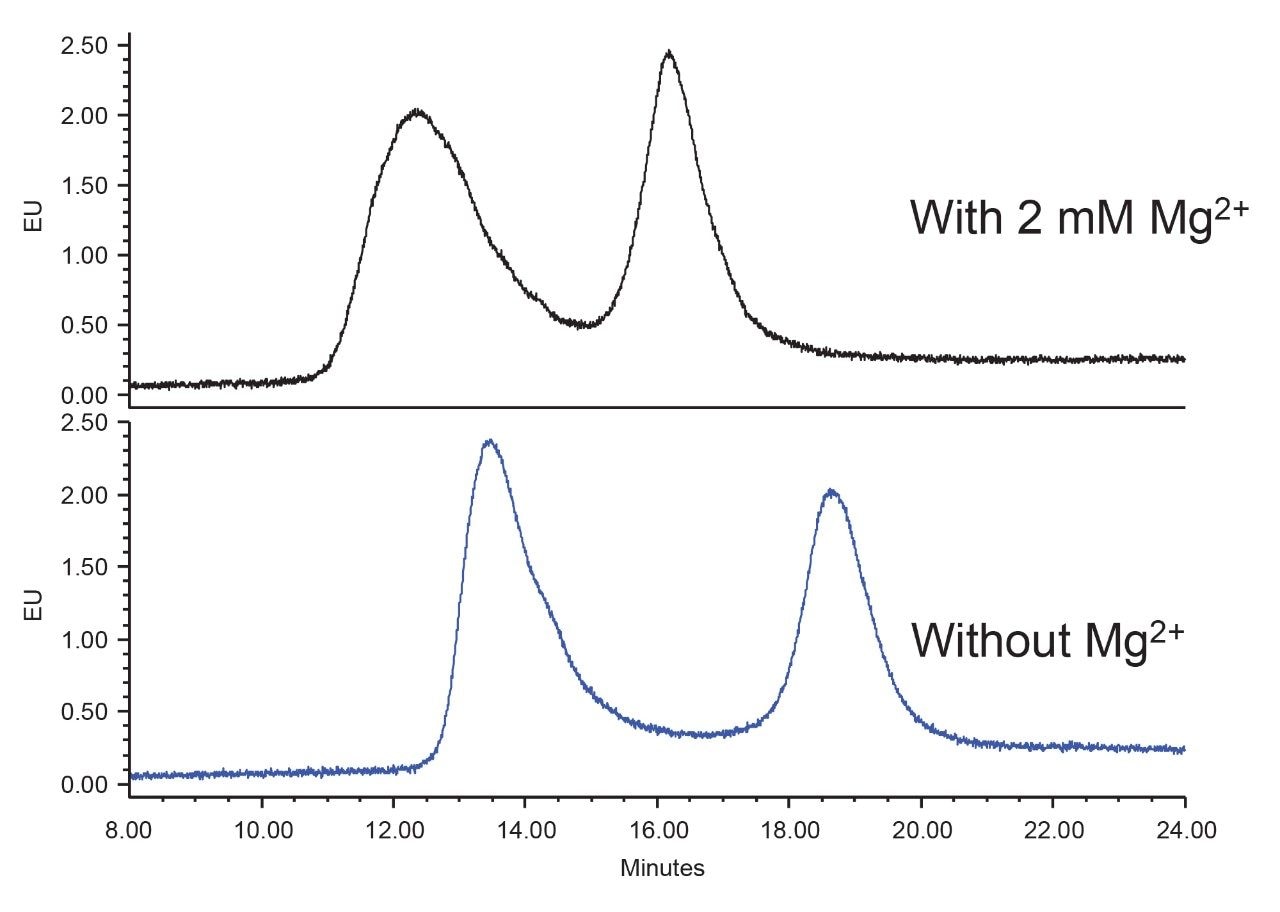
The addition of 2 mM magnesium (Mg2+) in the AEX mobile phase has been shown to improve the peak shape for AAV species.10 Under the conditions evaluated in this application note, the separation was not improved with the addition of Mg2+ (Figure 4) in the mobile phases for AAV8-GFP. However, the effectiveness of this low level of magnesium ion might still be worth considering for the separation of other AAV serotypes.
The separation of the empty capsids from the full capsids with the optimized method is shown in Figure 5, and a series of chromatograms for AAV8 samples with varying ratios between empty and full capsids is presented in Figure 6.
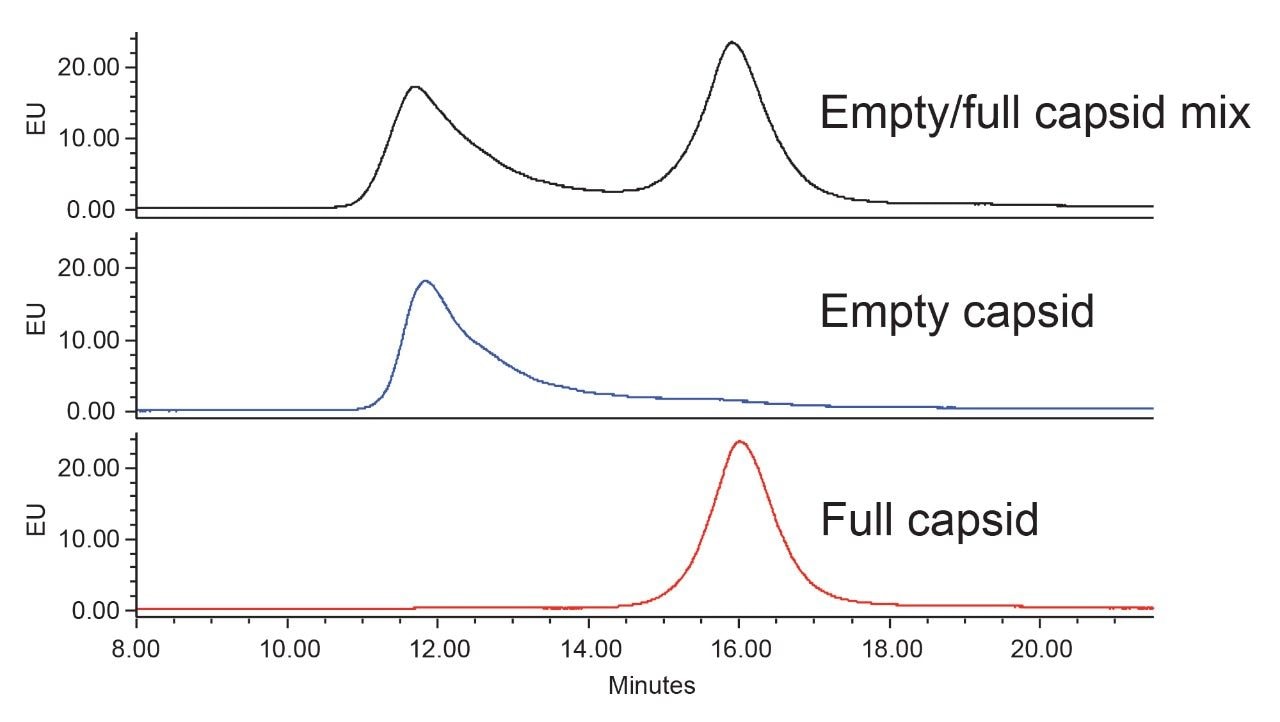
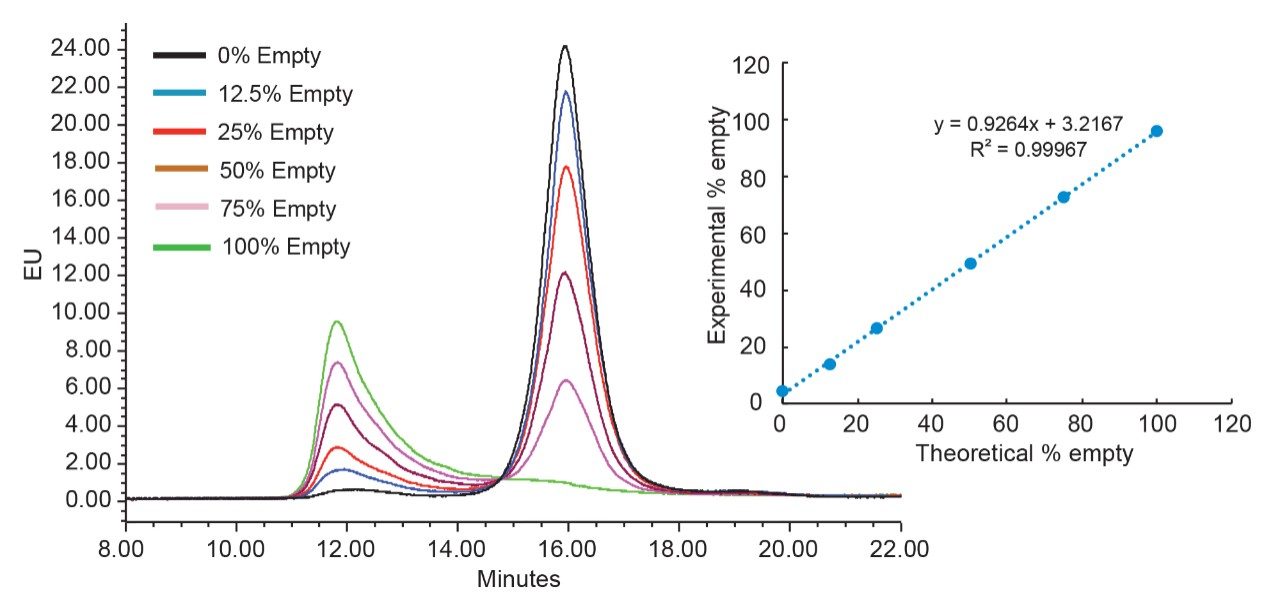
For the data shown in Figure 6, varying volumes of two standard samples comprised of predominately either empty or full capsids were mixed together to produce a series of samples for evaluation. To produce these mixtures with appropriate concentrations and ratios, a size exclusionchromatography (SEC) with intrinsic protein fluorescence (FLR) detection was employed to normalize the capsid concentrations of the empty and full AAV samples (data not shown). This concentration normalization was achieved based on the peak areas measured for both the empty and full AAV standard samples, which were assumed to be functionally pure, and in acceptance of a FLR response factor between full and empty capsid of 1.3.10 This response factor was previously reported for an AAV6 variant and was taken as a reasonable estimate for AAV8 capsid considering capsid protein homology. While this approximation was deemed adequate for the demonstration purposes of this study, it is important to point out that the assignment of empty and full AAV standard sample purities and content must be dutifully considered during method development.
Overall, Figure 6 demonstrates that this optimized AEX separation method can be used to monitor the ratios between empty and full capsids in an AAV8 sample. Also shown in Figure 6 is the linearity of the response. To produce this response curve, the peak areas for the empty and full standard were measured, after excluding the small peak area apexing around 16 min (green trace) and 12 min (black trace), respectively. A relative FLR response factor (RFF/E) of 1.9 was determined using the ratio of the full versus empty measured peak areas. This value was then divided into the area of the full capsid peak to normalize its response relative to the response of the empty capsid peak and then the relative abundances of empty and full capsids is calculated using Equation 1.
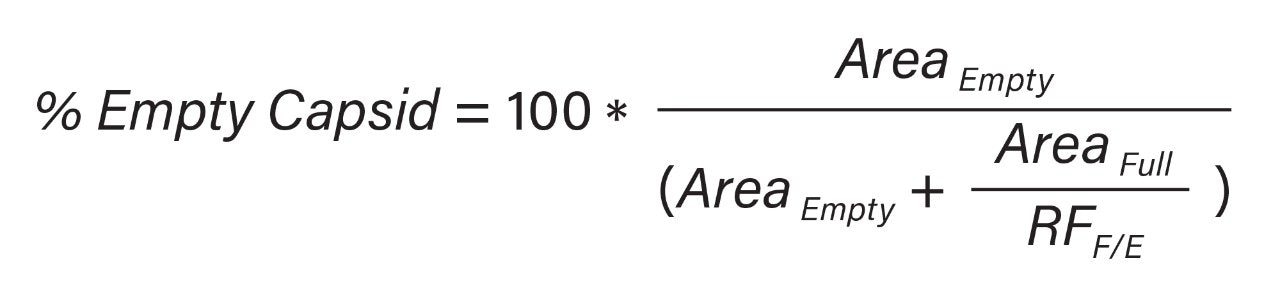
The differences between determined AEX response factor (1.9) and that observed by SEC-FLR (1.3) may be caused by the different mobile phases and column temperature used in respective experiments. This further highlights the importance of determining specific FLR response factors for empty and full capsids when establishing relative abundances of these two forms in an AAV sample.
Samples of several AAV serotypes were run individually on a Protein-Pak Hi Res Q Column under identical separation conditions (Figure 7). For comparison purposes, the chromatogram of AAV8 samples with empty and full capsids was also included in Figure 7a (bottom chromatogram). The separation for AAV8 was achieved after the method development efforts above. As can be seen, the retention time of the AAV capsid varies along with the AAV serotypes. While AAV2, AAV5, and AAV8 eluted approximately at the same time, AAV1 and AAV6 eluted later, and AAV9 eluted earlier (Figure 7a). AAV9 was retained better on the column when the salt concentration of the starting gradient was reduced (Figure 7b).
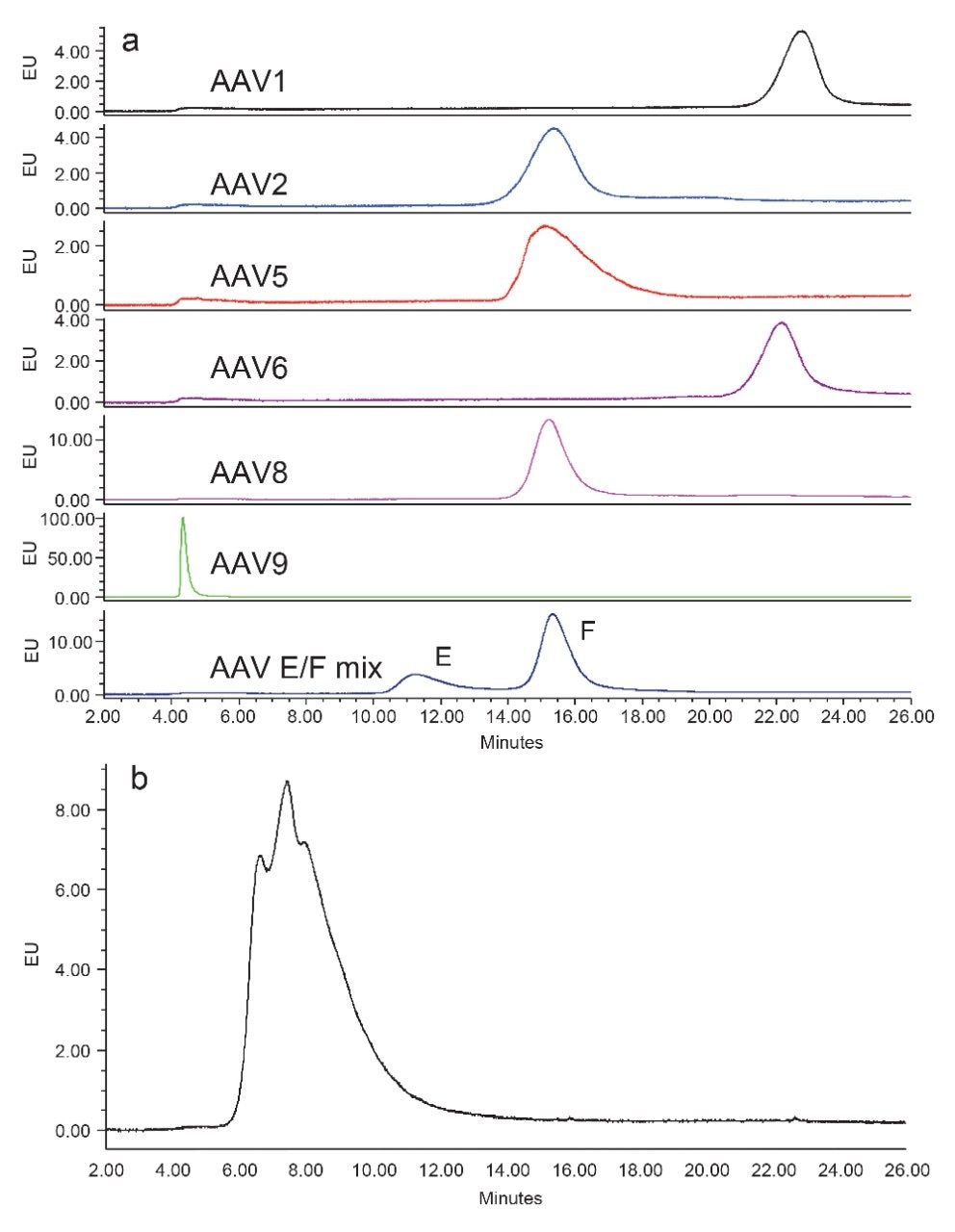
These results showed that further method development may be needed for the separation of the empty and full capsids for different AAV serotypes. This was not evaluated in the current study as the empty capsids of these serotypes were not available. However, because the peak shapes of these serotypes were largely similar to that observed for AAV8, the current optimized AEX method should serve as a useful starting point for other AAV serotypes in AEX method development.
Evaluating empty and full capsid content is important, because it can help to ensure the product quality as well as to help determine the appropriate dosage of the drug.
Here, we demonstrated that with proper method development, AAV8 empty and full capsids can be separated and their relative abundances determined using a Waters Protein-Pak Hi Res Q Column.
Due to differences in capsid protein surfaces, method evaluation, and perhaps some method optimization may be needed when separating empty and full capsids of other AAV serotypes, and the AEX method in this application note can serve as a useful starting point.
720006825, April 2020