This application note highlights the characteristics of a new mixed-mode RP/AX stationary phase that offers very different selectivity compared to traditional RP stationary phases.
While there is a great diversity of reversed-phase (RP) columns currently available, there are relatively few that are designed to retain and separate polar acidic analytes. Widely used silica-based RP packing materials typically exhibit both reversed-phase and cation-exchange behavior, with the latter arising from ionized silanols.1 The presence of ionized silanols creates a negative surface charge, which repels negatively charged analytes such as ionized acids. One strategy to retain ionized acids is to use ion-pairing reagents in the mobile phase.2 This approach has several disadvantages including long equilibration times and high background levels. Another strategy involves derivatizing the analytes to form more hydrophobic neutral species.3 The disadvantage of this approach is the greater time needed for sample preparation and the potential for increased variability in quantitative measurements. The limitations of these strategies led to the development of mixed-mode RP/anion-exchange (AX) columns, which contain stationary phases having both hydrophobic and AX functionalities.4 However, many mixed-mode RP/AX columns suffer from poor batch-to-batch reproducibility and a limited pH range. To overcome these issues, we have developed a new stationary phase based on hybrid organic/inorganic particles that has excellent batch-to-batch reproducibility and is stable over a wide pH range. The chromatographic selectivity of the new Atlantis BEH C18 AX stationary phase is described in this note. This material is packed into new MaxPeak High Performance Surfaces column hardware, which is designed to increase analyte recovery by minimizing interactions with stainless steel that can lead to losses. The packed columns are named Atlantis Premier BEH C18 AX Columns.
The sample contained 5 μg/mL thiourea, 12.5 μg/mL 5-fluorouracil, 25 μg/mL nicotinamide, 37.5 μg/mL adenosine- 5'-monophosphate, 37.5 μg/mL procainamide and 125 μg/mL resorcinol in 10 mM ammonium formate pH 3.00 (aq). Four consecutive separations were carried out, and the retention times from the last three injections were used to calculate the retention factors. Void time (t0) measurements were made using thiourea with an acetonitrile mobile phase.
|
Instrument: |
ACQUITY UPLC I-Class System with PDA Detector |
|
Data management: |
Empower 3 CDS |
|
Column size: |
2.1 × 50 mm |
|
Column temp.: |
30 °C |
|
Injection volume: |
1.5 μL |
|
Flow rate: |
0.2 mL/min |
|
Mobile phase: |
10 mM ammonium formate pH 3.00 (aq) |
|
UV detection: |
254 nm |
Samples containing 5 μg/mL thiourea, 50 μg/mL resorcinol, 10 μg/mL caffeine, 5 μg/mL 2-nitrobenzoic acid, 100 μg/mL metoprolol, 10 μg/mL 2-nitrobenzyl alcohol, 2 μg/mL papaverine, 125 μg/mL 2-chlorobenzoic acid, 20 μg/mL propranolol, 25 μg/mL 4-nitrophenol, 10 μg/mL berberine, 25 μg/mL 3-nitrophenol, 100 μg/mL 2-chlorophenol, 25 μg/mL 2-nitrophenol, 10 μg/mL amitriptyline, 500 μg/mL fluoxetine, 25 μg/mL diethylphthalate, 100 μg/mL fenoprofen, and 25 μg/mL dipropylphthalate were prepared in 15.4 mM ammonium formate pH 3.00 (aq). Three consecutive separations were carried out and the average retention times were used to calculate the gradient retention factors. Thiourea was used as the void time (t0) marker with a 100% acetonitrile mobile phase.
|
Instrument: |
ACQUITY UPLC System with PDA Detector |
|
Data management: |
Empower 3 CDS |
|
Column size: |
2.1 × 50 mm |
|
Column temp.: |
30 °C |
|
Injection volume: |
5 μL |
|
Flow rate: |
0.5 mL/min |
|
Mobile phase A: |
15.4 mM ammonium formate pH 3.00 (aq) |
|
Mobile phase B: |
Acetonitrile |
|
Gradient: |
5–95% B in 5.3 min (linear) |
|
UV detection: |
254 nm |
The sample contained 10 μg/mL thiourea and 12 μg/mL thymine in 10 mM ammonium formate pH 3.00 (aq). The instrument and separation conditions were the same as for the polar mixture separation. After separating the polar mixture four times, the flow was stopped for 10 min. The flow was then restarted at 0.2 mL/min. After 0.6 min, two injections of the thiourea/thymine sample were made. The retention factor of thymine was calculated using the average retention time of thymine and the t0 measurement previously obtained using thiourea with an acetonitrile mobile phase.
The sample contained 10 μg/mL thiourea, 100 μg/mL guanosine-5'-monophosphate, 100 μg/mL resorcinol, and 100 μg/mL nicotinamide in water.
|
Instrument: |
ACQUITY UPLC H-Class System with PDA Detector |
|
Data management: |
Empower 3 CDS |
|
Column size: |
2.1 × 50 mm |
|
Column temp.: |
30 °C |
|
Injection volume: |
2.0 μL |
|
Flow rate: |
0.2 mL/min |
|
Mobile phase: |
(95/5 v/v) 10 mM ammonium bicarbonate pH 10.08 (aq)/methanol |
|
UV detection: |
254 nm |
The sample contained 200 μg/mL metoprolol tartrate, 10 μg/mL papaverine, and 50 μg/mL amitriptyline in methanol/water (1/5 v/v). Three consecutive separations were carried out and the average peak widths at 13.4% peak height were determined for each analyte.
|
Instrument: |
ACQUITY UPLC System with TUV Detector |
|
Data management: |
Empower 3 CDS |
|
Column size: |
2.1 × 50 mm |
|
Column temp.: |
30 °C |
|
Injection volume: |
2 μL |
|
Flow rate: |
0.4 mL/min |
|
Mobile phase A: |
0.1% formic acid (aq) |
|
Mobile phase B: |
Acetonitrile |
|
Gradient: |
15–95% B in 7.37 min (linear) |
|
UV detection: |
260 nm |
The properties of the Atlantis BEH C18 AX stationary phase are summarized in Table 1, and compared to those of BEH C18, CSH C18, and HSS T3. The Atlantis BEH C18 AX stationary phase is based on a 95 Å ethylene-bridged hybrid (BEH) particle, which has a smaller pore size than the 130 Å particles used in BEH and CSH stationary phases. The smaller pore size particles have a 50% higher surface area, which leads to greater retention. The retention factors of neutrals for Atlantis Premier BEH C18 AX columns are very similar to those for HSS T3 columns. The Atlantis BEH C18 AX stationary phase contains not only C18 groups, but also tertiary alkylamine moieties, creating a positive surface charge below approximately pH 8. Like Atlantis BEH C18 AX, CSH C18 also has both C18 and anion-exchange groups. However, with anion-exchange groups containing pyridine moieties, CSH C18 is positively charged only below approximately pH 5. Atlantis BEH C18 AX is endcapped to reduce the concentration of residual silanols and to improve the base stability of the stationary phase.
To obtain sufficient retention of highly polar analytes in RP separations, highly aqueous mobile phases are often needed. Not all RP columns are compatible with these mobile phases, due to retention losses that occur when the flow is stopped and restarted.5,6 High-coverage C18 and C8 bonded phases based on particles with average pore diameters less than about 100 Å are particularly prone to this phenomenon, with retention losses approaching 100%. The use of an intermediate C18 surface concentration (1.6 μmol/m2) together with the AX groups makes Atlantis BEH C18 AX compatible with highly aqueous mobile phases. An experiment in which the flow of a 100% aqueous mobile phase (at 30 °C) was stopped and restarted for an Atlantis Premier BEH C18 AX, 1.7 μm, 2.1 × 50 mm Column found only a 3.7% decrease in retention for thymine. This is lower than the approximately 10% retention loss observed for HSS T3.
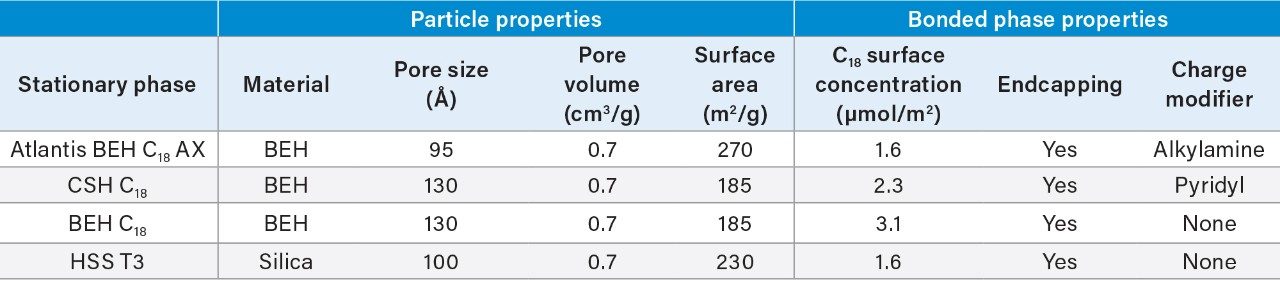
The Atlantis BEH C18 AX stationary phase has a positive surface charge below approximately pH 8. Relative to conventional reversed-phase materials, the positive surface charge gives increased retention of anions, such as ionized acids, and decreased retention of cations, such as protonated bases. These differences are readily observed in the chromatograms shown in Figure 1. The sample used for this study contains a mixture of six polar analytes, one which is negatively charged in the pH 3 mobile phase (adenosine 5'-monophosphate, AMP), two which are positively charged (nicotinamide and procainamide) and three which are neutral (thiourea, 5-fluorouracil, and resorcinol). Relative to HSS T3 and CSH C18 columns, the Atlantis Premier BEH C18 AX Column gives the highest retention for AMP. The neutral compounds have similar retention on the Atlantis Premier BEH C18 AX and HSS T3 columns, while the CSH C18 column has the lowest retention of these analytes, due to the lower surface area of the 130 Å particles used for this material. The positively-charged analytes nicotinamide and procainamide are most retained on the HSS T3 column, and least retained on the Atlantis Premier BEH C18 AX Column.
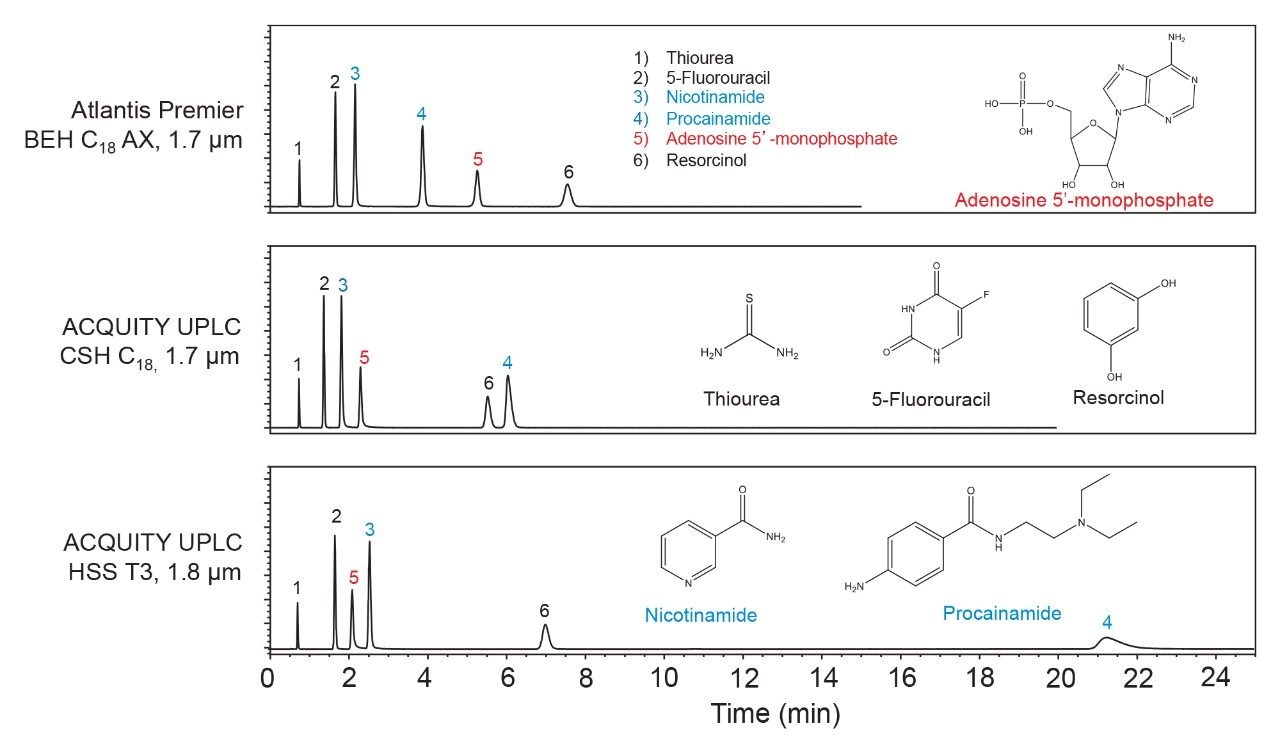
These trends have also been observed in studies with larger sets of analytes. For example, a set of 18 compounds covering a range of polarities and acid/base properties was separated using an acetonitrile gradient with an aqueous mobile phase containing a pH 3 ammonium formate buffer. The results are shown in Figure 2 as plots of gradient retention factors (kg) for Atlantis BEH C18 AX vs. kg for BEH C18 and CSH C18. The data points for the analytes that are negatively charged at pH 3 are shown as red circles, those for the positively charged analytes as blue diamonds and those for the neutrals as black triangles. The Atlantis Premier BEH C18 AX Column gives the highest kg values for the negatively charged compounds, and the lowest kg values for the positively charged analytes. To quantify the selectivity differences between these columns, we calculated the s2 values as
s2 = 1 - r2
where r is the correlation coefficient for the kg vs kg plots.7 The lower the correlation coefficient, the greater is the s2 value, indicating a larger selectivity difference between the columns. The s2 values range between 0 and 1. The results show s2 values of 0.58 for Atlantis BEH C18 AX relative to BEH C18 and 0.47 for Atlantis BEH C18 AX relative to CSH C18. These are large values, approximately four times greater than the largest value reported for a set of different RP bonded phases.7 Atlantis BEH C18 AX significantly expands the stationary phase selectivity range for reversed-phase columns when separating mixtures containing ionized analytes.
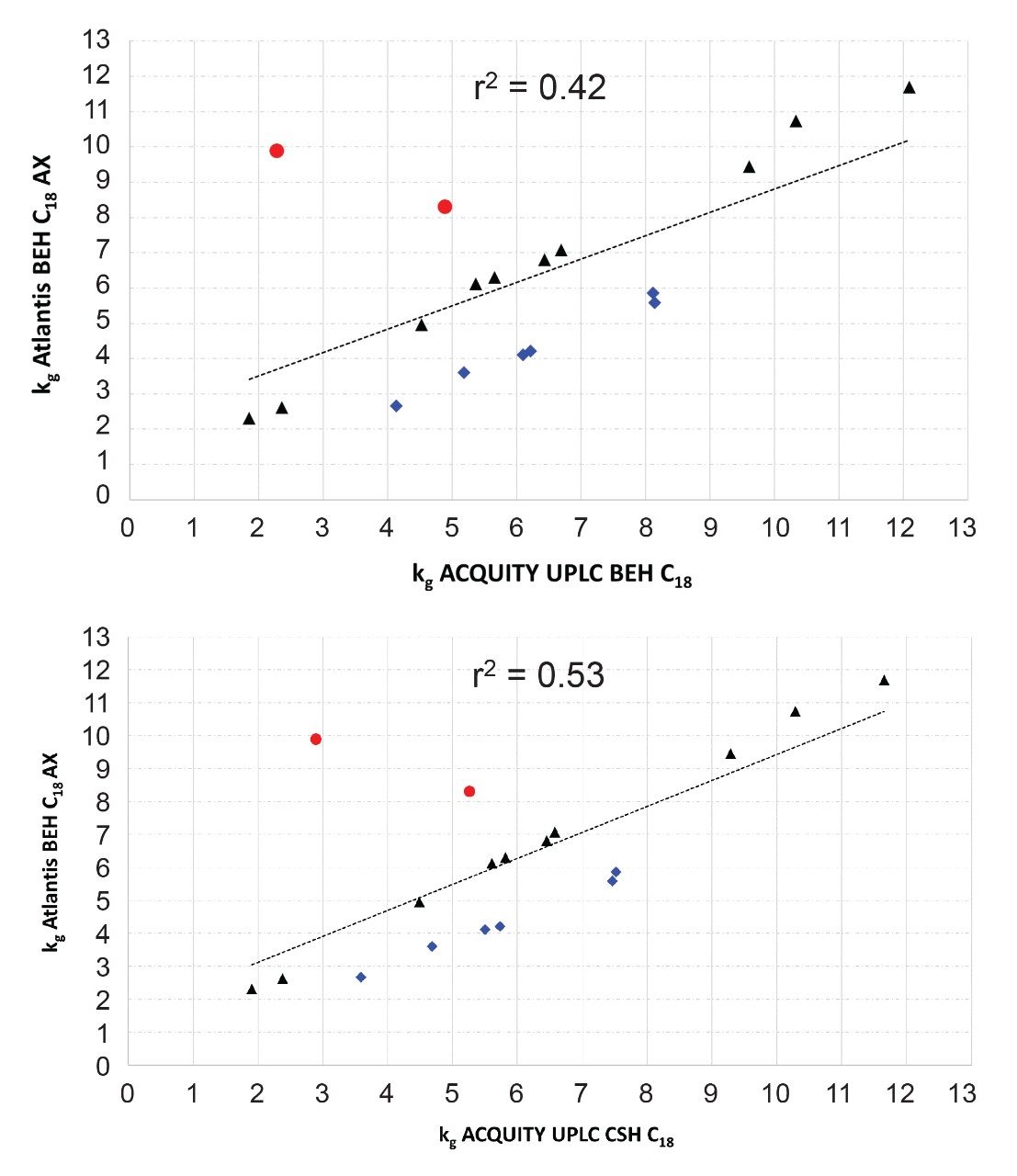
As for CSH C18,8 the surface modification of Atlantis BEH C18 AX involves separate steps to incorporate the charge modifier, the C18 groups and the endcap. This approach gives excellent control of the surface chemistry, resulting in high batch-to-batch reproducibility. The chromatographic reproducibility of Atlantis BEH C18 AX was assessed by testing twenty-seven different batches of 1.7, 2.5, and 5 μm materials using the separation of the polar mixture shown in Figure 1. For the relative retentions, the results showed relative standard deviations (RSD) ranging from 1 to 3% (Figure 3), indicating excellent reproducibility, comparable to some of the most reproducible conventional C18 bonded phases.9 No statistically significant differences were found in the relative retentions of the different particle sizes, facilitating scalability across columns packed with different particle sizes.
Combining the base-stable BEH particle10 with stable bonding chemistries, the Atlantis BEH C18 AX material shows exceptional resistance to both acidic and basic mobile phases for a mixed-mode stationary phase. The recommended pH range for Atlantis Premier BEH C18 AX Columns is compared to those of two leading mixed-mode RP/AX columns in Table 2.
The most notable differences are in the upper limits, with Atlantis BEH C18 AX recommended for use up to pH 10 vs. pH 4.5 or 7.5 for the other columns, which are based on silica particles. The pH range for Atlantis BEH C18 AX was established based on the results of accelerated tests similar to those which have previously been described.10,11 To verify the upper pH limit, we carried out a stability study using a pH 10 mobile phase and a temperature of 30 °C. A three-component sample mixture was injected 340 consecutive times, with a run time of one hour per injection.
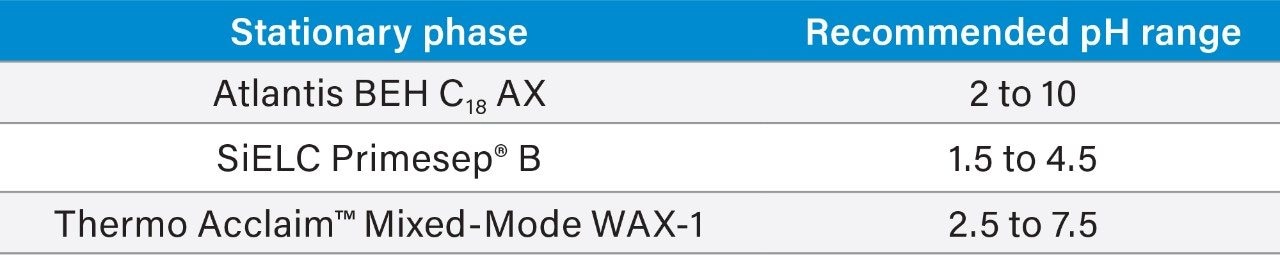
The changes in retention time for resorcinol, guanosine-5'-monophosphate, and nicotinamide were found to be +0.1%, -14.5%, and -8.5%, respectively (Figure 4). The extended high pH stability of Atlantis Premier BEH C18 AX Columns allows a wider range of mobile phase pH values to be used. For samples containing ionizable analytes, mobile phase pH has been shown to be the most powerful variable to adjust selectivity.12
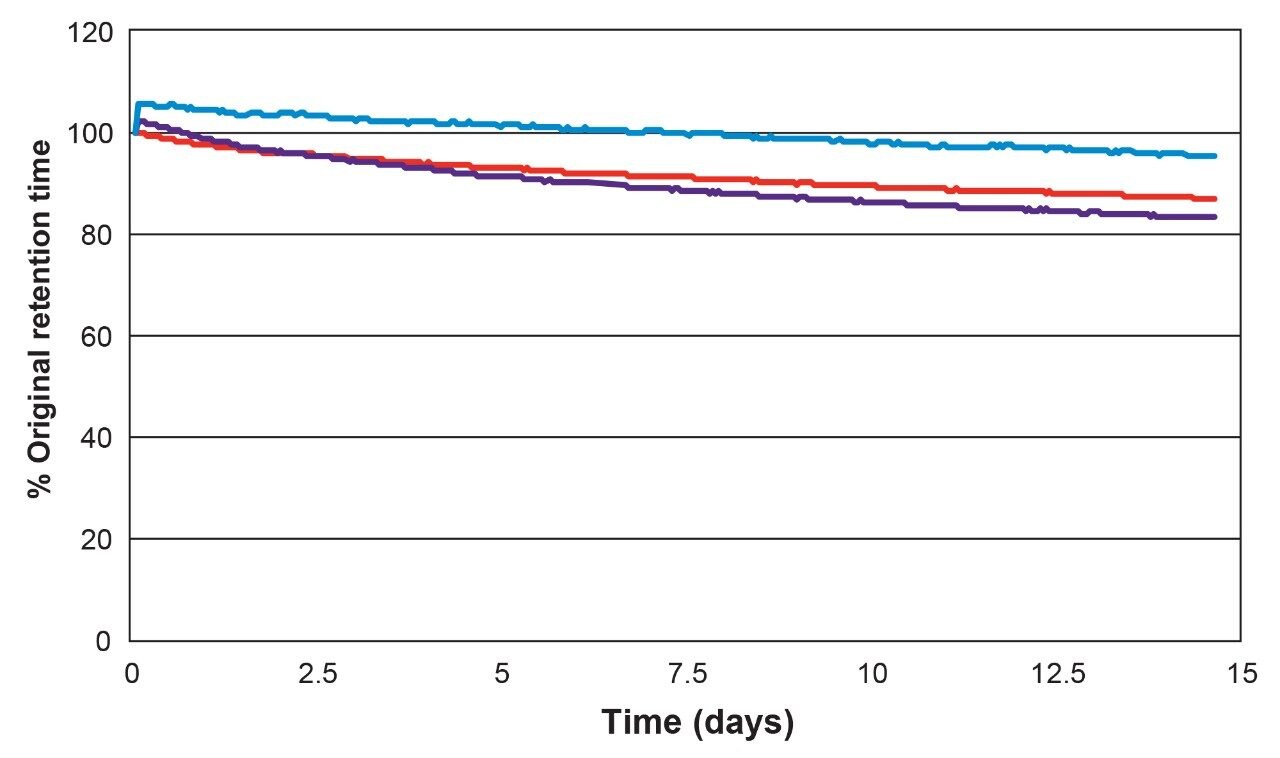
To investigate the dependence of retention on pH for an Atlantis Premier BEH C18 AX Column, the mixture of Figure 1 was separated using a series of mobile phases varying in pH, but with constant buffer concentration. The 100% aqueous mobile phases were prepared from ammonium formate with variable amounts of formic acid or ammonium hydroxide added to adjust the pH. The results are shown in Figure 5. The retention factors of three of the analytes (AMP, nicotinamide, and procainamide) show a pronounced dependence on pH, while the retention factors of the other two analytes (5-fluorouracil and resorcinol) vary only slightly.
The behavior of nicotinamide (pKa = 3.4) and procainamide (pKa = 4.6, 9.3) is similar to that of other bases in RP chromatography: the retention factors are lower below their pKa, where they are protonated, then increase above their pKa, where they are unprotonated.13 The differences in retention factors between the protonated and unprotonated forms are large: a factor of 9 for nicotinamide and more than 14 for procainamide.
The surface charge of the stationary phase also transitions from positive to neutral over this pH range, which may accentuate the retention factor difference since the protonated forms of the analytes are partially excluded from the positively-charged surface due to ionic repulsion. The retention factor of AMP shows a different dependence on pH. AMP has three ionizable groups: the two acidic P-OH groups (pKa = 0.9 and 6.2) and the basic adenine moiety (pKa = 3.8).
The ionic charge of AMP varies from 0 at approximately pH 2.5 to -2 at pH 7. The increasing negative charge of AMP accounts for its increasing retention factor between pH 2.5 and 5.5. The decrease in its retention factor above pH 5.5 is due to the decreasing positive charge on the stationary phase.
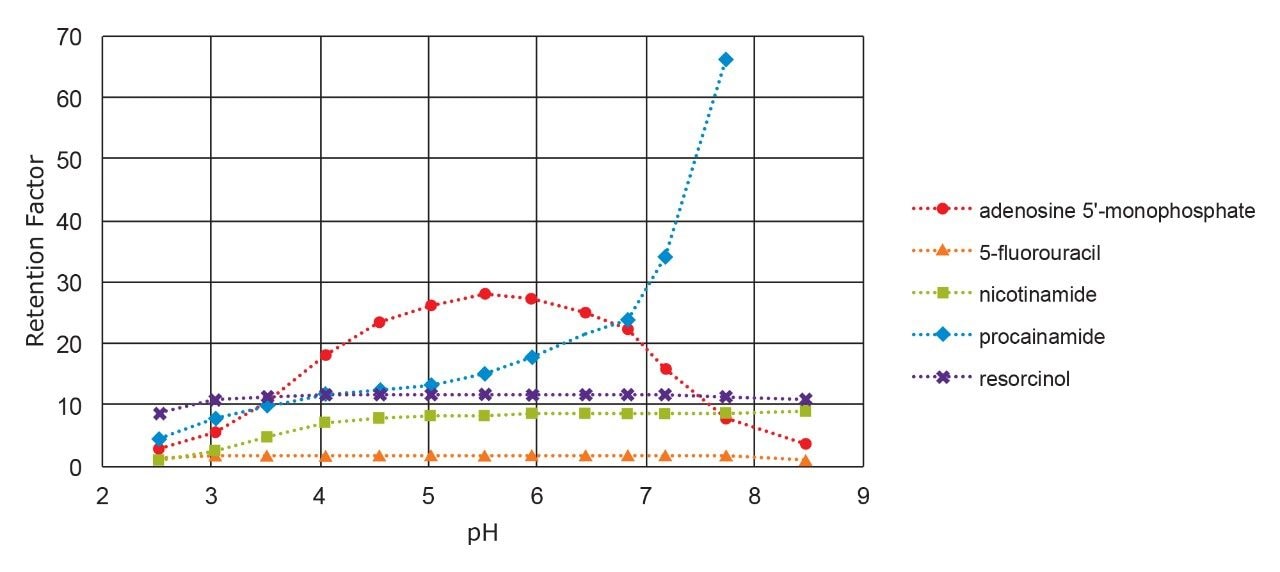
A second important mobile phase variable is the concentration of the buffer, which strongly affects the ion-exchange retention. To investigate this variable, the same polar mixture was separated using a series of mobile phases containing pH 3.00 ammonium formate at concentrations ranging from 5 to 50 mM. The results are shown in Figure 6. Two of the analytes (AMP and procainamide) show a strong dependence of retention on buffer concentration, with opposite trends. The retention factor of negatively charged AMP decreases with increasing buffer concentration, due to the formate anions displacing the AMP from the AX sites on the stationary phase. In contrast, the retention factor of positively charged procainamide increases with increasing buffer concentration. This is attributed to a weakening of the ionic repulsion between protonated procainamide and the positively-charged surface as the buffer concentration increases.
It has been demonstrated that CSH C18 columns give a large improvement in the sharpness and symmetry of peaks from cationic analytes, such as protonated bases, when using low ionic-strength mobile phases, like those containing formic acid.8 Atlantis Premier BEH C18 AX Columns also show this effect, but to an even greater extent. Shown in Figure 7 are chromatograms for the separation of three basic analytes on three different columns using an acetonitrile gradient and an aqueous mobile phase containing 0.1% formic acid. The results show that the metoprolol and amitriptyline peaks become narrower and more symmetric moving from BEH C18 to CSH C18 to Atlantis BEH C18 AX.
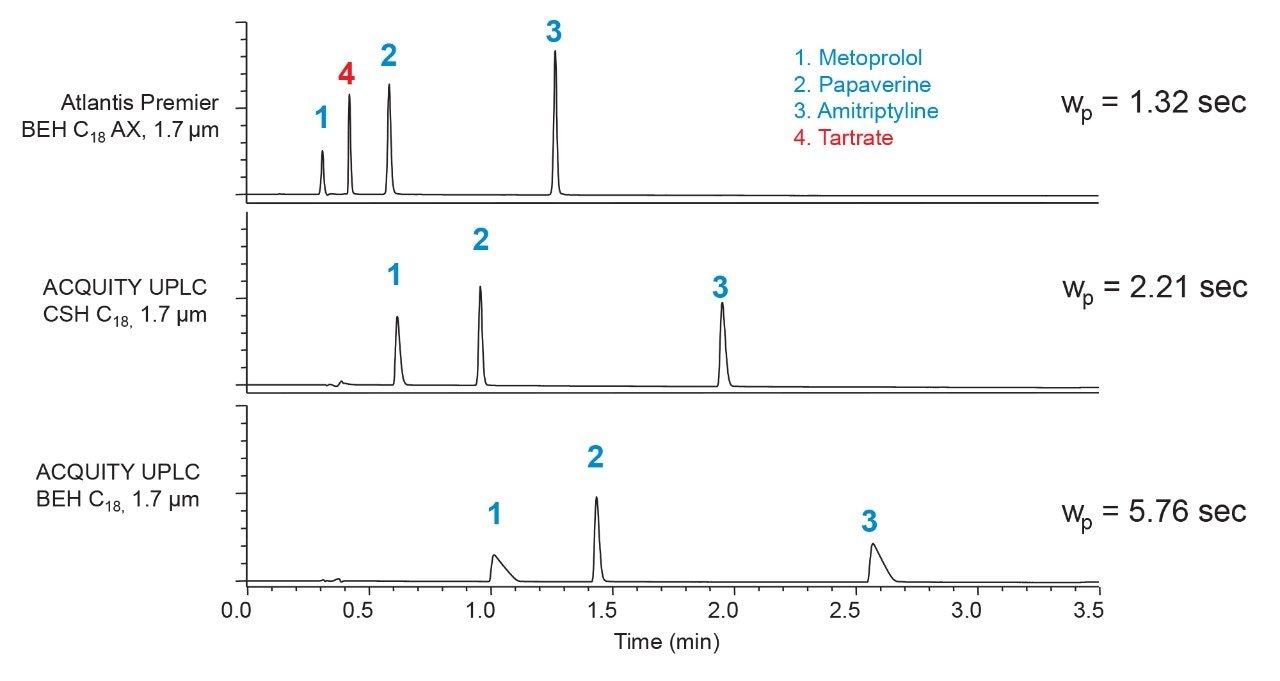
Figure 7. Gradient separations of three basic analytes using a mobile phase containing 0.1% formic acid. The average peak widths at 13.4% peak height (wp) were calculated from the metoprolol and amitriptyline peaks.
The retention times also decrease, as a result of ionic repulsion of the protonated analytes from the surfaces of the CSH C18 and Atlantis BEH C18 AX stationary phases. It’s notable that the tartrate counterion of metoprolol is retained on the Atlantis Premier BEH C18 AX Column, but not on the other two columns. To quantify the improved peak sharpness for the Atlantis Premier BEH C18 AX Column, we determined the average peak widths for the metoprolol and amitriptyline peaks. The results show a 40% decrease moving from CSH C18 to Atlantis BEH C18 AX. This is a large improvement that offers the possibility of higher resolution separations for basic analytes when using low ionic-strength mobile phases. However, the peak shapes for ionized acids may not be optimal when using such mobile phases. Buffer concentrations of at least 10 mM are recommended for ionized acidic analytes with Atlantis Premier BEH C18 AX Columns.
This application note highlights the characteristics of a new mixed-mode RP/AX stationary phase that offers very different selectivity compared to traditional RP stationary phases. Atlantis BEH C18 AX has excellent batch-to- batch reproducibility and exceptional high-pH stability relative to existing mixed-mode RP/AX products. Compared to conventional RP stationary phases, when used with mobile phase pH values less than about 8, Atlantis BEH C18 AX shows increased retention of anions, such as ionized acids, and decreased retention of cations, such as protonated bases. The selectivity for ionizable analytes may be varied over a wide range by adjusting the pH and buffer concentration of the mobile phase. When using low ionic strength mobile phases (e.g., containing 0.1% formic acid), Atlantis BEH C18 AX shows extraordinarily sharp and symmetric peaks for protonated bases. This new stationary phase is complementary to existing RP stationary phases, making it useful for column screening during method development.
720006742, January 2020