This is an Application Brief and does not contain a detailed Experimental section.
This Application brief demonstrates improve separations of IgG2 disulfide isoforms while also enhancing MS sensitivity.
IgG2 antibodies exhibit unique heterogeneity from the formation of disulfide isoforms. Improved analytical capabilities for detecting these isoforms can reveal new insights on efficacy and biotransformation.
Monoclonal antibody (mAb) therapeutics are widely used in the treatment of different types of cancers, autoimmune diseases, and other disorders. Immunoglobulin G (IgG) is the main type of mAb, of which there are four subclasses: IgG1, 2, 3, and 4. The majority of therapeutic mAbs developed so far belong to the IgG1 subclass, although some are IgG2 or IgG4. These different IgG subclasses differ in terms of the connectivity of their disulfide bonds.
Whereas IgG1 molecules generally only have one defined structure, IgG2 antibodies can exist as three isoforms (A, B, and the intermediate species A/B), each with a different pattern of disulfide bonds.1 The nature of the disulfide bonding in the IgG2 subclass has been shown to influence antigen binding activity in vitro.2 The heterogeneity associated with different disulfide isoforms adds to the structural complexity of an IgG2, posing an additional challenge for the characterization of these molecules.
Reversed-phase liquid chromatography (LC) coupled to high-resolution mass spectrometry (MS) is a powerful analytical approach for the profiling of mAb therapeutics. The combination of high performance separations and accurate mass information can provide detailed insights into antibody heterogeneity and stability.3 However, the highest resolution chromatographic separations of intact proteins have relied upon ion pairing reagents, which can compromise MS sensitivity. To facilitate the characterization of IgG2 isoforms, methods combining high resolution intact protein separations with high MS sensitivity are still required.
Newer column technologies have promised gains in resolving power for the reversed-phase separations of proteins, specifically monoclonal antibodies. Among the leading options is the BioResolve RP mAb Polyphenyl Column, which is based on a superficially-porous silica particle and unique, high coverage phenyl bonding.4,5 Because of its chemical properties, it is found to be amenable to milder elution conditions, namely lower temperatures and less acidic conditions.
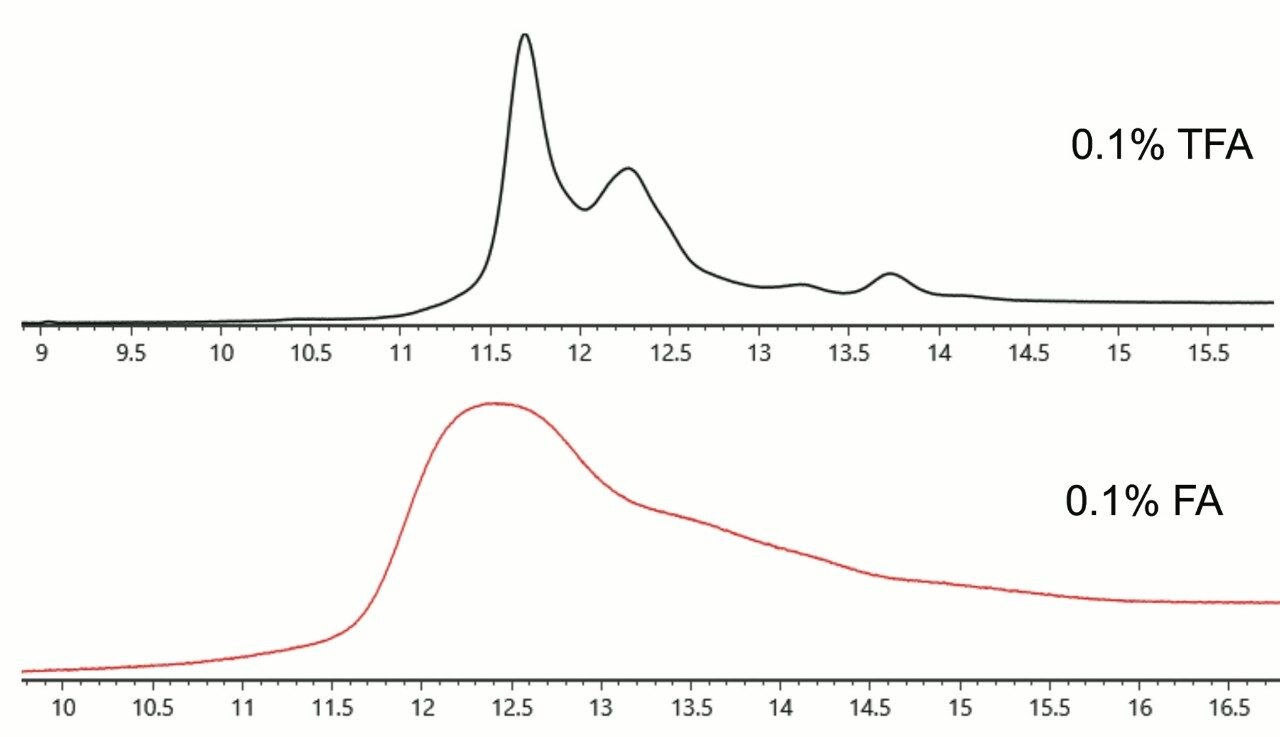
Previous methodologies for the profiling of IgG2 isoforms have often required the use of high column temperatures, raising concerns over the acid-catalyzed degradation of samples (particularly intermediate species) at column temperatures ≥80 °C. The use of trifluoroacetic acid (TFA) has been necessary to resolve IgG2 disulfide isoforms, but this compromises MS sensitivity as a result of ion suppression. Formic acid can be used to help improve ionization efficiency and reduce the acidity of the mobile phase, but it does so at the detriment of chromatographic resolving power. Few alternatives for more MS-friendly and more sample-friendly separations have existed with previous technologies. To this point, example separations of an IgG2 with a market-leading C4 column are displayed in Figure 1, as obtained using TFA and FA. The challenge to resolve disulfide isoforms is apparent in this example, wherein no resolution could be achieved with formic acid and partial resolution could only be observed if using TFA. To make use of this column technology, it was necessary to use TFA and also include saw-tooth, repeat gradients in the method to minimize carryover.
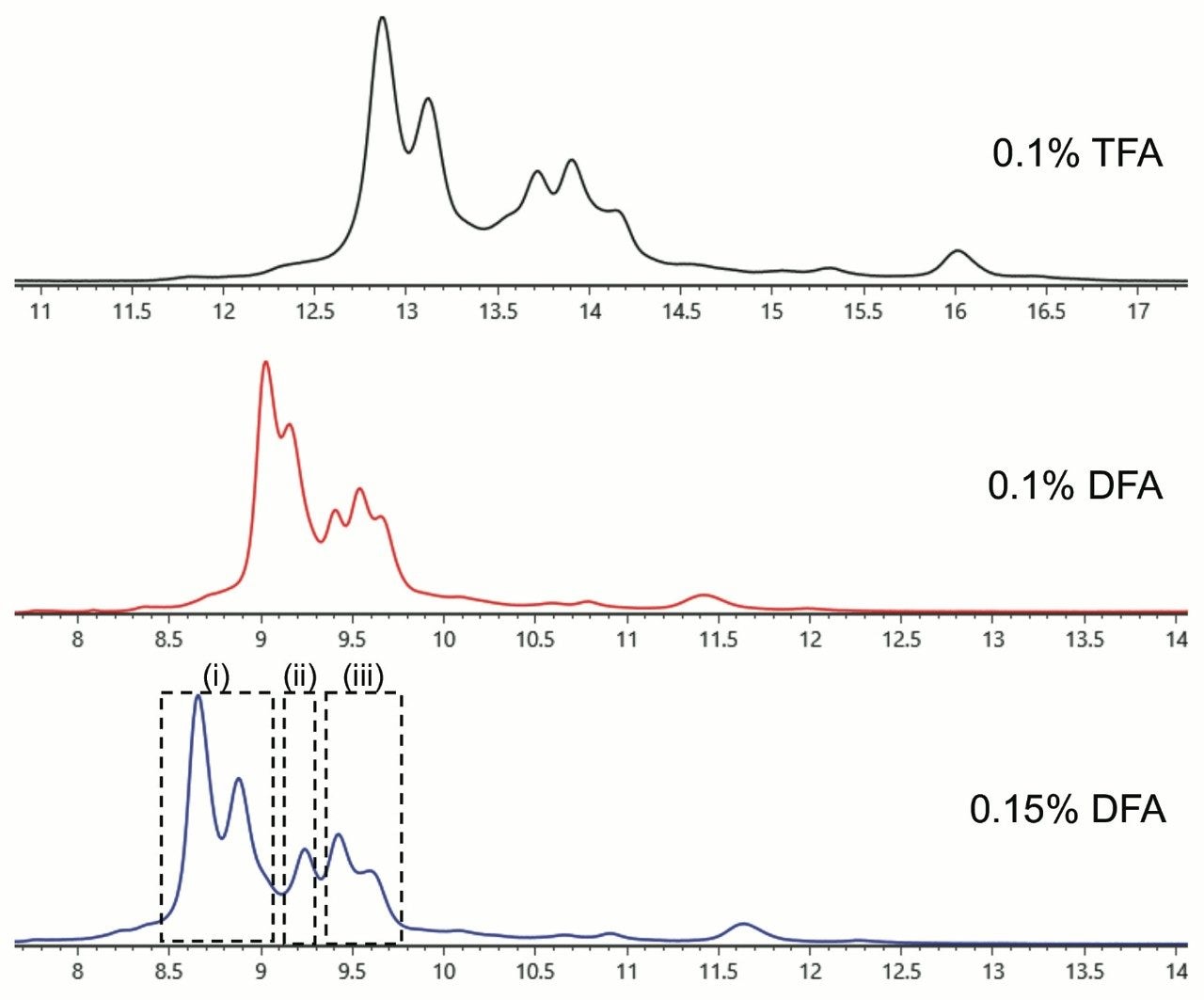
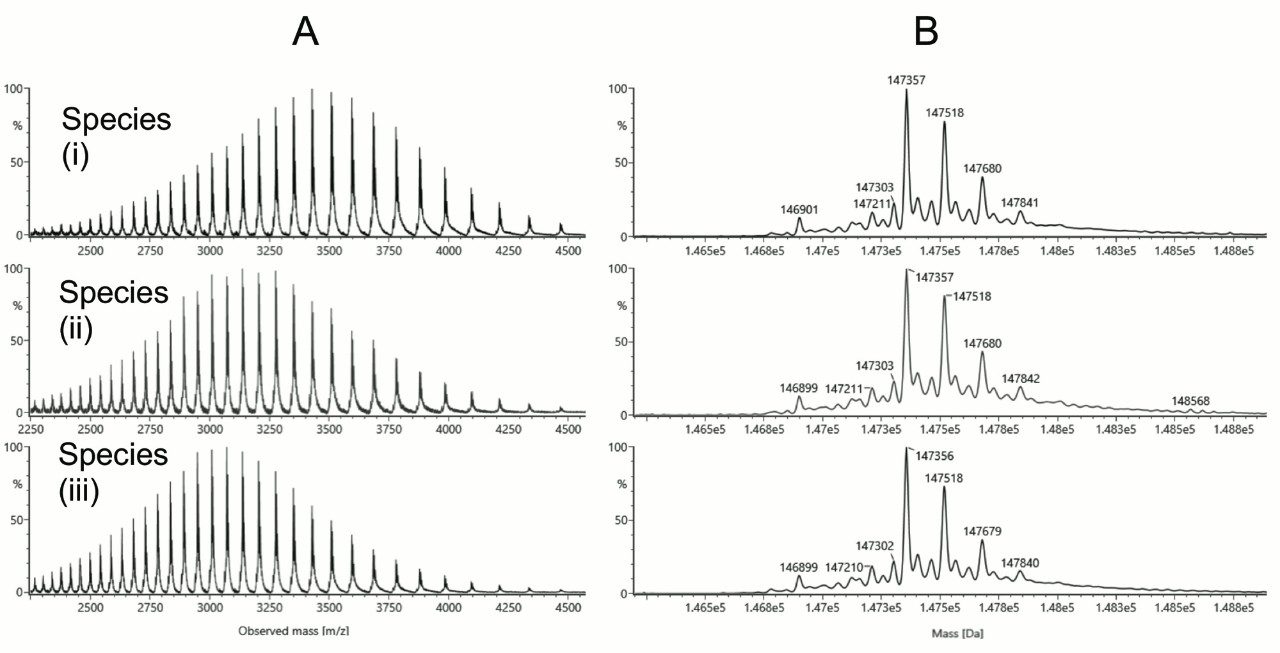
With a BioResolve RP mAb Polyphenyl 450 Å, 2.7 μm, 2.1 x 100 mm Column (p/n 186008945), it was possible to reduce the column temperature to 70 °C without detriment to recovery. Moreover, it was no longer necessary to perform saw-tooth gradients after an analysis, given that no carryover peaks were observed after changing the column chemistry. Excellent resolution of the expected IgG2 isoforms was observed. In fact, a number of additional species were detected. Combined with these method changes, we also explored the use of difluoroacetic acid (DFA), a novel ion pairing agent first proposed in 20026 for peptide mass spectrometry that has more recently garnered attention for its utility in protein LC-MS.7 With the availability of IonHance Difluoroacetic Acid (p/n 186009201), a first ever MS-grade DFA with <50 ppb K/Na content, it was possible to optimize the chromatography and achieve TFA-like resolving power (Figure 2) while also improving MS sensitivity and MS data quality (Figure 3). In sum, high quality mass spectra were achieved and there were no significant observations of salt adducts or gas phase ion pairing to challenge the interpretability of the data.
Each of the major chromatographic peaks exhibited the same glycoform profile and masses consistent with the intact protein without additional modifications. Thus, we could conclude that these peaks differ only in terms of their higher order structure. Using data from redox experiments (not shown), it has been possible to attribute peaks in the regions (i), (ii), and (iii) to disulfide isoforms B, A/B, and A, respectively. The structural basis for the presence of two distinct peaks in regions (i) and (iii) is not yet clear. Among the possibilities, these may correspond to different disulfide arrangements between the closely spaced cysteines in the hinge region.
Quite interestingly, the high signal to noise spectra also made it possible to clearly observe differences in the charge state distributions for each species. Under the denaturing conditions of reversed-phase LC-MS, isoform A (region iii) exhibits a higher mean charge than isoform B, consistent with the more open structure predicted for this disulfide pattern.2
It is critical that disulfides isoforms be well understood in IgG2 therapeutics as they can be tied to efficacy. Rearrangement is also known to occur upon dosing,8 suggesting that an assay to profile such might be essential to pharmacodynamic and biotransformation studies.
Given its purposefully designed attributes, the BioResolve RP mAb Polyphenyl Column makes for a robust starting point for modernizing LC-MS techniques for assaying IgG2 isoforms. Complemented with MS-grade IonHance Difluoroacetic Acid, impressive assay capabilities have been demonstrated herein. DFA confers advantageous ion pairing with less ion suppression than TFA. In turn, an LC-MS technique is achieved that has the capacity and sensitivity to help glean new information about IgG2 disulfide isomerization.
720006473, January 2019