
For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
A rapid UPLC-MS/MS method for the analysis of steroid hormones in dried blood spots is demonstrated using the Xevo TQ-S micro Mass Spectrometer.
Rapid separation and analysis of steroid hormones in dried blood spots was achieved using the ACQUITY UPLC I-Class System/Xevo TQ-S micro Mass Spectrometer and CORTECS Column.
Dried blood spots (DBS) are an established micro sampling technique providing a low-cost approach of collecting, shipping, and analyzing samples for clinical research. Ligand-binding assays (LBAs) are the established frontline testing methodologies for DBS samples in steroid hormone analysis. Although rapid, the relatively low analytical specificity of the LBAs may necessitate follow-up, using liquid chromatography – tandem mass spectrometry (LC-MS/MS). The multiplexed LC-MS/MS analysis of steroid hormones panels provides greater detail about the underlying enzyme activity, compared with single-analyte LBAs, which is important for the assessment of biomarkers in clinical research. The challenge is to create a LC-MS/MS methodology which separates key analytes from matrix and isobaric interferences, while maximizing throughput.
UltraPerformance Liquid Chromatography (UPLC) on the ACQUITY UPLC I-Class System enables high pressure chromatographic separations in short analysis times. Combining the ACQUITY UPLC with a CORTECS 2.7 µm particle Column provides UPLC separations at high linear velocities without a loss in column performance, particularly in regard to critical pair separations.
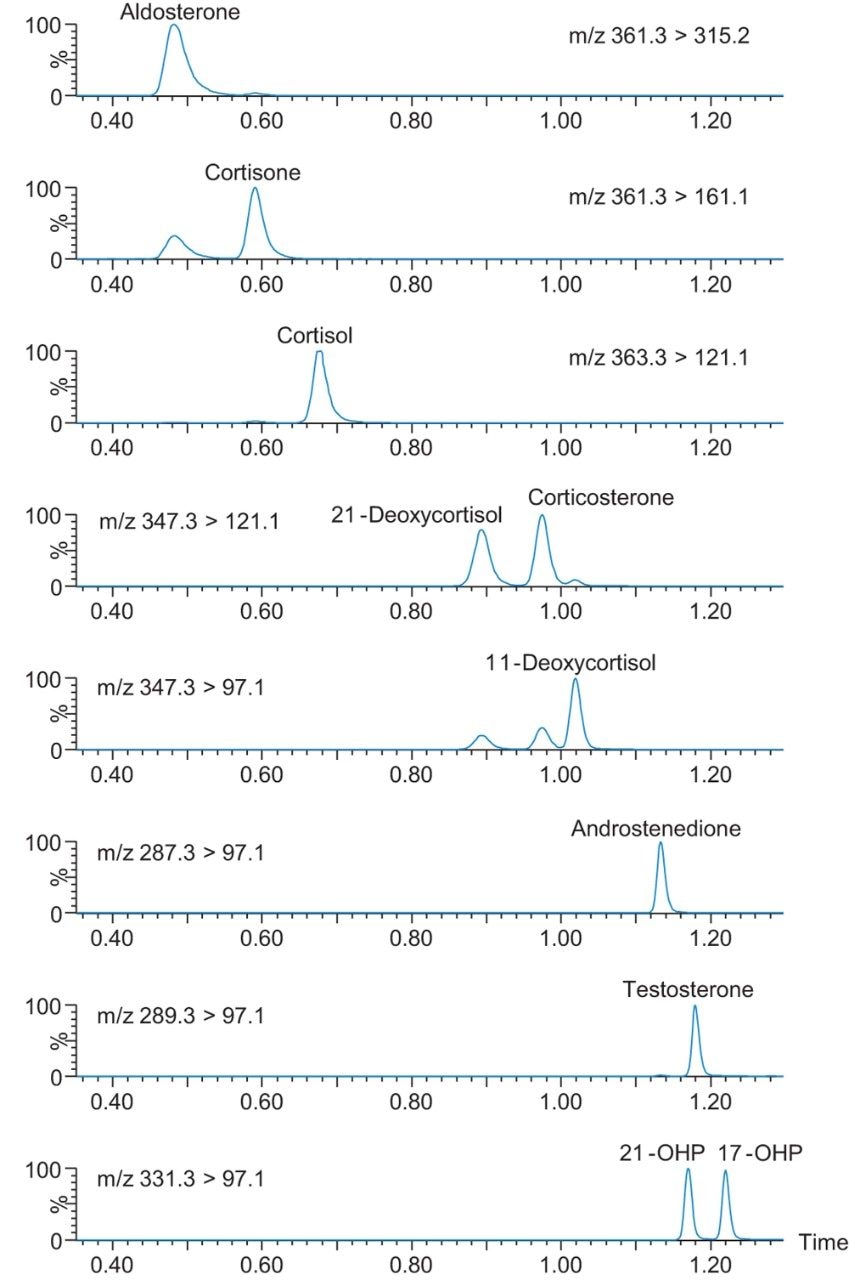
Through utilization of the ACQUITY UPLC I-Class System and the CORTECS Column technology, separation of cortisol, 21-deoxycortisol, 11-deoxycortisol, androstenedione, and 17-hydroxyprogesterone (17-OHP) from other endogenous steroid hormones was achieved within a 2.3 minute injection cycle time (Figure 1). Analytically sensitive detection between 0.5–1.0 ng/mL in DBS samples was achieved for the steroid hormones using the Xevo TQ-S micro, following extraction employing Oasis MAX µElution SPE on the Tecan Freedom Evo 100 Liquid Handling System (LHS).
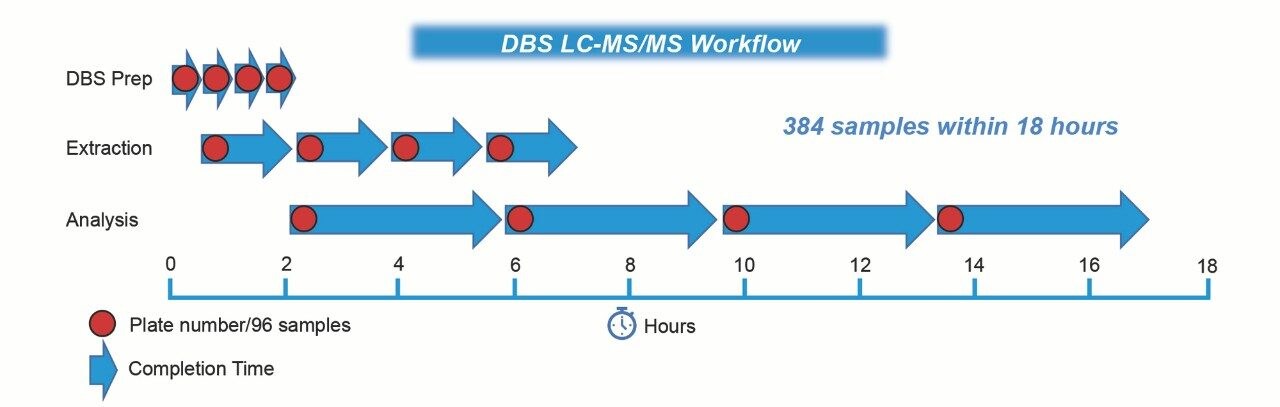
Using the LHS, 150 µL internal standard was added to two 3 mm DBS samples, shaken for 5 minutes and diluted with water. The Oasis MAX SPE µElution Plate was conditioned with methanol prior to 700 µL supernatant being applied. This was followed by a wash with 1% (v/v) ammonia in 10% (v/v) acetonitrile (aq). Samples were eluted with 70% acetonitrile and diluted with water. 25 µL of extracted sample was injected on the ACQUITY UPLC I-Class System utilizing a 0.05 mM Ammonium fluoride/methanol gradient and a 2.7 µm, 2.1 mm x 50 mm CORTECS C18 Column with VanGuard Pre-Column. Detection was performed using Multiple Reaction Monitoring (MRM) on the Xevo TQ-S micro. The complete analytical workflow is shown in Figure 2.
DBS calibration lines were linear from 0.5–500 ng/mL for androstenedione and 11-deoxycortisol; and 1.0–500 ng/mL for cortisol, 17-OHP, and 21-deoxycortisol with correlation coefficients (r2) >0.99 over five occasions.
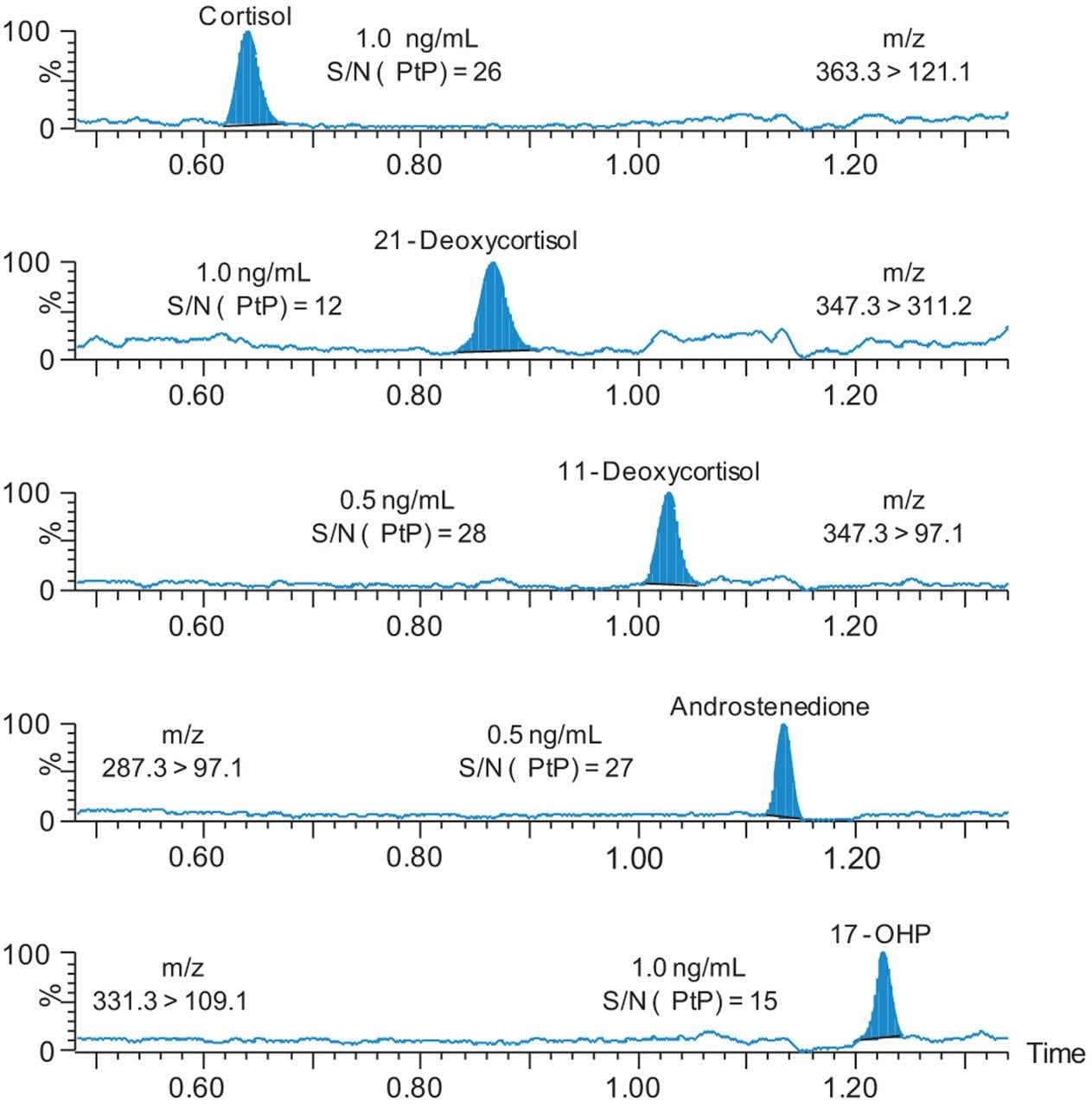
Signal/Noise (S/N) at the LLOQ calibrators are shown in Figure 3, demonstrating the analytical sensitivity of the method from two 3 mm dried blood spots.
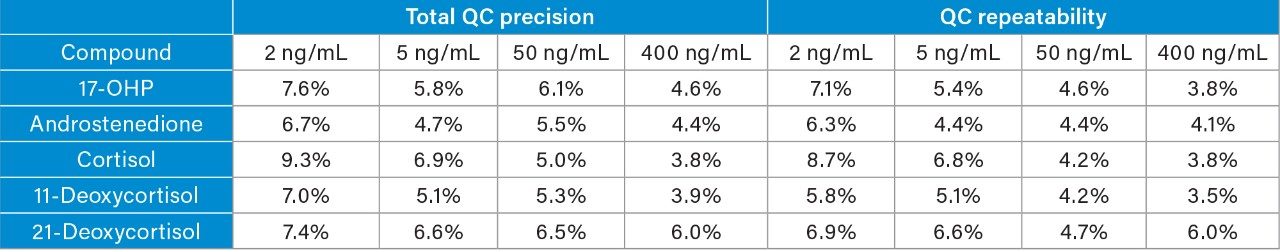
Total precision and repeatability of the method for analyzing the steroid hormones over five occasions was ≤9.3% CV (Table 1).
A UPLC-MS/MS method for the separation and detection of steroid hormones in dried blood spots has been developed for clinical research purposes. The benefits of this method include:
We would like to thank Susan Johnston and colleagues from NHS Glasgow and Clyde for the provision of 17-OHP DBS calibrators for use during initial investigations.
720006551, April 2019