This application note demonstrates a robust method for budesonide, formoterol, and related compounds was developed using a Quality-by-Design approach on an ACQUITY Arc System running Empower 3 and Fusion Software. This development was performed using an ACQUITY Arc System that is equipped with a column heater and solvent select valve to allow for automated exploration of a wide range of conditions.
Analytical methods are required to be developed at different stages of the pharmaceutical product life cycle. Two of the most widely used strategies for method development are: one-factor-at-a-time (OFAT) and analytical quality-by-design approach (AQbD). In OFAT, one variable is changed sequentially until a suitable method is obtained. This may create an adequate method, but it provides a limited understanding of method capabilities and method robustness. The AQbD, on the other hand, is a comprehensive, systematic, and risk-based approach for method development that starts with predefined objectives and provides rational understanding.1 In this approach, multiple variables are screened to provide a broad knowledge about the impact of the studied factors on the method performance. This knowledge is used to establish the method operable design region (MODR) which corresponds to the multi-dimensional combination of variables that have been verified to meet the method performance criteria. The outcome of this approach is a fit-for-purpose, well-designed, understood, and robust method that delivers the expected performance throughout its lifecycle.2,3
In this application note, a software assisted AQbD approach was implemented to develop a method for the analysis of formoterol, budesonide, and related compounds. This development was performed using an ACQUITY Arc System that is equipped with a column heater and solvent select valve to allow for automated exploration of a wide range of conditions. Fusion QbD method development software was used with an AQbD software in this study. Details about the use of Fusion QbD as an AQbD software for method development on an ACQUITY UPLC H-Class PLUS System has recently been described.4
Budesonide and formoterol fumarate reference standards were both purchased from USP (Rockville, USA). Budesonide related compounds G, E, and L were also all purchased from USP (Rockville, USA). Stock solutions of these compounds were prepared by accurately weighing the desired amounts of each standard and dissolving them in acetonitrile as a solvent. The stock solutions were then used to make a test mixture that contains all the previously mentioned APIs and impurities. This mixture was prepared by diluting the stock solutions of each standard in 70/30 (v/v) water/acetonitrile as sample solvent. The final concentrations of each analyte in the test mixture were approximately: 0.4 mg mL-1 budesonide, 0.15 mg mL-1 formoterol, 0.005 mg mL-1 related compounds (E and L), and 0.01 mg mL-1 related compound G.
| Data management: | Empower 3 Chromatographic Data System (CDS) and Fusion QbD |
|
System: |
ACQUITY Arc with Quaternary Solvent Manager (rQSM), Sample Manager (rFTN), Column Heater, PDA Detector, ACQUITY QDa Mass Detector |
|
Columns: |
(Five) 3.0 × 100 mm columns: BEH C18, 2.5 μm, pH range: 1–12 BEH Shield RP18, 2.5 μm, pH range: 1–11 CORTECS T3, 2.5 μm, pH range: 2–8 CORTECS Phenyl, 2.5 μm, pH range: 2–8 HSS PFP, 2.5 μm, pH range: 2–8 |
|
Flow rate: |
0.5 mL/min |
|
Mobile phase A: |
First screening: trifluoroacetic acid 0.1% Second screening: ammonium hydroxide 0.1% |
|
Mobile phase B: |
Acetonitrile (strong solvent [SS]) |
|
Mobile phase D: |
Solvent select valve that was used to select between several solvents at the different stages of the method development |
|
Screening: |
D1, 20 mM ammonium acetate |
|
Optimization: |
D1, 20 mM ammonium acetate/ammonium hydroxide buffer, pH = 8.0 D2, 20 mM ammonium acetate/ammonium hydroxide buffer, pH = 8.5 D3, 20 mM ammonium acetate/ammonium hydroxide buffer, pH = 9.0 |
|
Profile: |
Equilibrate at 10% organic for 3 min Isocratic at 10% organic for 1.0 min Gradient from 10–60% organic for gradient times ranging from 10–20 min Isocratic at 60% SS for 1.0 min Ramp down from 60% to 5% SS for 0.5 min Isocratic at 10% SS for 3 min Note: % Strong Solvent plus % Weak Solvents sum to 100% |
|
Column temp.: |
Constant 40 °C |
|
UV detection: |
244 nm |
|
Injection volume: |
3 μL working solution |
|
System: |
ACQUITY QDa Mass Detector |
|
Ionization mode: |
ESI+ |
|
Capillary voltage: |
0.8 kV |
|
Cone voltage: |
15 V |
|
Source temp.: |
600 °C |
In this part of the study, chromatographic parameters that are predicted to have significant effects on the method performance were selected for study. Since the stationary phase chemistry and the pH are strong affecters in chromatography, these two were set to be screened in this experiment. Five low pH levels of 2.0, 2.5, 2.9, 3.6, and 4.2 were all explored in this part of the study. For stationary phases, a wide range of versatile column chemistries was selected in order to achieve optimal separations of the analytes and their impurities. Five stationary phases were selected for study in this stage: BEH C18, BEH Shield RP18, CORTECS T3, CORTECS Phenyl, and HSS PFP. It is worth mentioning that the appropriate column geometries that match the ACQUITY Arc capabilities of 3.0 mm with a 2.x μm particle size were used in this experiment. The last chromatographic parameter that was selected to be varied in this experiment was the gradient time (10–20 minutes). All other chromatographic parameters including flow rate, temperature, and injection volume were set to be constant.
Using these parameters, an experimental design (DOE) was generated within Fusion QbD for all the required screening experiments. A statistical sampling approach was implemented by the software to obtain the maximum amount of information about the constants and the variables entered during the experimental setup. The total number of runs the software created for this screening experiment was 44. Note that this DOE design is highly efficient, since 125 methods would be required for all possible combinations (5 levels of tG × 5 levels of pH × 5 columns). When the experimental design was created, it was exported to Empower, creating all the methods and method sets that are needed for these runs. It also created and exported all the necessary conditioning/equilibration methods and method sets. This is particularly important as it significantly shortens the method development time by eliminating the time needed to manually generate methods and method sets for such a large number of experiments. Fusion QbD created two separate sample sets for Empower to run this screening on the five columns because the column heater (CH) can accommodate a maximum of only three columns at a time.
After the initial process and integration, results from the two sample sets were imported into Fusion QbD where mathematical models were automatically built and combined to predict the “Best Overall Answer” (BOA). The performance goals selected in this screening included both peak count-based and peak result-based “Trend Responses™”. The peak count-based Trend Responses used in this analysis were (1) the total number of peaks, and (2) the number of peaks with a USP resolution of ≥1.50 in each chromatogram.
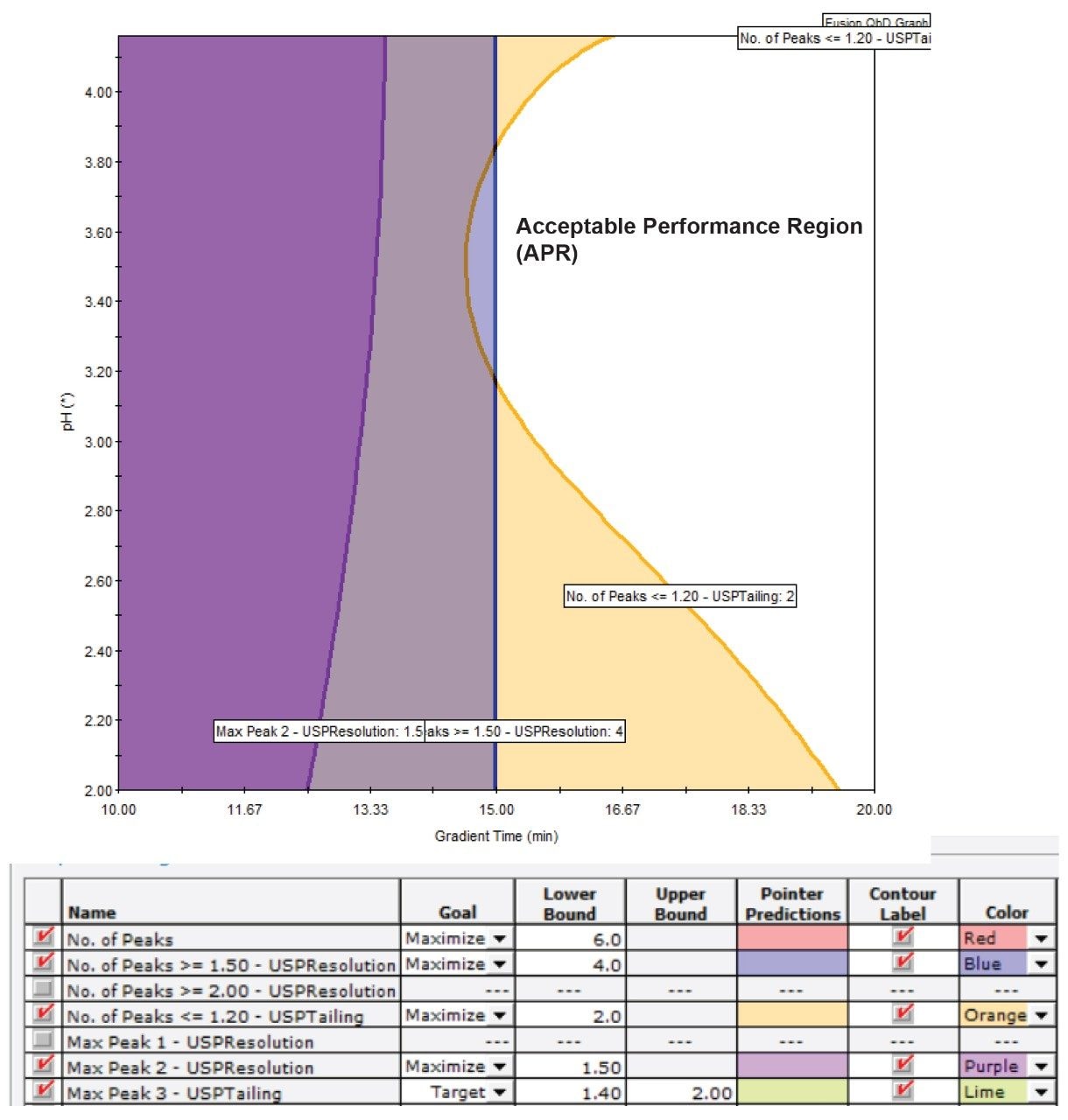
The next step was “Executing Search” in Fusion QbD. In this step, the software uses the measured responses to calculate models for each chromatographic result. These models calculated a “Cumulative Desirability Result” which is a numerical value that ranges from zero to one and represents the probability that the chromatographic conditions will meet the user specified performance goals. In this study, it was found that the best combination of conditions to achieve the set performance goals are: BEH C18 column, acetonitrile solvent, pH 2.0, and gradient time of 20 minutes. Fusion QbD models also provide a “Performance Map” that shows the “Acceptable Performance Region” (APR) – the unshaded region around the BOA where the method meets or exceeds the predefined screening performance goals (Figure 1).
The first chemistry screening experiment was reasonably successful in separating some of the analytes of interest, however, it was not able to achieve all the desired performance goals. For example, some analytes were not baseline resolved and the formoterol peak was unsymmetrical with tailing factor values between 1.38–2.13. As a result, a second screening experiment that explores different chromatographic conditions was performed to see if further improvements can be achieved.
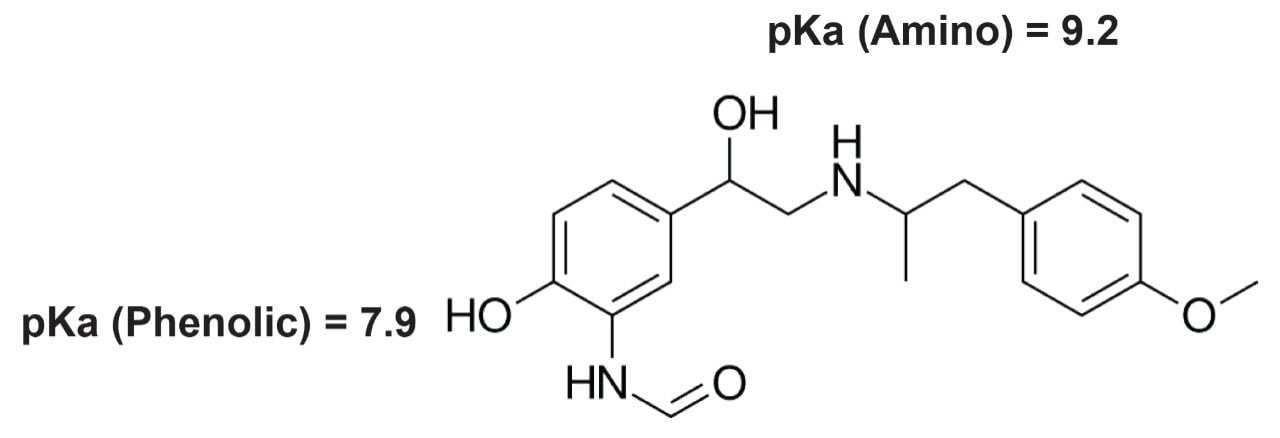
In particular, the main goal of this experiment was to improve the peak shape of formoterol fumarate and reduce the tailing. Since formoterol fumarate is a base (structure shown in Figure 2), it was logical to perform a second screening experiment under high pH conditions because the pH is a primary chromatographic parameter that has a major influence on the chromatographic performance. This screening was done by using the best column that was found in the previous experiment (BEH C18) and running the screening under high pH conditions (6.7 to 10.7). All other variables and constants were maintained at the settings used in the first experiment except for the gradient time that was adjusted from 10–20 to 12–25 minutes. Results of this experiment have shown that the formoterol peak shape improved significantly when high pH mobile phases were used. For example, the tailing for the formoterol peak was less than 1.5 for all 15 high pH screening runs. Figure 3 shows a “best looking” chromatogram from this screening experiment where the tailing of the formoterol peak was 1.2.
The results of these screening runs were next processed in Fusion QbD in order to find the BOA and the APR. The major performance goals that were set in this search included baseline resolution for at least four peaks and a target tailing of 1.2 for the formoterol peak. It was found that the tailing of formoterol decreases as the pH increases. The BOA search identified that using the BEH C18 column, a pH of 9.6 and a 25-minute run time were the best conditions for the next optimization experiment.
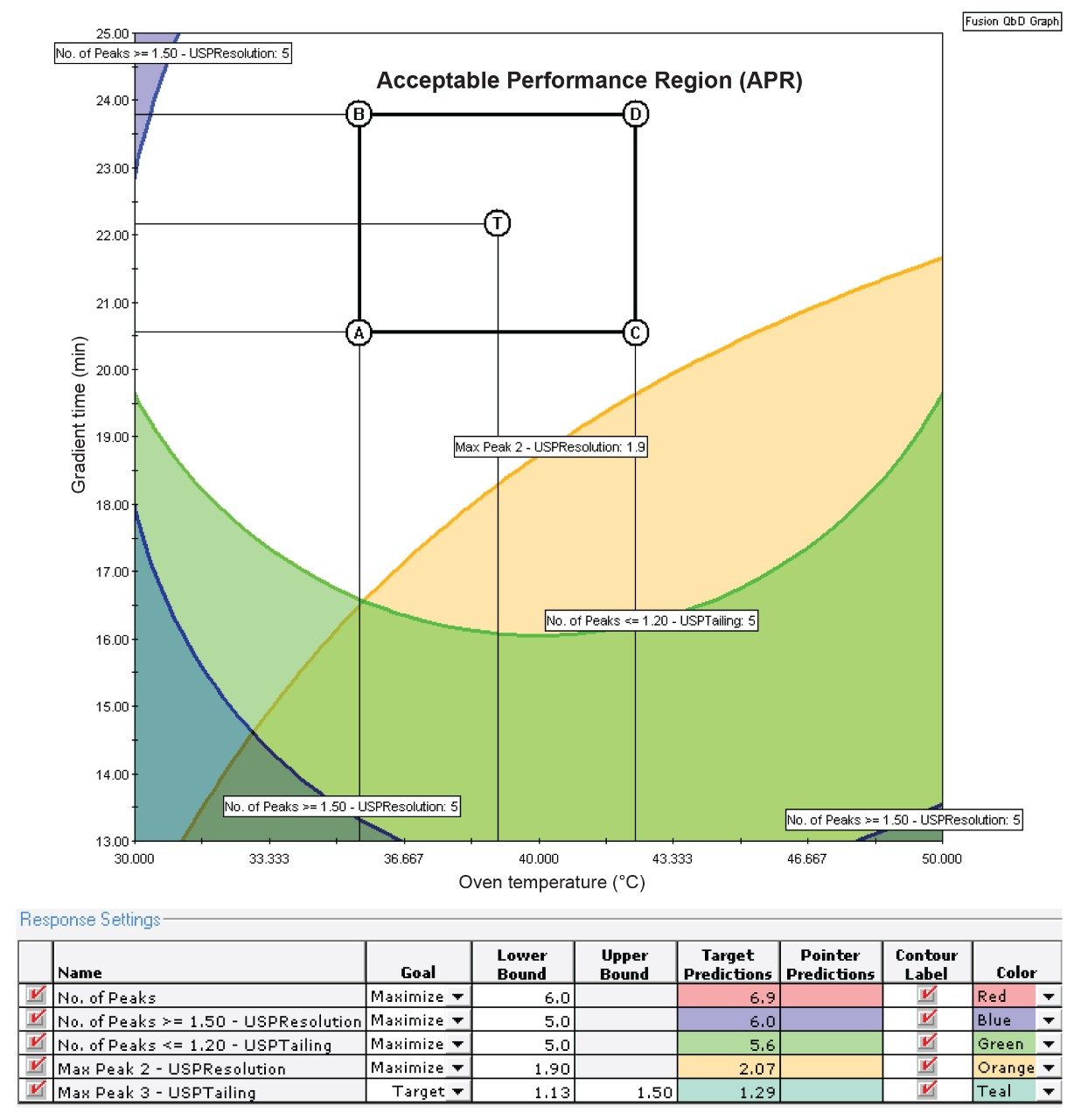
Optimization in LC method development is commonly done by fine tuning some of the less important parameters such as the temperature, gradient slopes, gradient times, and flow rates. In this optimization experiment, the flow rate and the temperature were varied. Two flow rates of 0.35 and 0.5 mL/min-1 and temperatures ranging from 30–50 °C were included. Three buffers at pH levels of 8.0, 8.5, and 9.0 were also studied. As the BEH C18 stationary phase has shown to be efficient at separating the active ingredients and the impurities, it was selected as the stationary phase in this optimization experiment. The experimental design was created again using the Fusion QbD Software and the results were also processed using the software to find the APR and the BOA. Results have shown that the desired performance goals can be achieved under wide ranges of experimental conditions. For example, as can be seen in Figure 4, all analytes of interest (seven peaks) can be separated with a minimum resolution of 2.0 and with a tailing factor of less than 1.3 over a wide range of temperatures (30–50 °C) and a suitable range of gradient times (20–25 minutes). The graph in Figure 4 also shows the location of the final method within the design space – the point designated as “T” – for target method. The rectangle within the final design space illustrates the joint “Proven Acceptable Ranges” (PARs) – the subset of the design space within which the graphed parameters can be independently adjusted post approval without requiring full re-validation of the method. These findings indicate that a robust method that achieves all the desired performance goals can be created in the middle of the APR space. It should be noted here that the models obtained from the optimization experiment are more precise than those obtained from the screening which enable the APR to now be considered a final design space. The analysis results identified the design space shown in Figure 4, which encompasses wide ranges of the experimental factors within which the desired performance goals can be achieved.
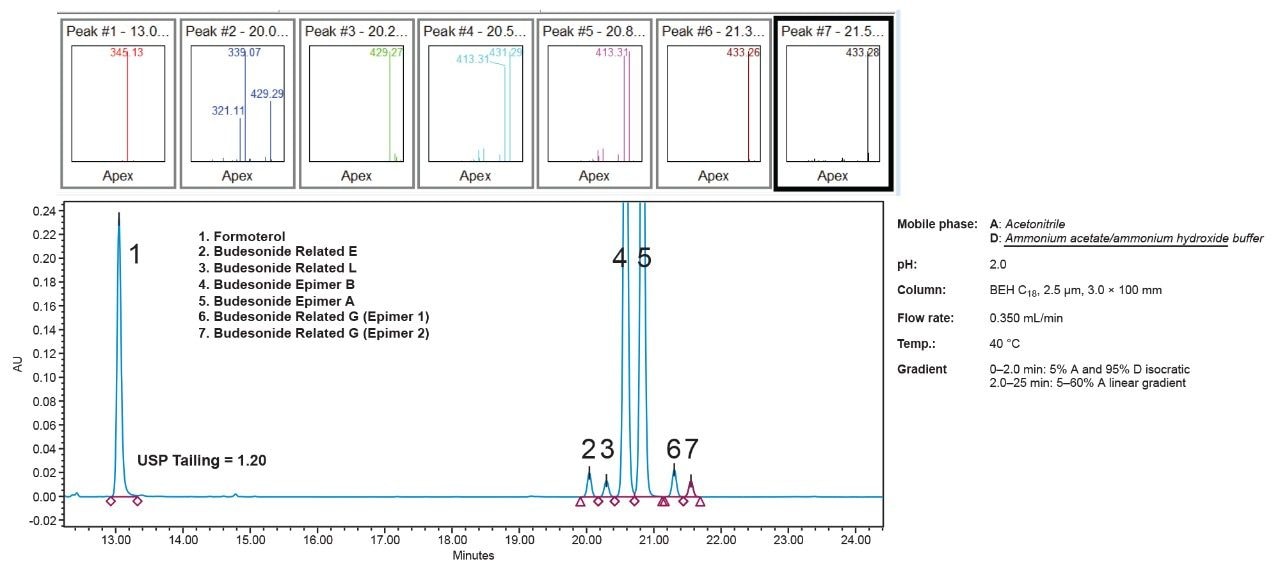
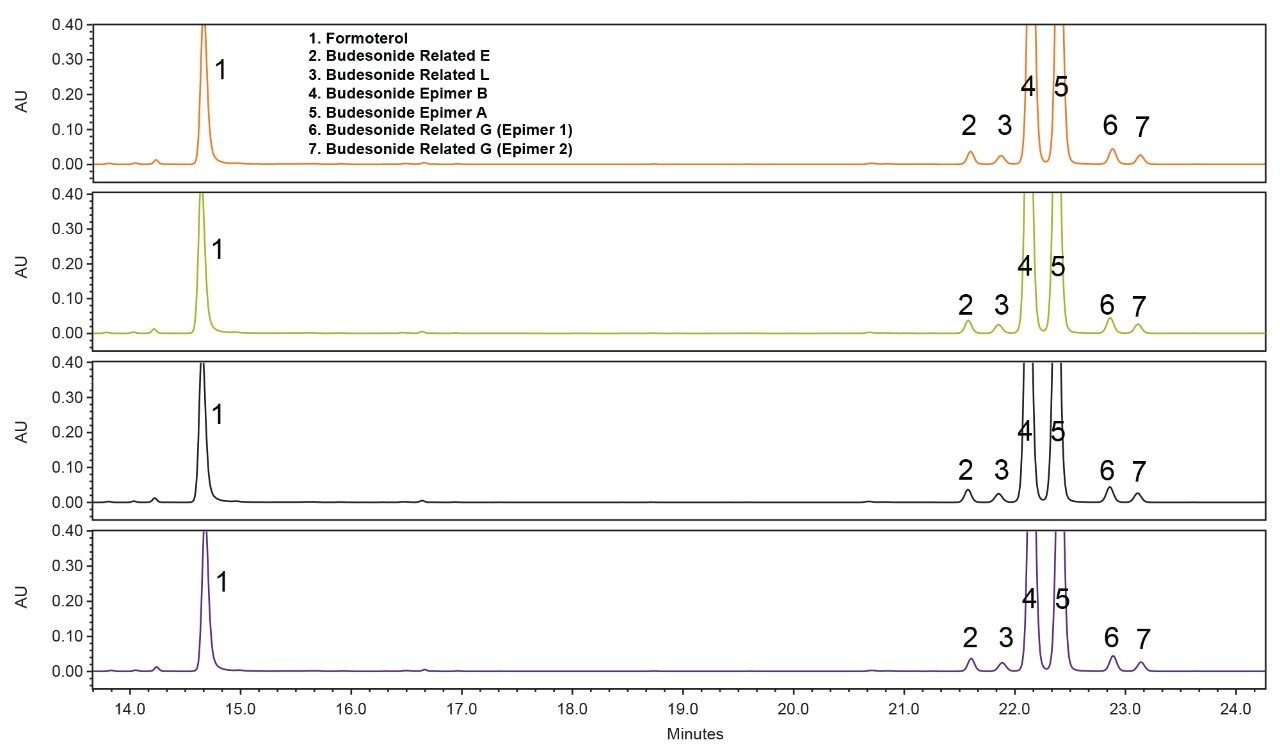
In order to verify the software-predicted results, it was necessary to run verification experiments and to compare their outcomes with the predicted results. This was done by running several experiments under the conditions that were predicted to give acceptable performance, and comparing the results of these experiments with the predictions. Results have shown (Figure 5) that the predicted performance matches the real performance reasonably well. For example, as can be seen in Figure 5, Fusion QbD predicted that the number of peaks under the BOA conditions (detailed in the graph) will be seven peaks and results of the real chromatogram (Figure 5) also showed seven peaks. Further, Fusion QbD predicted that six out of seven of these peaks will have a USP resolution of ≥2 and the real chromatogram have shown the same result. Finally, the Fusion QbD Software predicted that seven out of the seven peaks will have a tailing factor of equal or less than 1.2 and the real chromatogram has shown six out of the seven peaks had a tailing factor of less than 1.2. It is worth noting here that Fusion QbD Software can also successfully predict the performance for specific peaks in the chromatogram. For example, the predicted value of the resolution of budesonide epimer A was within less than 3% difference from the actual value when five different verification runs were performed.
— Automates the entire method development process
— Saves time by determining the minimum number of experiments needed for valid results
— Provides tools to quantify and visualize all important instrument parameter effects on all critical method performance characteristics
720006696, October 2019