
This application note evaluates the performance of three high-pressure binary LC systems, specifically in terms of retention time reproducibility, using a generic peptide mapping method. It evaluates the performance of the ACQUITY UPLC I-Class PLUS System along with two other vendor binary UHPLC systems when running a method which requires a long shallow gradient for separation of a complex sample.
Increased retention time reproducibility for long, shallow gradient methods using the ACQUITY UPLC I-Class PLUS System.
Generating liquid chromatographic methods which provide reliable and reproducible results is the end goal of method development. There are many factors which affect the reproducibility of a method, including but not limited to, mobile phase additives, pH, gradient slope/time, temperature, column geometry, particle size, and morphology, as well as proper column re-equilibration. The robustness of a method can also be impacted by the design of the LC system itself. This is often reflected in the performance specifications of a system or module outlining the conditions under which optimal performance is achieved. For example, high pressure binary pumps are known to provide the best performance for methods which require the use of long, shallow gradients, or those which impart a small change in solvent composition per column volume.
There are a variety of applications which may require the use of relatively long, shallow gradients. Some of these common application areas include biomolecule analyses, specifically peptide mapping1 and glycan analysis,2 as well as profiling of natural products, including traditional Chinese medicines.3 This application note evaluates the performance of three high-pressure binary LC systems, specifically in terms of retention time reproducibility, using a generic peptide mapping method. The LC systems ideally should perform identically from one injection to the next, as well as from analysis to analysis, which can extend over a period of anywhere from days to years depending on the aim of the analysis. This evaluation will highlight the exceptional reproducibility of the ACQUITY UPLC I-Class PLUS System as compared to other vendor offerings for methods which utilize long, shallow gradients.
Waters MassPREP Enolase Digestion Standard (p/n: 186002325) was reconstituted in 100 µL of 0.1% trifluoroacetic acid in water (per the Care and Use Manuals). Fresh sample was prepared daily by taking a number of vials, reconstituting in mobile phase A, vortexing, then pooling all samples. The resulting pooled sample was then divided among the three LC systems.
|
LC conditions |
|
|---|---|
|
LC systems: |
Vendor A binary UHPLC system Vendor B binary UHPLC system ACQUITY UPLC I-Class PLUS System configured with a CH-A |
|
Detection: |
Vendor A and Vendor B used UHPLC diode array detection while the ACQUITY UPLC I-Class PLUS used an ACQUITY UPLC TUV Detector |
|
Sample: |
MassPREP Enolase Digestion Standard |
|
Column: |
Waters ACQUITY UPLC Peptide BEH C18, 130Å 1.7 um, 2.1 × 100 mm (p/n: 186003555) |
|
Column temp.: |
65 °C |
|
Mobile phase A: |
0.1% Trifluoroacetic acid in water |
|
Mobile phase B: |
0.1% Trifluoroacetic acid in acetonitrile |
|
Flow rate: |
0.200 mL/min |
|
Injection volume: |
10 μL |
|
Wavelength: |
214 nm |
|
Collection rate: |
10 Hz |
|
Needle wash: |
25/75 Mobile phase A/Mobile phase B |
|
Seal wash: |
80/20 Water/methanol |
Empower 3 CDS, FR2, and Chromeleon CDS, 7.2
Note: Each day, fresh mobile phases were prepared in one large pool and then divided among the three systems to reduce any potential variability.
Long, shallow gradients are often required to resolve complex samples. One such application area is peptide digests, which can be quite complex depending on the size of the starting protein and the digestion enzyme used. For very complex peptide digests, where the separation of many peaks is required, run-to-run retention time reproducibility is of the utmost importance for tracking peaks. The reproducibility of any separation is dependent on many factors, including sample preparation, column, LC method conditions, as well as the LC instrumentation used. For this work, the impact of the LC instrument is the main focus; therefore all other variables were controlled. For an in depth review of the protocol used, refer to reference 4.
As stated in the introduction, due to the potential complexity of peptide digests, LC methods often consist of very shallow gradients run at low flow rates in an effort to separate as many components as possible, leading to improvements in characterization. Additionally, the retention of peptides and proteins in reversed-phase chromatography is strongly dependent on small changes in solvent strength,1 which is another reason why shallow gradients are employed. Separation conditions commonly start with a low % of organic solvent, often times less than 5%, to retain highly polar or early eluting peptides. Binary systems were chosen for the evaluation since these pumps, which utilize high pressure mixing, offer better compositional accuracy and precision than do their low pressure mixing counterparts. Specifically, the compositional accuracy and precision of a binary pump depends only on the flow accuracy for each pump head. In contrast, for a ternary or quaternary mixing pump, the compositional precision and accuracy depends on the ability of the gradient proportioning valve or apparatus to perform in a consistent and correct manner. For these reasons, a set of three UHPLC binary systems were chosen for the evaluation, including the ACQUITY UPLC I-Class PLUS System as well as two other vendor offerings.
The three binary systems were configured as similarly as possible. Solvent preheating was used across all systems. Active solvent preheating was used when available; however, in its absence, passive preheating was employed. Three columns from the same manufacturing lot were used, again to avoid any variability arising from the method conditions as opposed to the specific LC system.
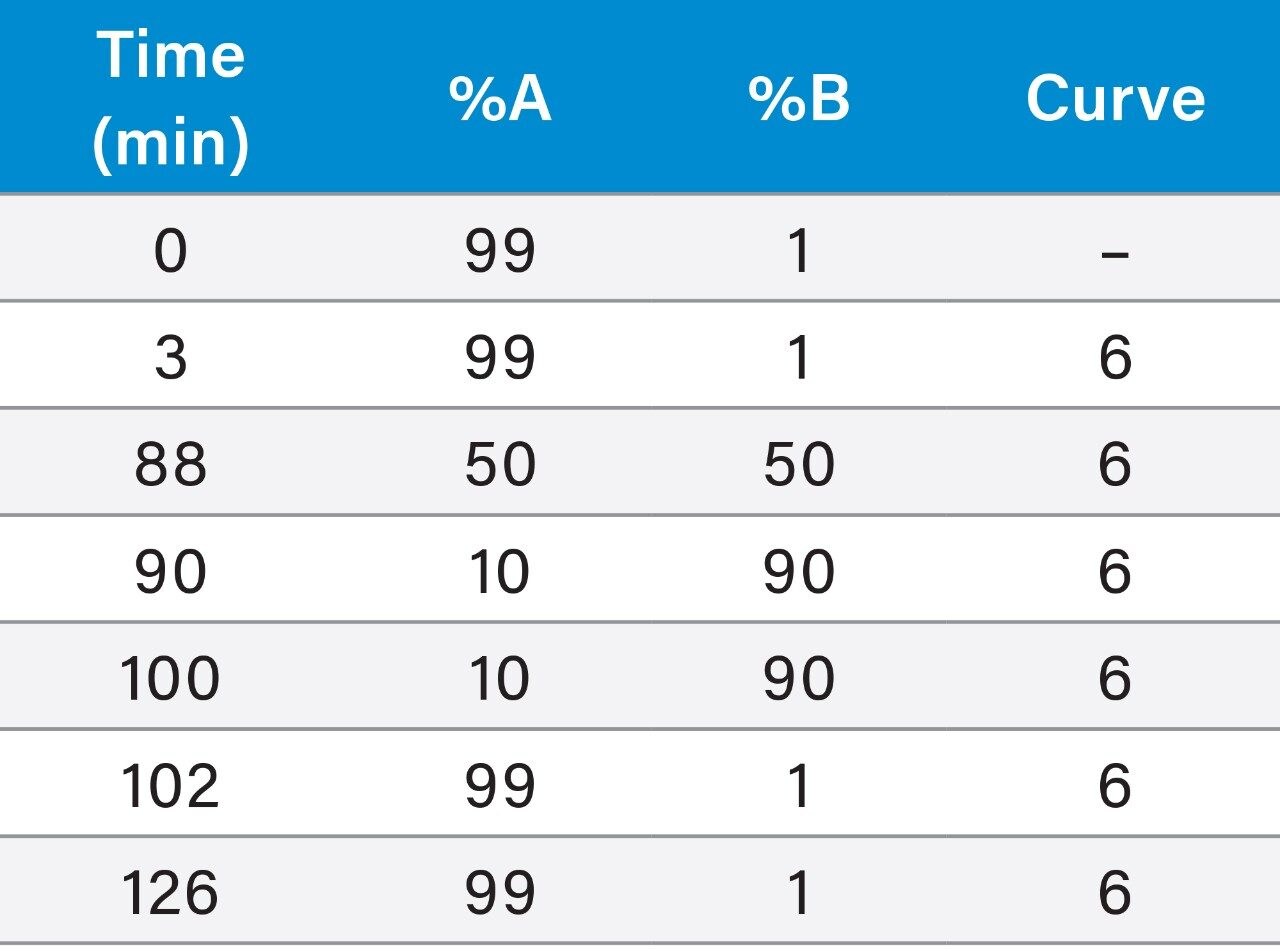
Example chromatograms from the three binary systems are shown in Figure 1. There were slight differences in the elution pattern or selectivity for the three systems, as well as differences in retention time. This is due to differences in the specific system volume, gradient delay or dwell volume, system dispersion, and mixing behavior of the three systems.
The ability of an LC system to provide a precise and reproducible result over a number of injections is important for a number of reasons including identification purposes. For example, if gradient delivery varies slightly from injection to injection, small selectivity differences may appear. These can impact the resolution between peaks, particularly for critical pairs or peaks which are only partially separated since the retention of specific peptides can be greatly affected by a small change in % organic. As a result, peak assignment based solely on retention time or elution order can be challenging.
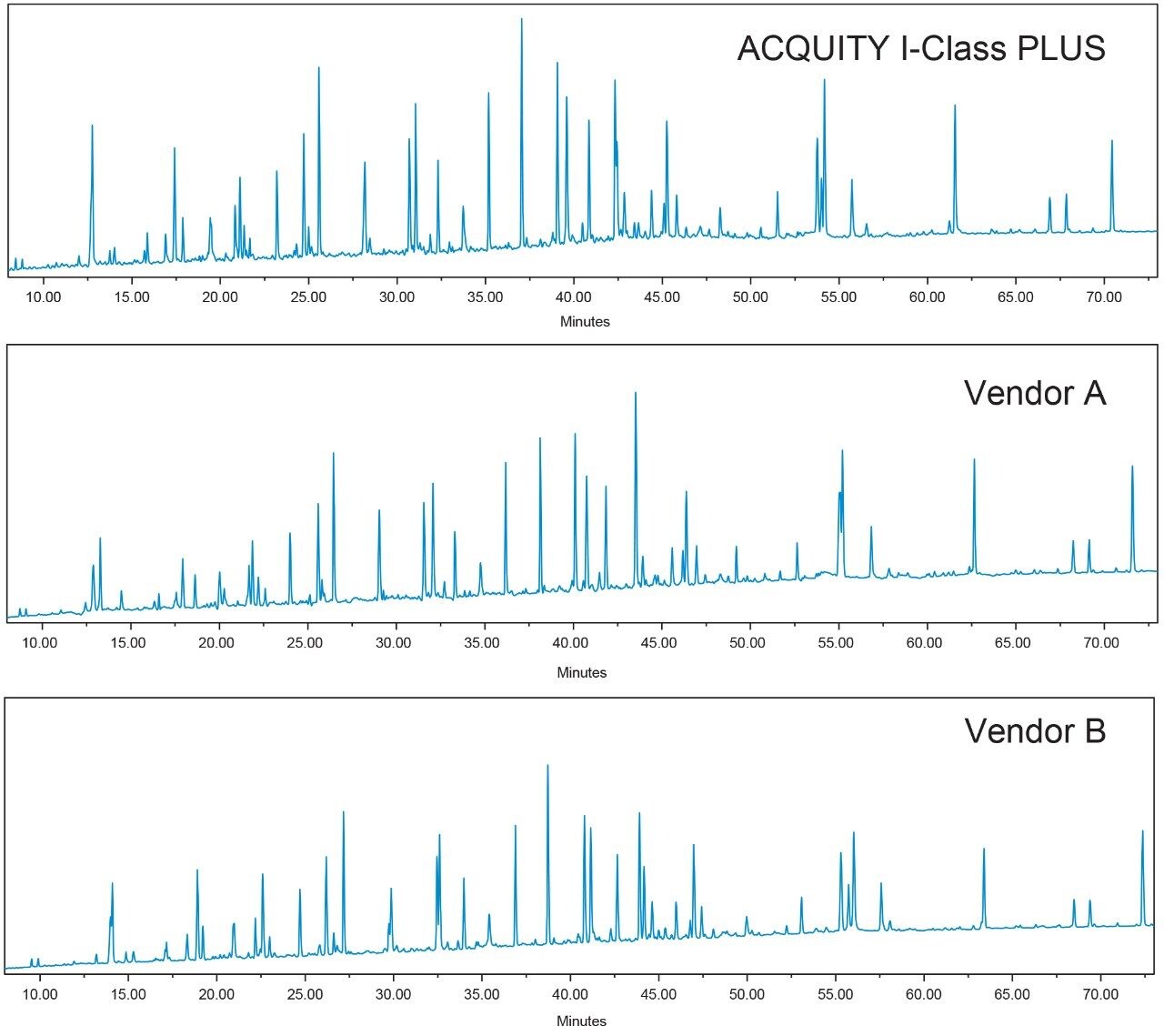
In these instances, the inability of a pump to deliver accurate and reproducible gradient performance will be seen in the resulting retention time reproducibility. For the peaks that were resolved and identified on all three systems, the retention times of eight replicate injections were compared and the resulting retention time standard deviations (RT SDs) are graphed in Figure 2. In addition to the standard deviation of the individual peaks, the average standard deviation across all peaks was calculated and determined to be 0.012, 0.062, and 0.033 min for the Waters ACQUITY UPLC I-Class PLUS System, Vendor A binary system, and Vendor B binary system respectively.
The ACQUITY UPLC I-Class PLUS System overall showed the best retention time reproducibility of the three systems evaluated. The average standard deviation was determined to be 0.012 minutes, or 0.7 seconds. In contrast, the Vendor A binary system showed the highest standard deviation, with an average RT SD of 0.062 minutes, or 3.7 seconds. The very small standard deviation values of the ACQUITY UPLC I-Class PLUS System highlight the consistent gradient delivery across injections.
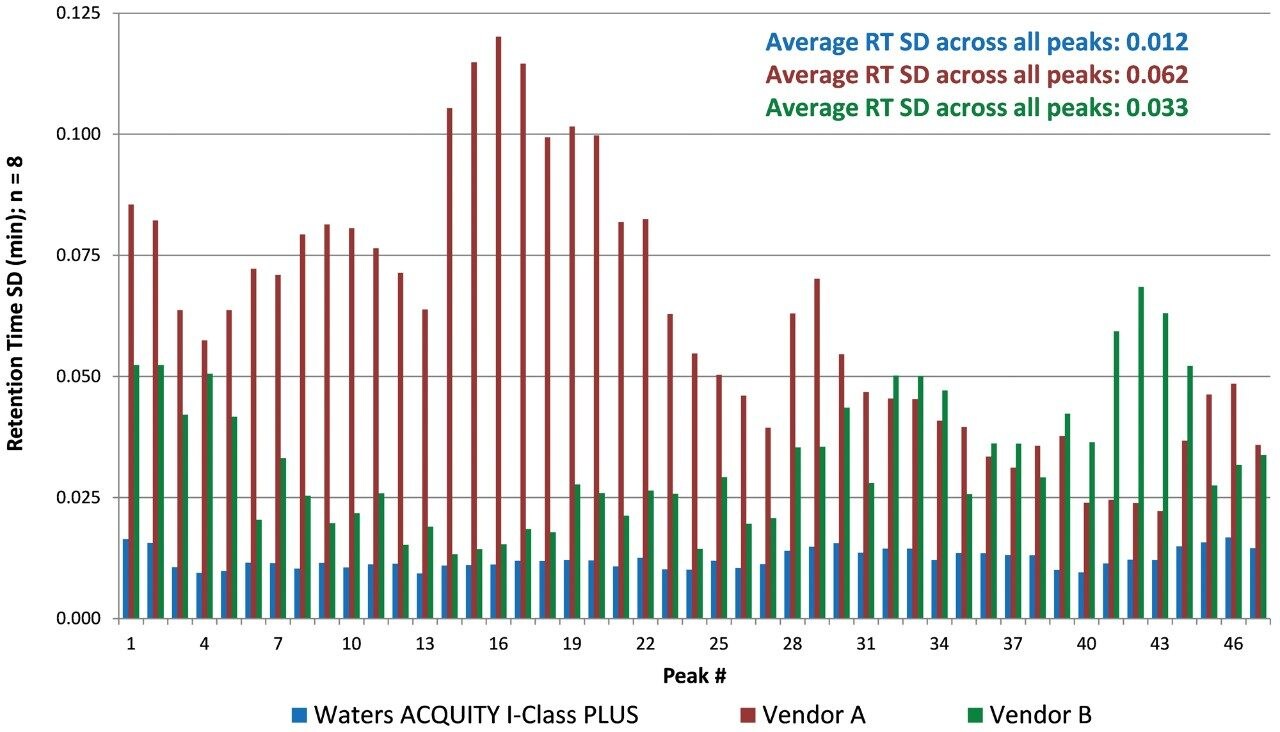
The reproducibility of each system is more apparent when looking at a smaller retention time window. Specifically, the overlaid chromatograms (time window of 30–48 min) are shown in Figure 3, as well as the retention time window for a specific peak. The retention time windows vary for each system due to differences in the gradient delay volume. These chromatograms illustrate the RT reproducibility of the three systems. For the ACQUITY UPLC I-Class PLUS System, all injections overlay with very little disparity in RT between injections, as indicated by the low RT SD value of 0.015 minutes. For Vendor A, the RTs spanned a wider range, as seen in the RT SD value of 0.070 minutes, indicating differences in the gradient delivery from injection to injection. Vendor B produced intermediate retention time results over the eight injections, as indicated by a RT SD of 0.035 minutes. It should also be noted that the shifting retention times seen on Vendor A and B systems did not follow a specific trend, but rather were random throughout the replicate injections (data not shown).
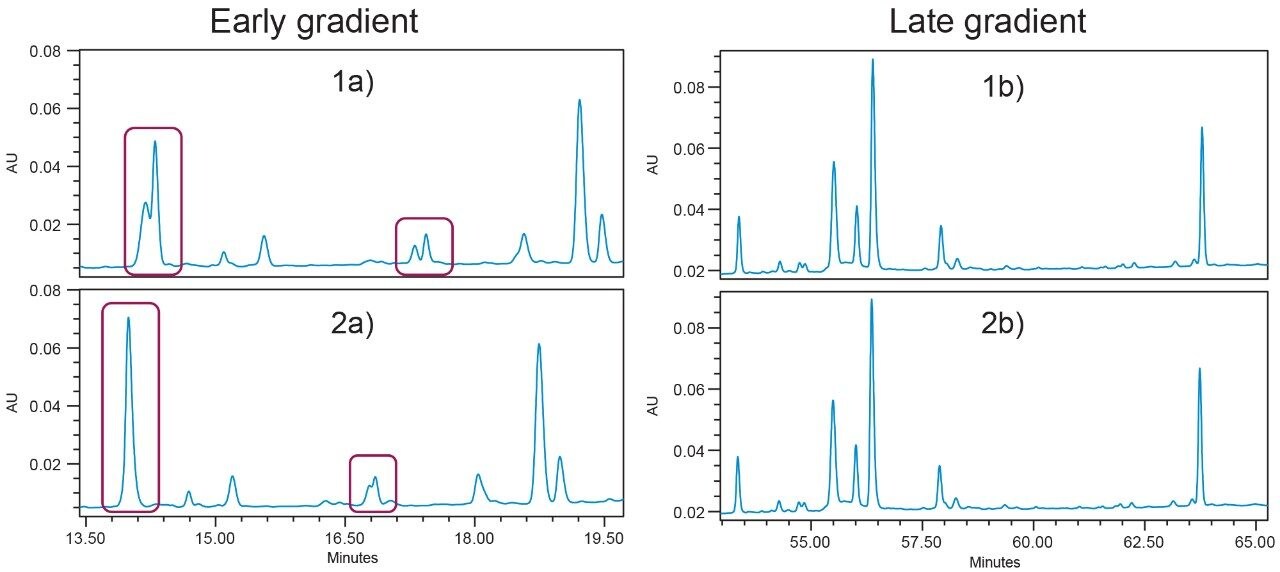
As previously stated, reproducible gradient delivery is important for peak identification and tracking. It is also important that the gradient delivery be accurate so that the method developed will perform the same regardless of when it is run. Figure 4 depicts two subsequent injections of the same enolase digest sample run with the same method conditions acquired on the Vendor B binary system. Chromatograms A and B (left) illustrate elution behavior early in the gradient, while C and D show elution behavior late in the gradient (right).
In the top left chromatogram (1a), there are two distinct peaks eluting at approximately 14.15 and 14.25 minutes. However, the next injection (2b) shows a single peak eluting at approximately 14 minutes. With only this information, it is impossible to identify the peak in chromatogram 2a as corresponding to either peak seen in chromatogram 1a. All peaks in the early part of the gradient (2a) show retention times which have shifted towards earlier elution and there is a loss of resolution between critical pairs (red boxes). By the end of the gradient (1b and 2b) however, all peak retention times are much more similar. Any inconsistency in the gradient delivery that affects the chromatography can lead to ambiguous or incorrect results that ultimately will require additional data to be collected. This impacts not only the quality of the data, but also increases the amount of work required to obtain the final result.
The performance and reproducibility of an LC system has the potential to impact peak identification, quantification and peak tracking when running an analytical procedure. Additionally, challenging method conditions, such as long shallow gradients, may negatively impact retention time reproducibility. This study evaluates the performance of the ACQUITY UPLC I-Class PLUS System along with two other vendor binary UHPLC systems when running a method which requires a long shallow gradient for separation of a complex sample. The ACQUITY UPLC I-Class PLUS System showed the most consistent gradient formation, enabling the generation of reproducible retention times, thereby generating high quality and reproducible data.
720006369, August 2018