
In this application note we present the use of a simple sample extraction and dispersive solid phase extraction (dSPE) cleanup procedure followed by UPLC-MS/MS analysis for rapidly monitoring the Oregon Cannabis Pesticide Guide List in cannabis matrix. With so many compounds to monitor, method generation can be a tedious task. In this study, the pre-existing LC and MS methods from Waters' Quanpedia Database were used to develop and implement a rapid solution for the Oregon pesticide list.
The increased use of both medical and recreational cannabis in combination with its expanding legal acceptance in most US states1 has led to rigorous cannabis safety and quality control testing. Pesticides are widely used in the cultivation of cannabis plants to safeguard against harmful insects and to promote better crop yields. The application of pesticides is regulated,2 and their residues in cannabis products are closely monitored by state regulatory agencies. The number of pesticides and their action limits varies from state to state. In Oregon, 59 pesticides are monitored with action limits ranging from 100 to 2000 ppb. Therefore adopting a robust and rapid procedure for monitoring the Oregon pesticide list in cannabis products is critical.
Multi-residue pesticide detection is routinely performed using tandem quadrupole mass spectrometry (MS/MS) in combination with liquid chromatography (LC) and gas chromatography (GC). Both LC-MS/MS and GC-MS/MS are commonly used for multi-residue pesticide analysis as some pesticides are only amenable to either LC or GC. Tandem quadrupole MS is the detector of choice as it provides high sensitivity and selectivity for simultaneous analysis of hundreds of pesticides at low ppb (ng/g) levels in a single analysis.
In this application note we present the use of a simple sample extraction and dispersive solid phase extraction (dSPE) cleanup procedure followed by UPLC-MS/MS analysis for rapidly monitoring the Oregon Cannabis Pesticide Guide List3 in cannabis matrix. With so many compounds to monitor, method generation can be a tedious task. In this study, the preexisting LC and MS methods from Waters Quanpedia Database were used to develop and implement a rapid solution for the Oregon pesticide list.
Standard compounds for the 59 pesticides monitored on the Oregon list were combined to produce a stock solution which was sequentially diluted to prepare the spiking solutions. Cannabis buds were first ground using a hand grinder. A 0.5 g portion of the ground material were weighed into 50 mL centrifuge tubes and spiked with 200 ppb of the acetonitrile spiking solutions. A 5-mL volume of acetonitrile was added and the samples were processed using a Geno Grinder (two stainless steel grinding balls, 11 mm) for 5 minutes (1500 rpm). The samples were then centrifuged at 5000 rpm for 5 minutes. For experiments where no further cleanup was performed, the supernatant was filtered using a 0.2 μm PTFE filter in preparation for analysis.
A 1 mL aliquot of the supernatant was added to a dSPE tube (2 mL centrifuge tube containing 150 mg MgSO4, 50 mg PSA, 50 mg C18, 7.5 mg graphitized carbon black), vortexed for 1 minute, centrifuged, and the supernatant transferred to a sample vial for analysis by UPLC-MS/MS.
All separations were performed on the Waters ACQUITY UPLC H-Class System and the Xevo TQ-S micro Tandem Quadrupole Mass Spectrometer. MassLynx MS Software (v4.1) was used for data acquisition and processing. The Quanpedia Database can be used to automatically generate LC, MS acquisition, and TargetLynx data processing methods to reduce method setup times with minimal user interaction.
|
UPLC system: |
ACQUITY UPLC H-Class |
|
Separation mode: |
Gradient |
|
Column: |
XBridge BEH C18 XP, 130Å, 2.5 μm, 2.1 mm × 100 mm, P/N: 186006031 |
|
Solvent A: |
5 mM Ammonium formate with 0.020% formic acid in water |
|
Solvent B: |
Methanol |
|
Flow rate: |
0.50 mL/min |
|
Column temp.: |
30 °C |
|
Injection volume: |
5 μL |
|
Time (min) |
%A |
%B |
Curve |
|---|---|---|---|
|
0.00 |
98% |
2% |
– |
|
0.20 |
98% |
2% |
6 |
|
11.50 |
1% |
99% |
6 |
|
13.00 |
1% |
99% |
6 |
|
13.25 |
98% |
2% |
1 |
|
15.00 |
98% |
2% |
1 |
|
MS system: |
Xevo TQ-S micro |
|
Ionization mode: |
ESI+/ESI- |
|
Capillary voltage: |
2.5 kV (+); 2.4 kV (-) |
|
Cone voltage: |
Various V |
|
Collision energy: |
Various eV |
|
Desolvation temp.: |
450 °C |
|
Source temp.: |
150 °C |
|
Desolvation gas flow: |
1000 L/Hr |
|
Cone gas: |
50 L/Hr |
Quanpedia Database was used to automatically create the LC, MS, and data processing methods (Figure 1) for the various target pesticides monitored using the transitions listed in Table 1. Users can quickly generate pre-defined LC-MS/MS methods in three easy steps, which greatly reduces the potential for error and level of complexity involved in method development for large numbers of target analytes. As a result, it decreases the amount of work, time, and resources required for laboratories to set up methods.
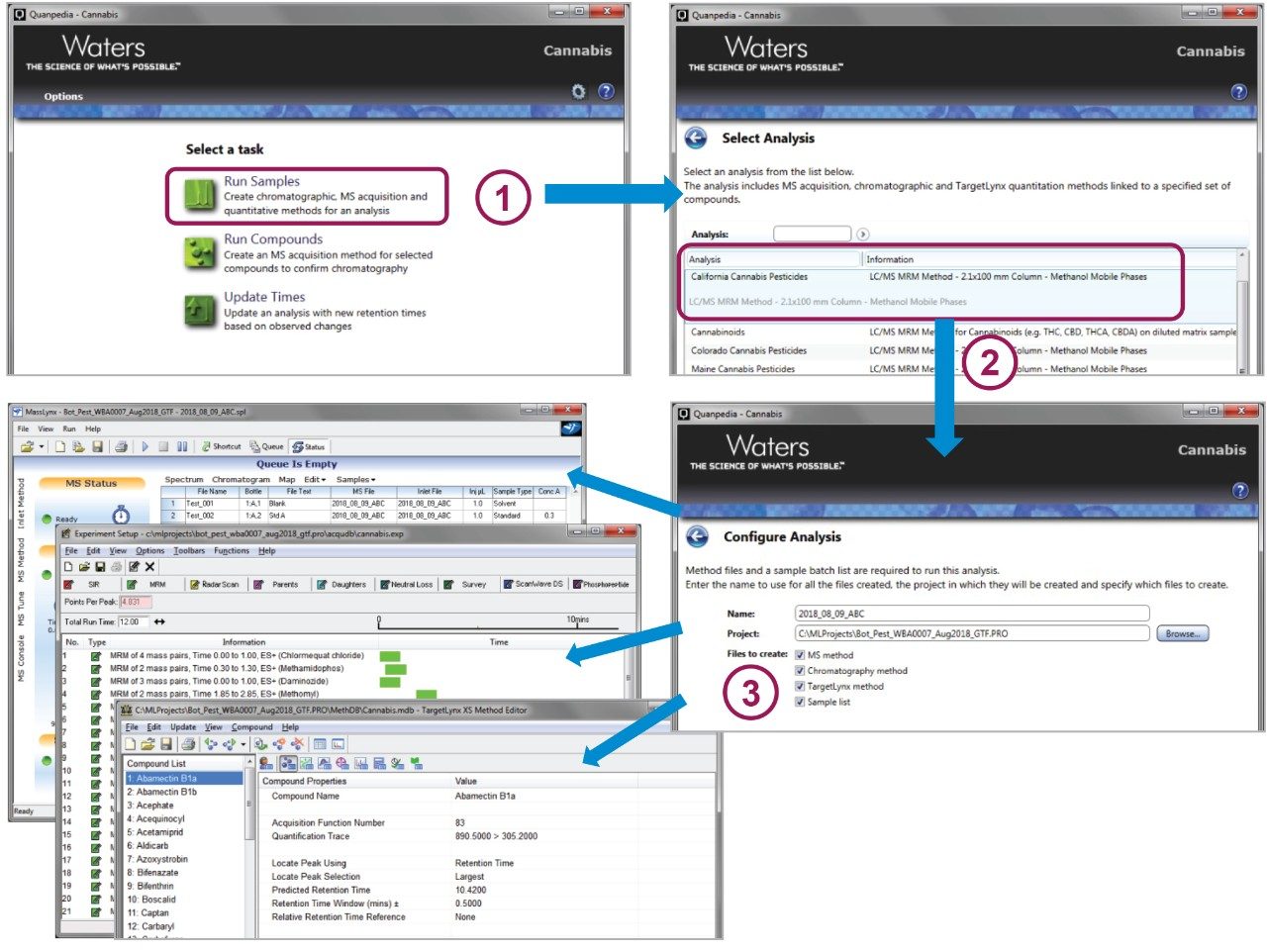
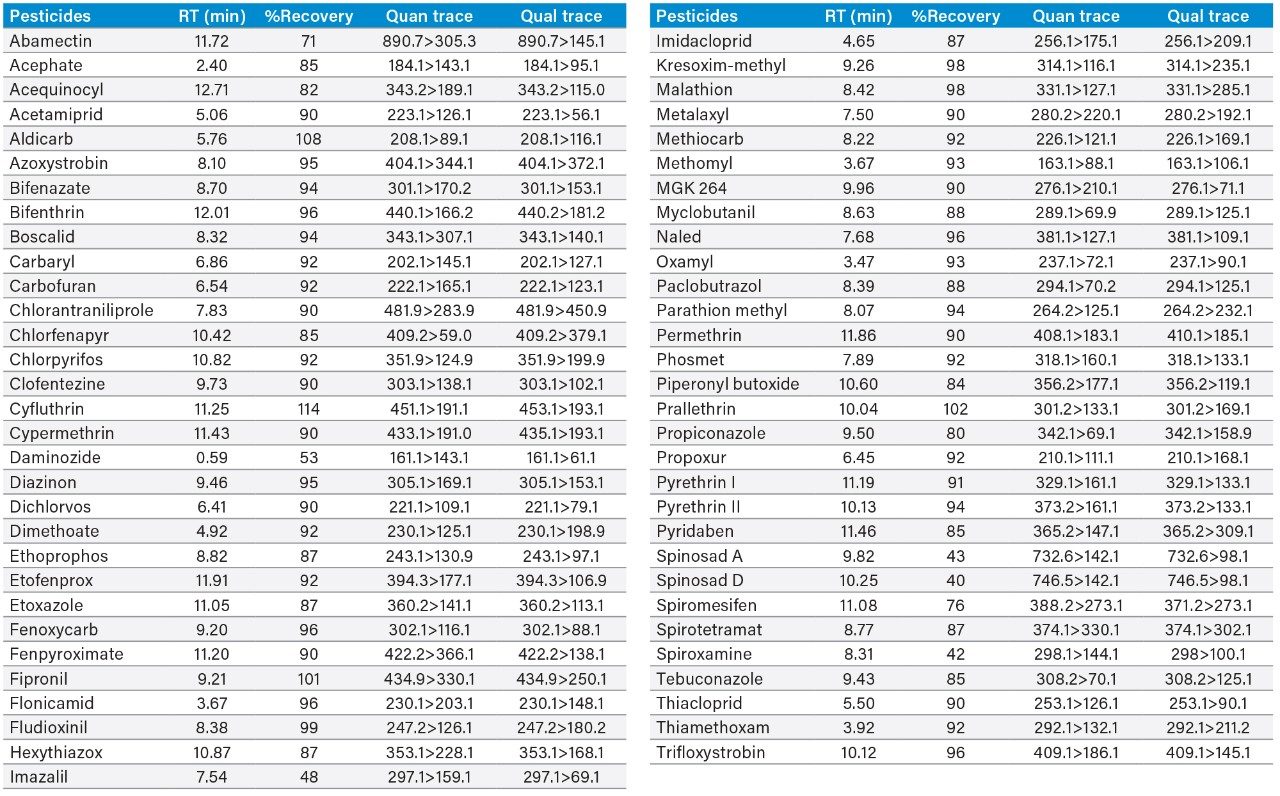
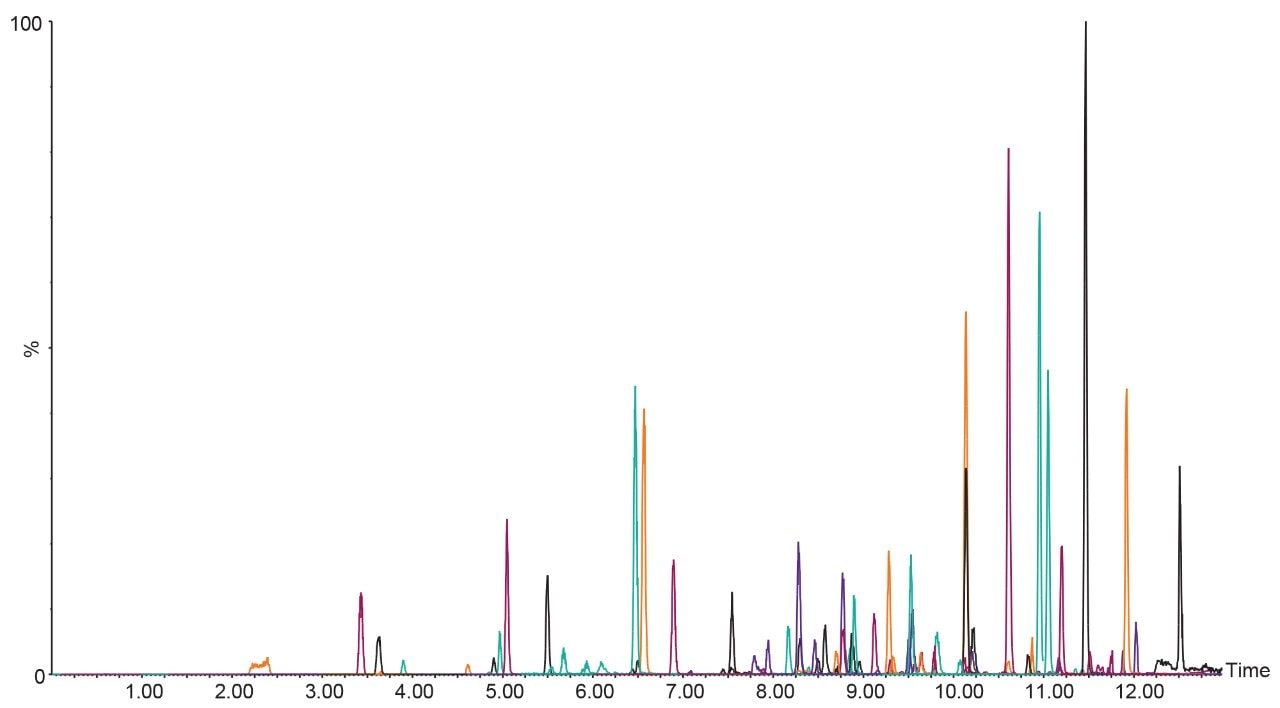
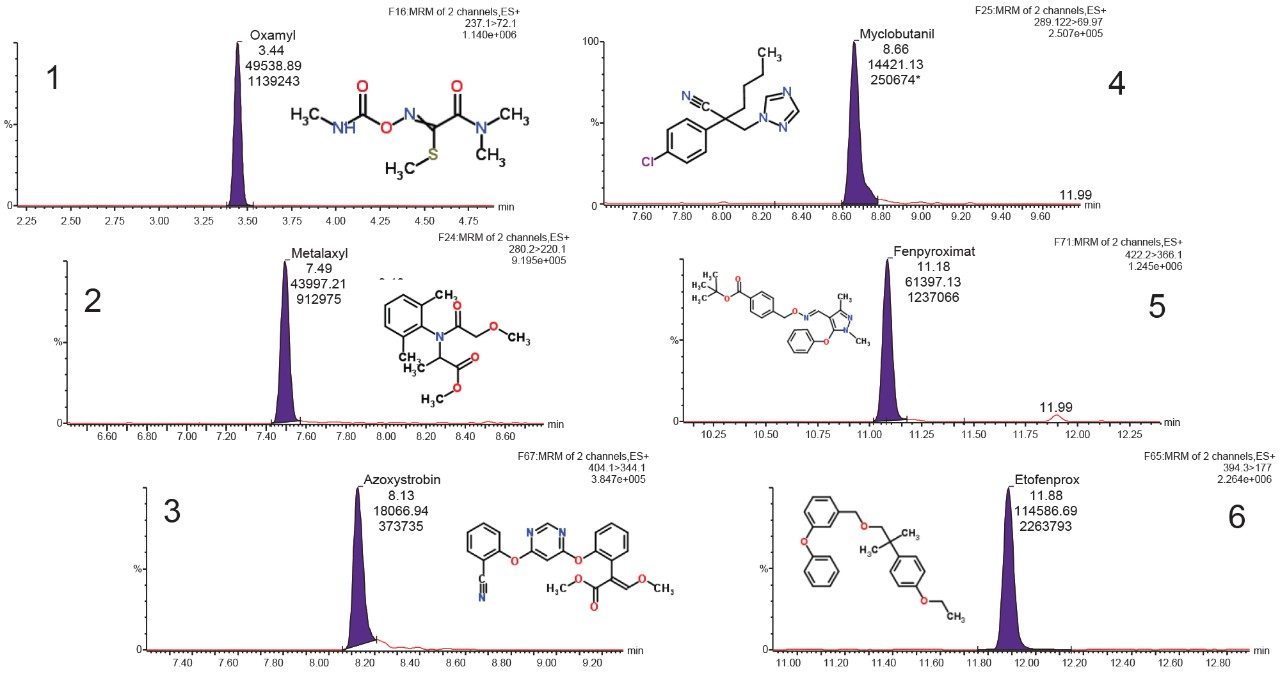
Figure 2 shows an overlay chromatogram of 59 pesticides analyzed by UPLC-MS/MS. MRM chromatograms of selected pesticides in cannabis matrix are shown in Figure 3.
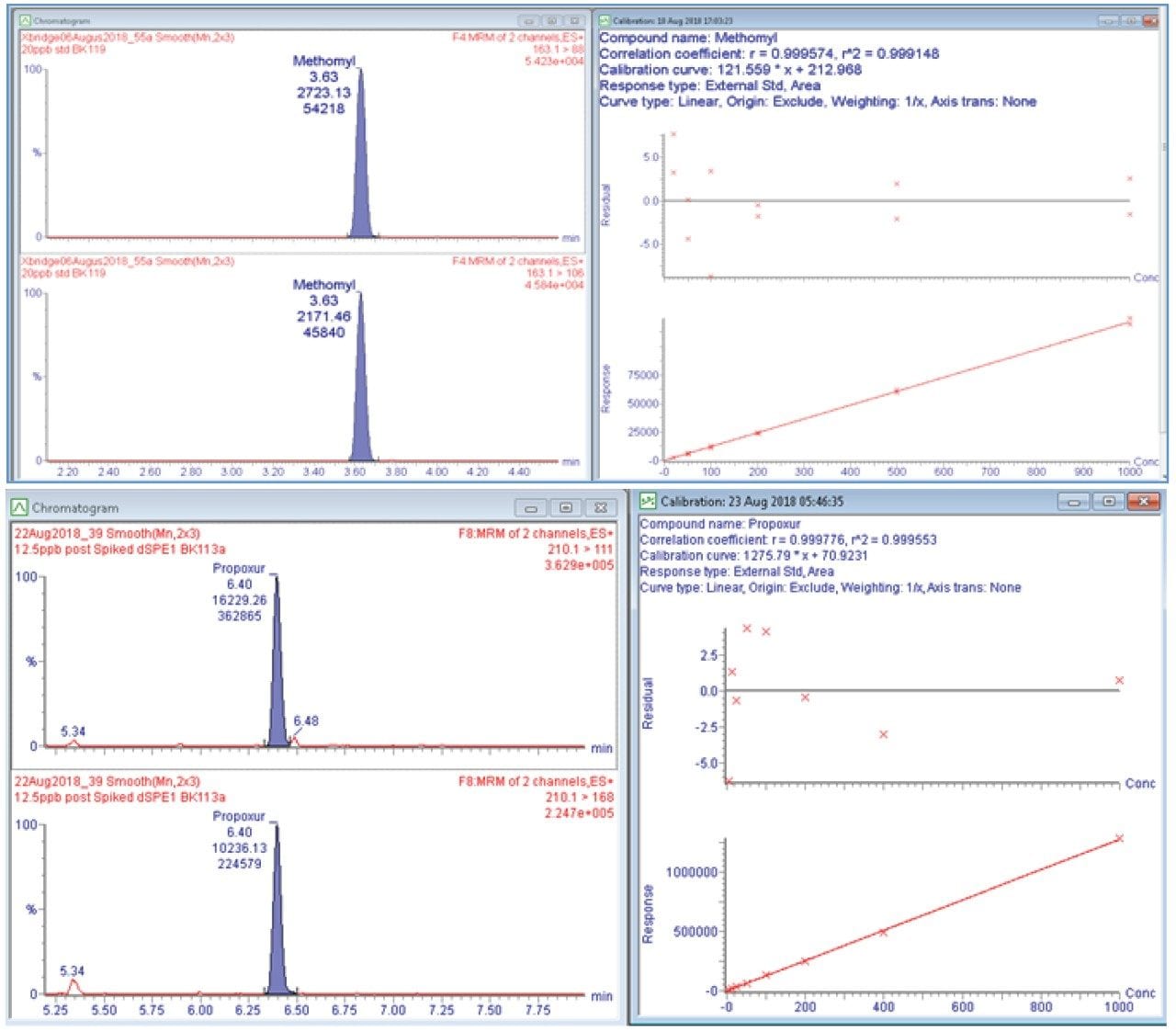
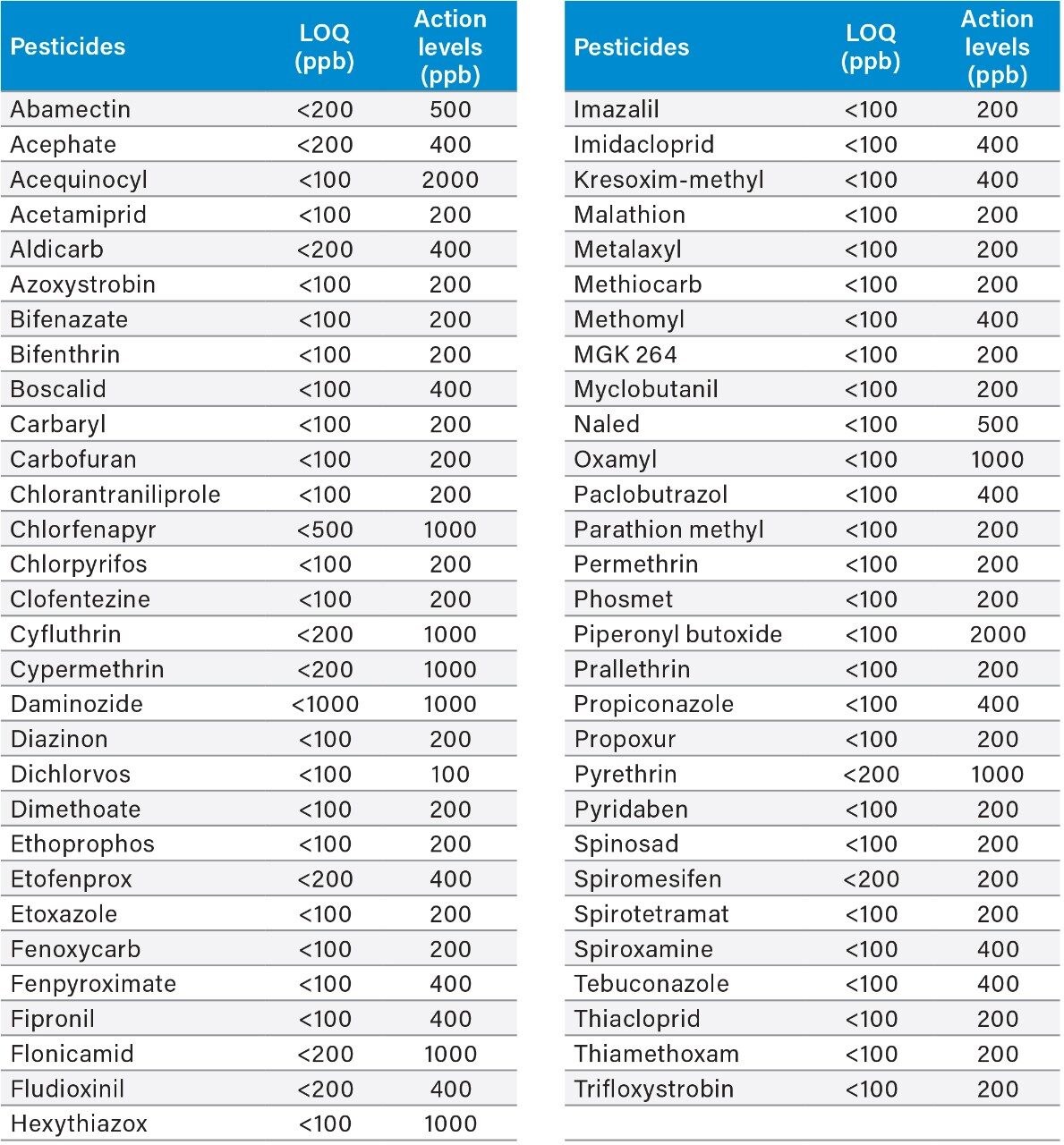
An example of the quantitation curve for methomyl and propoxur are shown in Figure 4. Linear calibration curves (R2>0.990) for each pesticide were obtained over the range tested 6.25 to 1000 ppb in matrix. Table 2 highlights the limit of quantitation (LOQ) and action limits per the Oregon Cannabis Pesticide Guide List.3
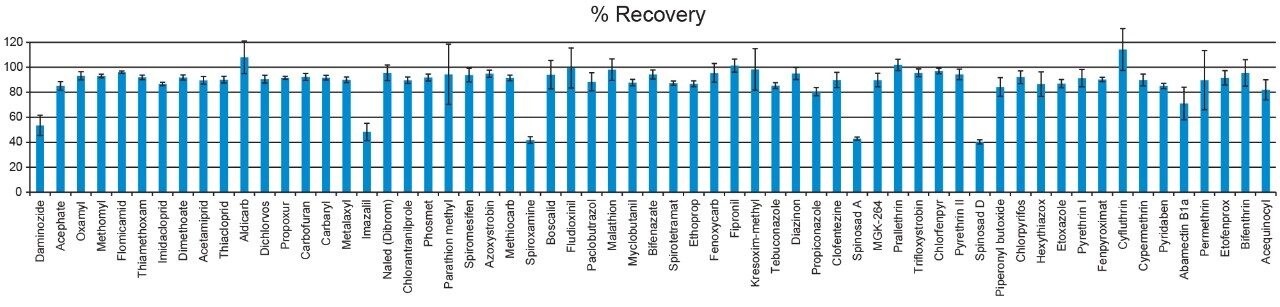
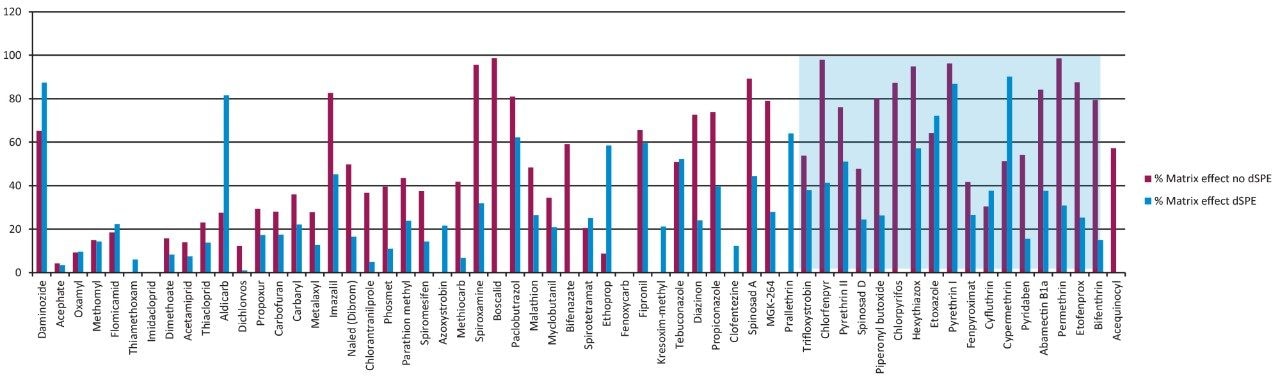
Method recovery was assessed by spiking pesticides at the 200 ppb and 1000 ppb levels in a cannabis flower matrix and comparing the response to that observed from spiked matrix blanks (matrix-matched standards). As shown in Figure 5, the recoveries observed for most of the pesticides were in the range of 80% to 120%. Matrix suppression was determined at the 200 ppb level by comparing the response observed in matrix-matched standards to the response observed in the solvent standards. Matrix suppression data is presented in Figure 6. Those compounds that co-eluted with the cannabis resin constituents (retention times from 9 to 12 minutes) showed the greatest suppression before dSPE cleanup. The dSPE cleanup provided a significant reduction of suppression for most of the compounds.
This simple sample extraction and dSPE cleanup method followed by UPLC-MS/MS analysis using the ACQUITY UPLC H-Class System coupled to the Xevo TQ-S micro Tandem Quadrupole Mass Spectrometer provides a rapid, sensitive, and robust method for determination of the Oregon Cannabis Pesticide Guide List in a challenging cannabis matrix. Matrix suppression was significantly reduced by dSPE cleanup for many of the pesticides; thereby improving the data quality. This method is capable of meeting the MRLs for Oregon’s pesticide list in cannabis matrix.
720006373, October 2018