This application note focuses on reversed-phase (RP) column selection considerations for peptide mapping applied to the routine characterization and analysis of biotherapeutic proteins.
In this study, ten distinctly different RP column types, representing variations in ligand characteristics, base particle composition, and particle size, were evaluated. Among these, we have identified four Waters RP Particle Technologies with notable applicability to peptide mapping. Unlike other options, these column technologies provide both high peak capacity separations and sizeable differences in retentivity and selectivity, which can in turn be used to expedite the development of RP peptide mapping separations.
The peptide mapping of proteins is a fundamental tool that has been applied to both the proteomics-based discovery of new biotherapeutic proteins and for monitoring the modification and degradation of those proteins as they are developed and commercialized.1 When developing a reproducible and informative peptide map, the enzymatic digestion protocol and the separation of the resultant peptides need to be optimized. In this note, we will focus on reversed-phase (RP) column selection considerations for peptide mapping applied to the routine characterization and analysis of biotherapeutic proteins. These analyses that are based on either optical (UV absorbance) detection, mass spectrometric (LC-MS) detection, or both. Many of these column selection considerations can also be directly applied to the selection of columns for LC-MS based proteomics studies and synthetic peptide purity analyses.
In this study, ten distinctly different RP column types, representing variations in ligand characteristics, base particle composition, and particle size, were evaluated using acetonitrile gradients and either 0.1% trifluoroacetic acid (TFA) or 0.1% formic acid (FA) ion-pairing reagents. Separations of both a peptide standard mix and the tryptic digest of a reference monoclonal antibody were used for these comparisons. The performance metrics for this evaluation included both peak capacity and peptide retention. In addition, several of the selectivity differences observed between selected columns are also highlighted.
MassPREP Peptide Mixture Sample was reconstituted in 0.1% formic acid to a concentration of ~15 µg/mL per peptide. National Institute of Standards and Technology (NIST) monoclonal antibody (NISTmAb) reference material, RM 8671 was reduced, alkylated (iodoacetamide), and then digested with trypsin. The sample was acidified prior to analysis with 1:9 ratio of 1% formic acid. The final concentration of injected mAb sample was ~0.1 mg/mL.
|
LC system: |
ACQUITY UPLC H-Class Bio |
|
Detection: |
ACQUITY UPLC TUV Detector with 5 mm titanium flow cell |
|
Wavelength: |
214 nm |
|
Columns: |
ACQUITY UPLC Peptide BEH C18, 130Å, 1.7 µm, 2.1 x 150 mm ACQUITY UPLC Peptide CSH C18, 130Å, 1.7 µm, 2.1 x 150 mm XSelect Peptide CSH C18 XP, 130Å, 2.5 µm, 2.1 x 150 mm ACQUITY UPLC Peptide BEH C18, 300Å, 1.7 µm, 2.1 x 150 mm ACQUITY UPLC Peptide HSS T3, 100Å, 1.8 µm, 2.1 x 150 mm ACQUITY UPLC CSH Phenyl-Hexyl, 130Å, 1.7 µm, 2.1 x 150 mm CORTECS C18, 90Å, 2.7 µm, 2.1 x 150 mm CORTECS UPLC C18+, 90Å, 1.6 µm, 2.1 x 150 mm CORTECS C18+, 90Å, 2.7 µm, 2.1 x 150 mm CORTECS UPLC T3, 120Å, 1.6 µm, 2.1 x 150 mm |
|
Column temp.: |
60 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
15 µL for MassPREP Peptide Mixture and 50 µL for NIST mAb digest |
|
Flow rate: |
0.2 mL/min |
|
Mobile phases: |
A: 0.1% (v/v) trifluoroacetic acid (TFA) or 0.1% (v/v) formic acid (FA) in water B: 0.1% (v/v) trifluoroacetic acid (TFA) or 0.1% (v/v) formic acid (FA) in acetonitrile(Peptide standard) |
|
Time (min) |
%A |
%B |
|---|---|---|
|
Initial |
99.5 |
0.5 |
|
2.0 |
99.5 |
0.5 |
|
32.0 |
50.0 |
50.0 |
|
35.0 |
5.0 |
95.0 |
|
36.0 |
5.0 |
95.0 |
|
38.0 |
99.5 |
0.5 |
|
50.0 |
99.5 |
0.5 |
|
Time (min) |
%A |
%B |
|---|---|---|
|
Initial |
99.5 |
0.5 |
|
2.0 |
99.5 |
0.5 |
|
62.0 |
50.0 |
50.0 |
|
65.0 |
5.0 |
95.0 |
|
66.0 |
5.0 |
95.0 |
|
68.0 |
99.5 |
0.5 |
|
80.0 |
99.5 |
0.5 |
Peptide mapping separations can be exceedingly complex. As an example, the tryptic peptide map of the NIST monoclonal antibody reference material (NISTmAb) is comprised of over 50 predicted peptides of three amino acid residues or more. In addition, there are numerous , lower abundance modified or degraded peptides, incompletely and non-specifically digested peptides, and autolytic trypsin peptides. This can result in a separation with well over a hundred components many of which represent critical attributes of the protein structure and must be detected at low abundances in the presence of closely eluting peptides at nearly two orders of magnitude higher mass loads. Also problematic in these separations are the minimal differences in molecular structure that can exist between variants of a given peptide, such as the deamidation of a single amino acid residue in a peptide with more than 20 residues.2
Given these challenges, a column that provides the narrowest chromatographic peaks for the peptides separated over the length of the gradient elution provides the greatest probability of success in these complex separations. This performance characteristic for a separation is referred to as “peak capacity” (PC) which is a dimensionless value determined by dividing the average peak width of the separation by the length of the gradient, in a general sense PC can be thought of as being proportional to the average resolution of the separation.3,4 In this study 4σ peak capacities were determined based on the following equation:
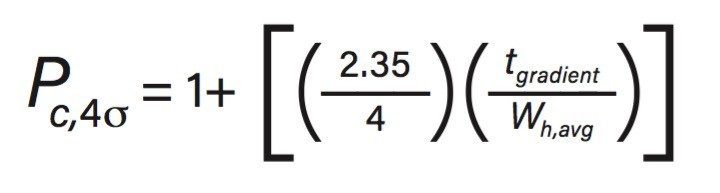
Where tgradient is the time of the gradient and wh,avg is the average peak width at half-height.
PC values were determined for a set of columns (Table 1) having the same 2.1 mm I.D. and 150 mm length.
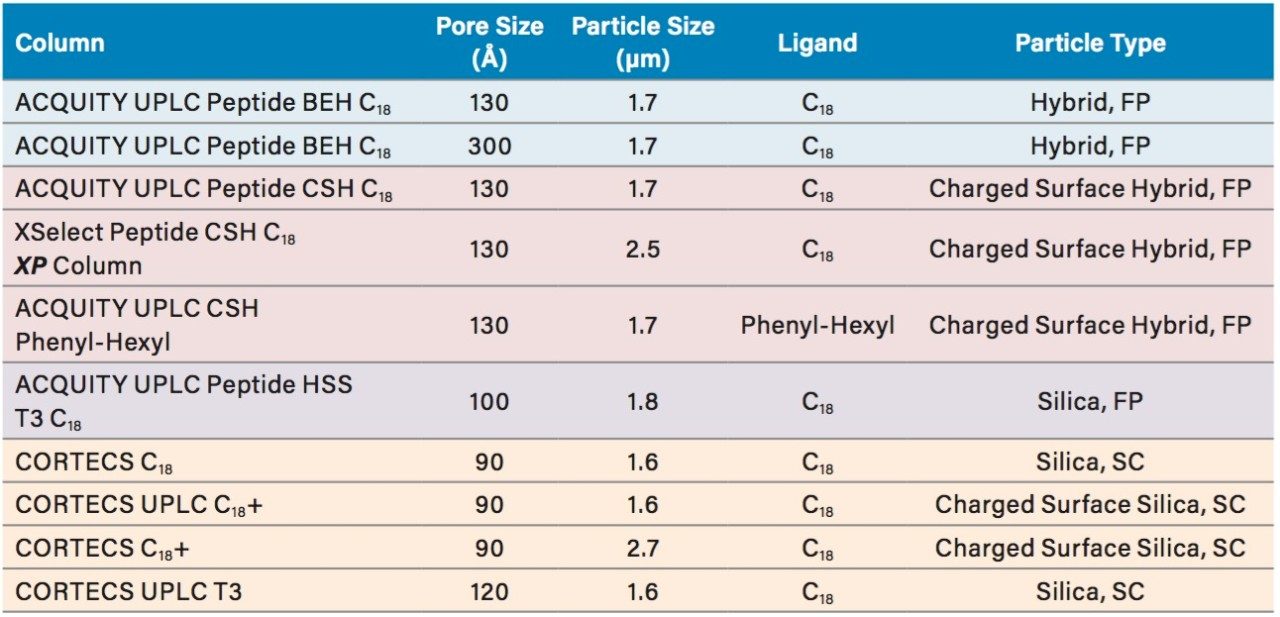
Peak capacity values were determined for all of the columns evaluated using a mixture of peptide standards (MassPREP Peptide Mixture) which was comprised of the components shown in Table 2. This mixture contains peptides of varied sizes and charges. The use of peptide standards facilitated the determination of PC by providing well resolved and predominantly pure peptides for which suitability parameters such as peak width can be more readily determined using UV absorbance data. Further assessments of the performance of the columns were also carried out by visually evaluating the tryptic digest of the NIST reference mAb (IgG1). Selected columns were also more quantitatively interrogated by determining the PC for the extracted ion mass chromatograms (XIC) of selected peptides (Table 3) from the tryptic digest of the NIST reference mAb. These peptides represent an even broader range of molecular weights than the peptides present in the MassPREP Peptide Mixture.
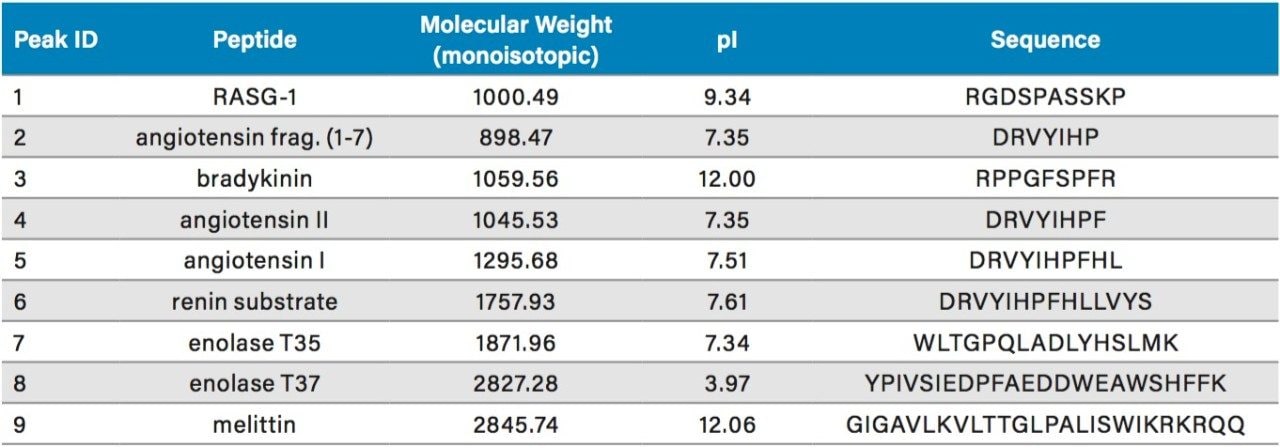
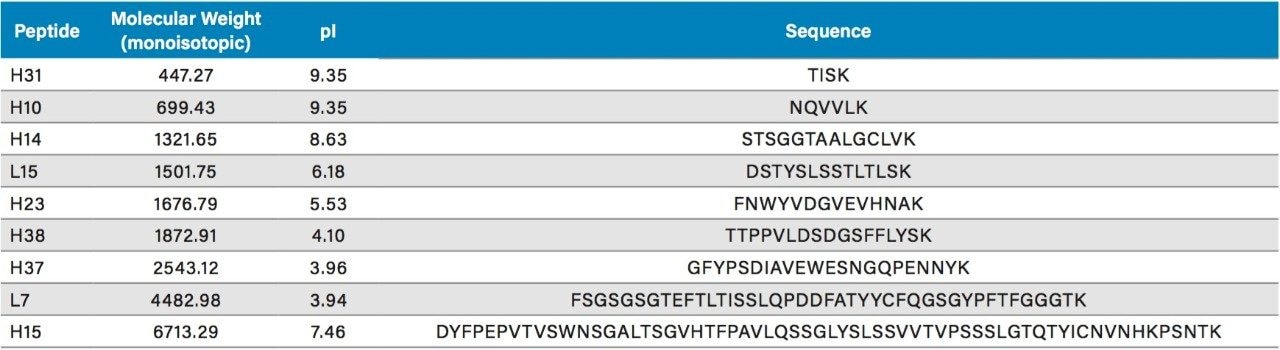
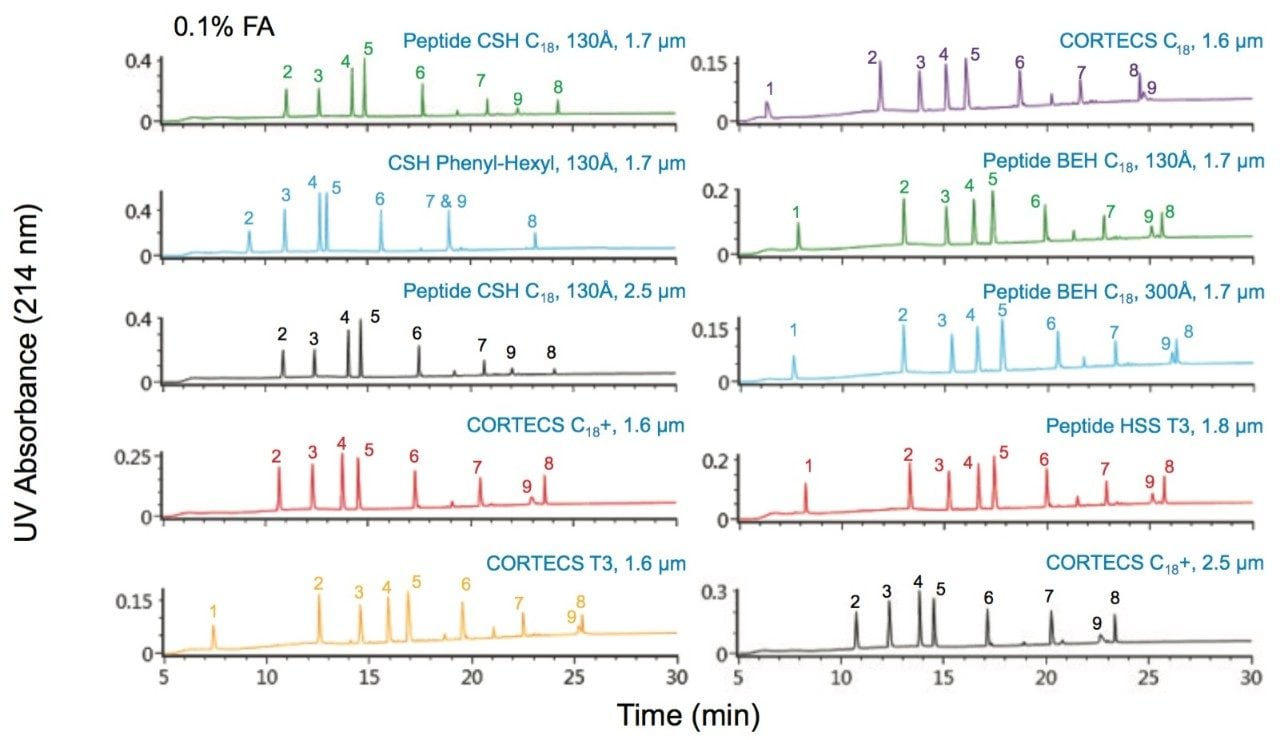
Comparisons of the chromatograms and PC values obtained for the ten columns for both 0.1% TFA or 0.1% FA-based separations of the MassPREP Peptide Mixture are shown in Figures 1 through 4. TFA and FA are the predominant ion-pairing reagents used in peptide mapping experiments. The advantages of TFA in terms of chromatographic performance (PC, retention, and baseline noise) and the advantages of FA with regards to MS sensitivity have been well documented, as has the capability of chromatographic phases using BEH (ethylene bridged hybrid) particles with a positively charged surface chemistry, which Waters refers to as a CSH (charged surface hybrid) particle, to provide high PC separations in FA and mixed FA plus TFA mobile phases.4,5
All of the columns evaluated provide high PC peptide separations in both TFA and FA containing mobile phases as shown by the charts in Figures 2 and 4. However, in both mobile phases the Peptide CSH C18, 130Å columns in 1.7 µm and 2.5 µm particle sizes, and the CSH Phenyl-Hexyl, 130Å, 1.7 µm particle size columns provide the highest average PC. This performance advantage is significantly more differentiated in the FA-containing mobile phase as compared to the TFA containing mobile phase.
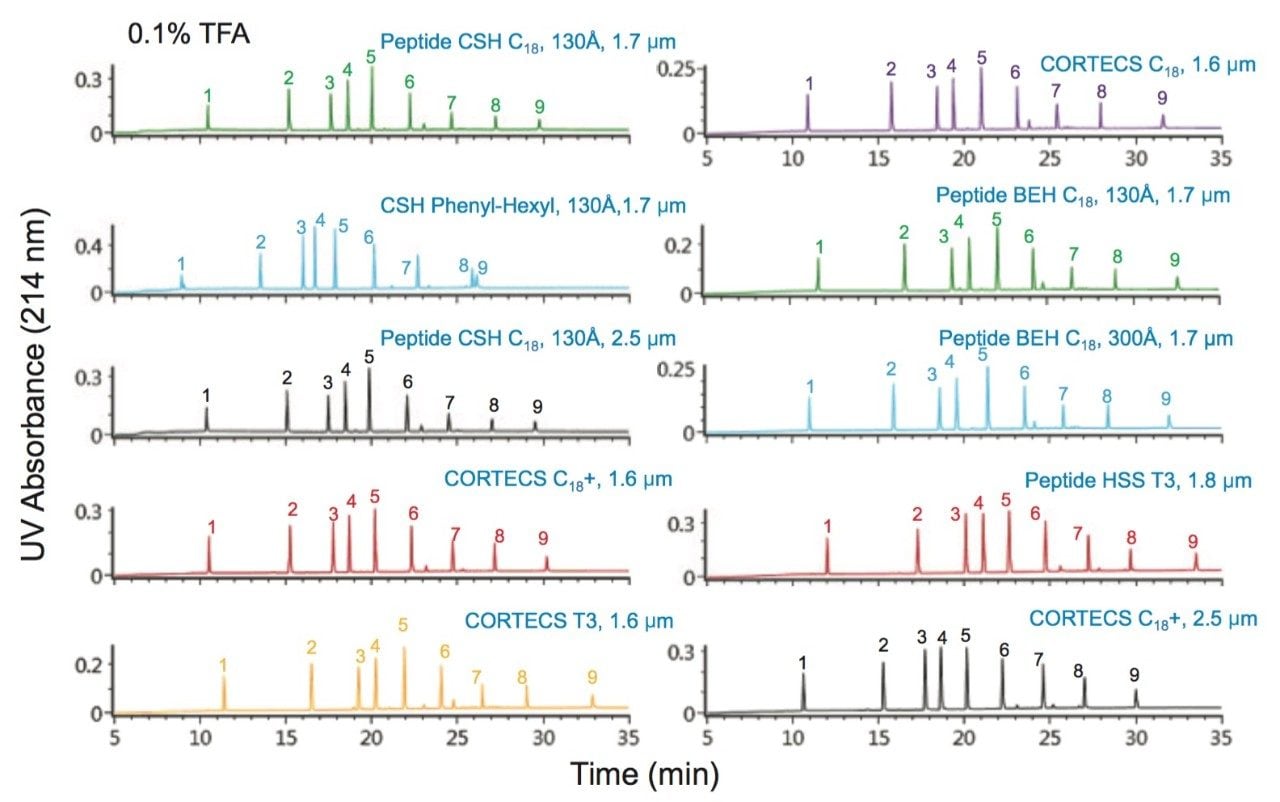
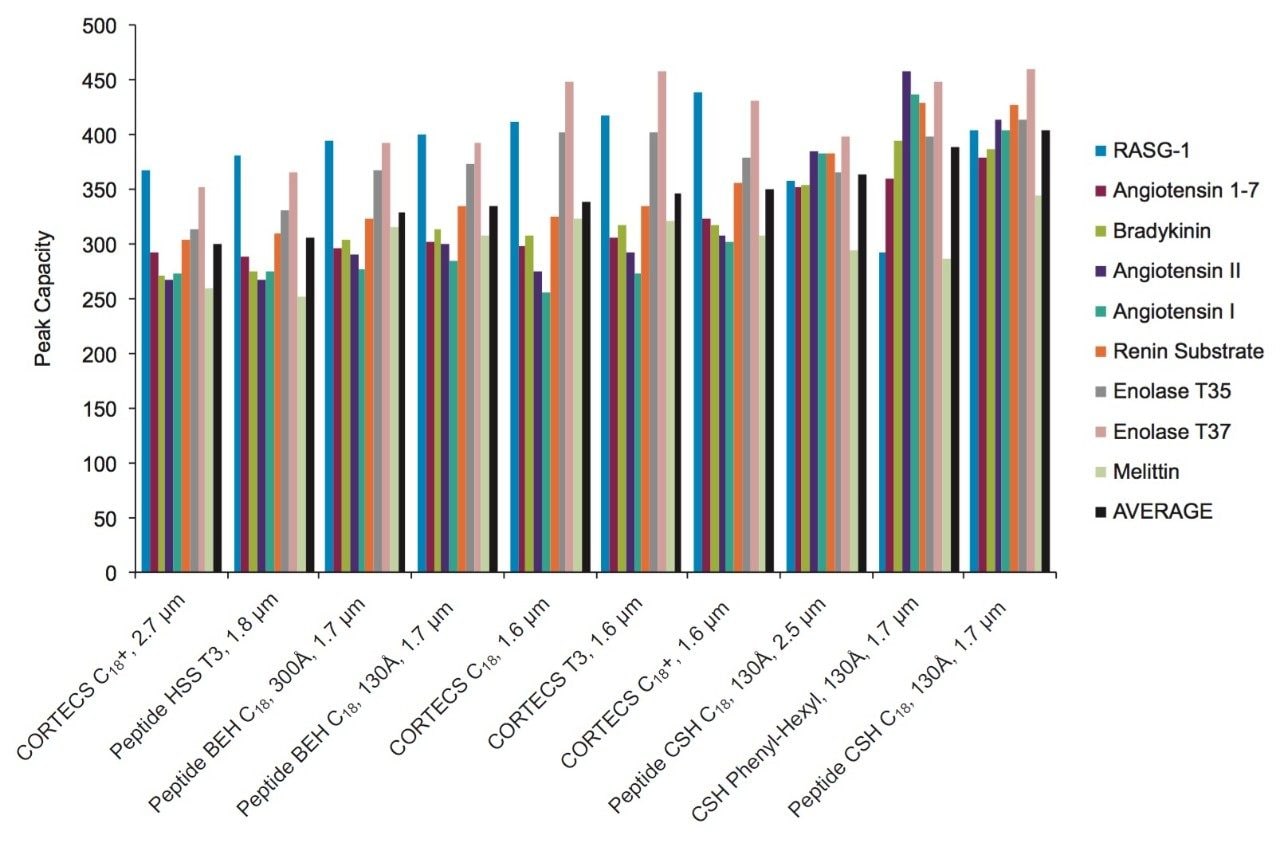
The PC values for individual peptides varied from column to column. As a clear example, the PC results for the peptide melittin with a FA mobile phase (Figure 3) is significantly higher for the Peptide BEH C18, 300Å and Peptide BEH C18, 130Å columns than is for the Peptide CSH C18, 130Å, 1.7 µm column. However, the PC for the other seven peptides is significantly higher for the latter. In this case, melittin is a fairly unusual peptide in that it contains a sequence motif of four strongly basic residues (KRKR). It is reasonable to hypothesize that this highly basic set of residues causes there to be significant charge repulsion on the CSH Column such that effective pore size is reduced, restricted diffusion effects are increased, and PC is diminished for what is a fairly large peptide (2854 Da). That slightly higher PC values were observed for melittin and the enolase T37 peptide (2827 Da) on the 300Å pore size BEH column in comparison to the 130Å pore size BEH particle suggest that restricted diffusion effects can in fact be observed for a 3 kDa peptide. We will further examine pore size effects in the NIST mAb peptide mapping results presented later in this application note.
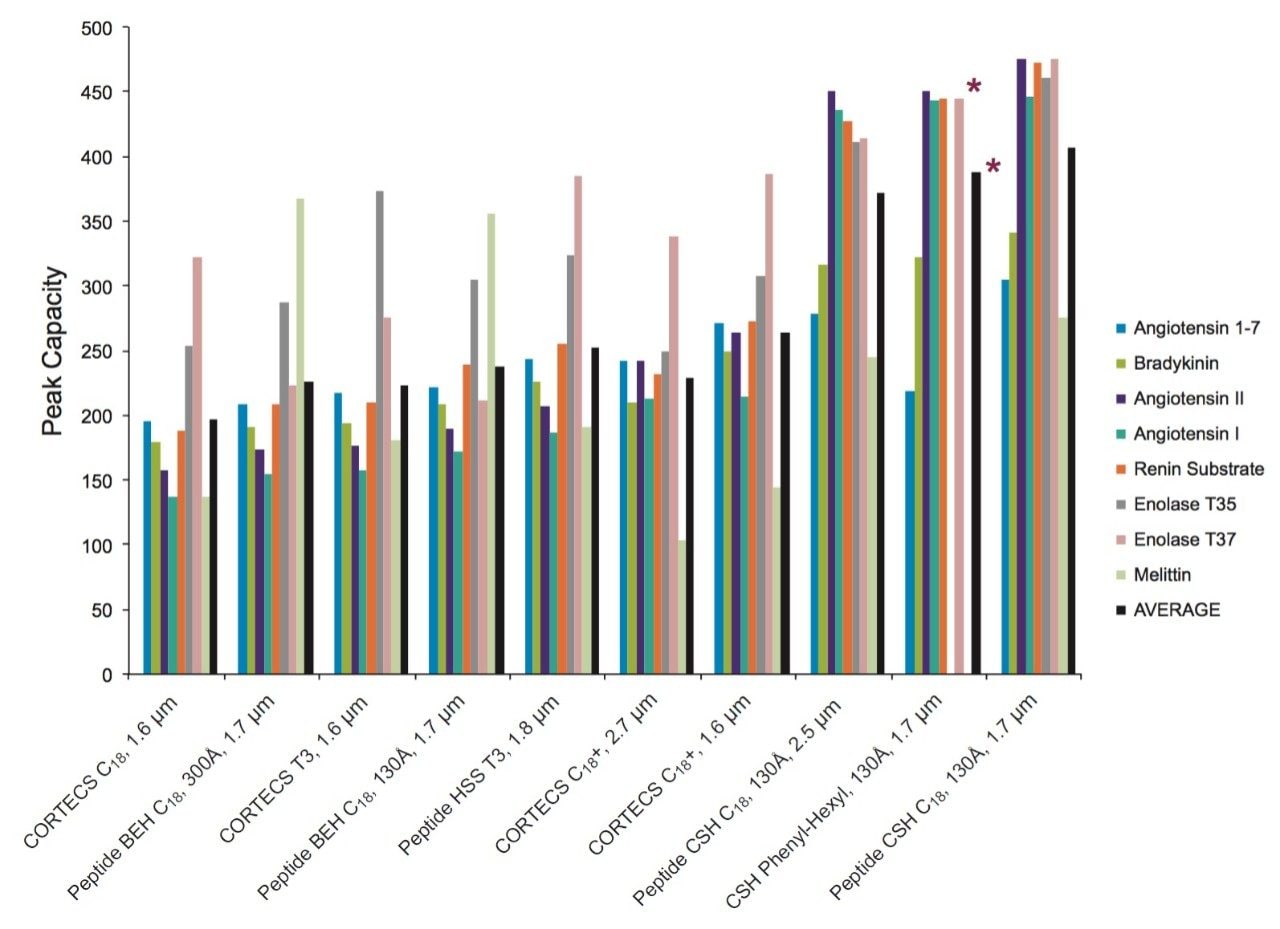
In other instances, these variations in individual peptide PC between different columns can be due to the characteristics of the peptide such as charge, size, and hydrophobicity, in addition to characteristics of the phase such as pore size, particle morphology, and ligand characteristics. As an example, the unusually low PC observed for melittin on the CORTECS T3, 120Å Phase when using a FA mobile phase may be predominantly the result of secondary ionic interactions between the particle surface and this strongly basic (pI =12.1) peptide. It is important to emphasize that these column chemistry differences can also be used to an advantage when developing an optimal peptide separation.
Particle size is also a key component in improving PC, which has an inverse square-root dependence on particle size. Within the set of columns evaluated there are two pairs of columns that are packed with different sized particles but have comparable pore size and surface chemistries. One of the column pairs is the fully-porous Peptide CSH C18, 130Å column in 1.7 µm and 2.5 µm particle sizes, and the other column pair is the solid-core CORTECS C18+, 90Å Column in 1.6 µm and 2.7 µm particle sizes. The individual peptide PC values (Figures 2 and 4) followed similar trends for both column pairs; therefore, we can assess the effect of particle size using the average PC values. For the CSH C18 Columns, increases in average PC of 11% and 16% were observed in TFA and FA when comparing the 1.7 µm to the 2.5 µm particle size columns. Similarly, for the CORTECS C18+ Columns, increases in average PC of 17% and 15% were observed in TFA and FA when comparing the 1.6 µm to the 2.7 µm particle size columns. In both cases, the increase in PC was below the predicted values based on particle size of 21% and 30% for the CSH C18 and the CORTECS C18+ Columns. These discrepancies may be partly attributed to the effect of post-column dispersion volume (tubing and detector flow-cell) on the small volume peaks (~20 µL) generated in this study, variations in peptide behavior and separation conditions, and column packing efficiencies. Nonetheless, these data indicate that smaller particle sizes can provide significant gains in PC. Additionally, it can also be surmised from the comparison of the CSH C18 and CORTECS C18+ chromatograms that while the use of solid-core particles may have some advantages over fully-porous particles, other factors in this case, such as surface chemistry, can have a greater influence on chromatographic performance.
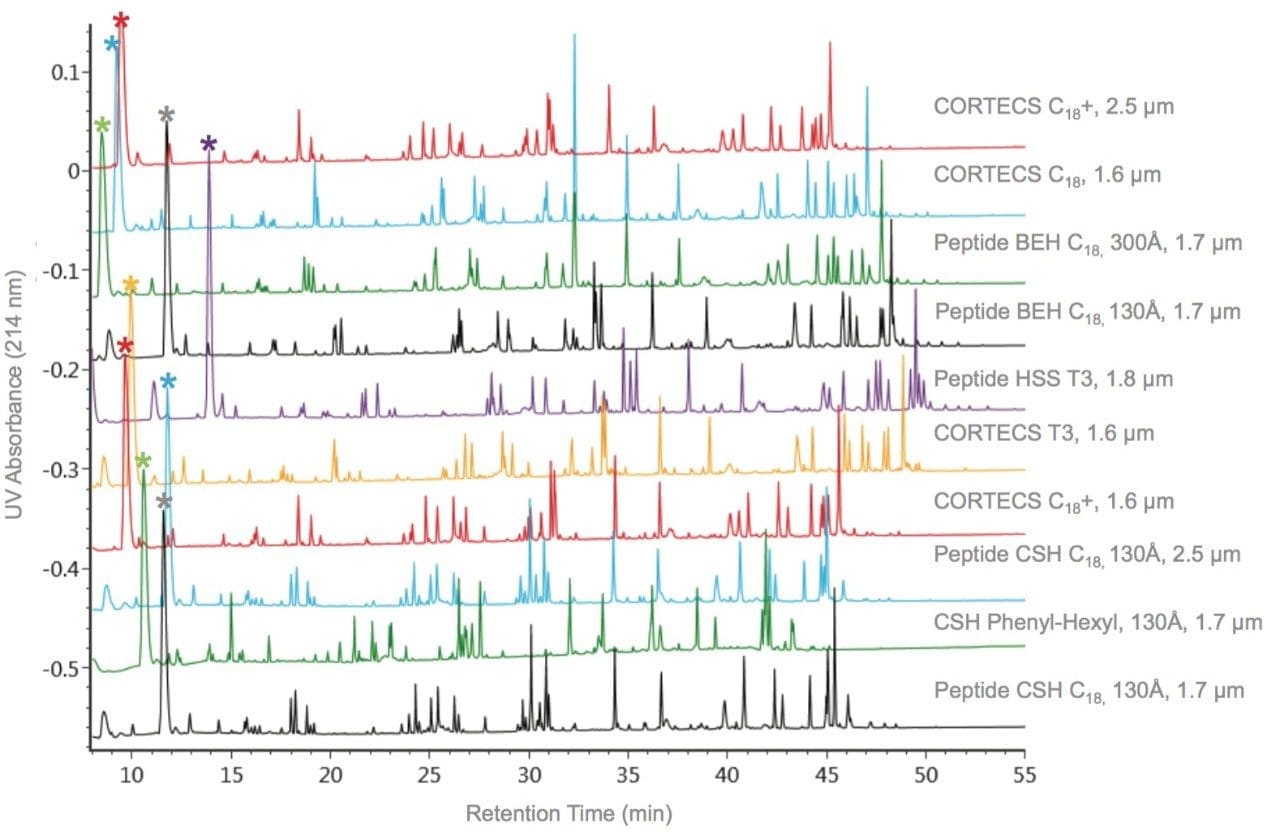
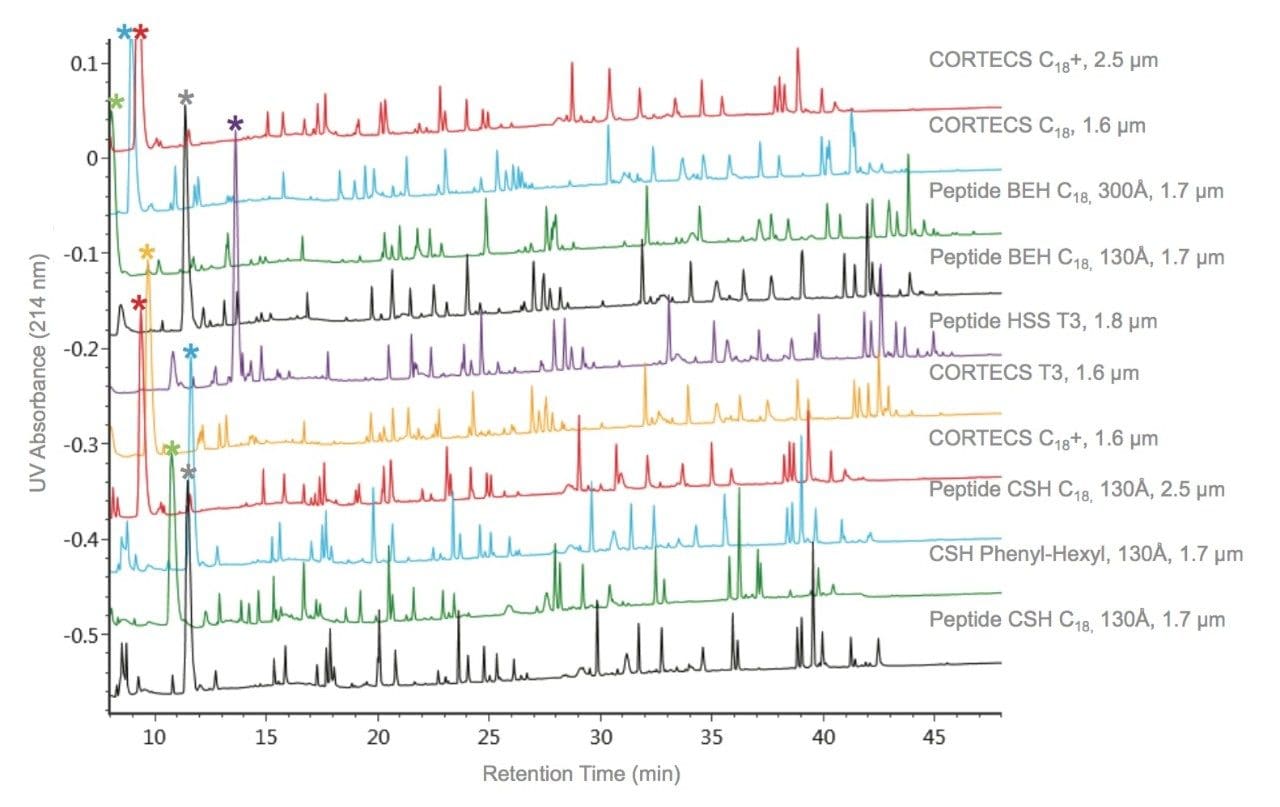
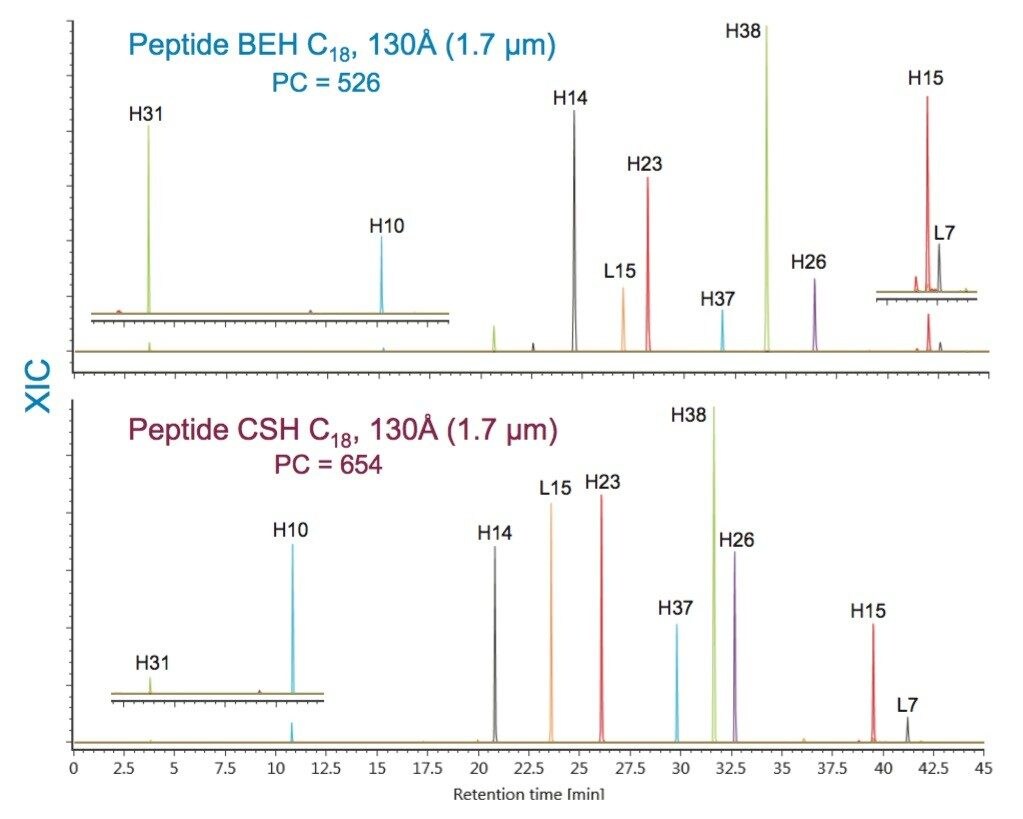
To specifically address the effect that particle surface charge and pore size can have on PC for a peptide map, we will evaluate selected peptides (Table 3) from the trypsin-digested peptide maps of reduced and alkylated NIST mAb. A comparison of the UV absorbance based peptide maps generated for this sample using either TFA or FA ion-pair mobile phases are presented in Figures 5 and 6. Consistent with previous observations for the peptide standard separations (Figures 1–4), all of the columns provide functional peptide separations with adequate peak capacity and a broad range of selectivity in both TFA and FA mobile phases. Nevertheless, due to the complexity of this sample, it is through extracted ion chromatograms (XIC) of selected peptides that the PC values of the various separations can be best elucidated. By using this selective detector, the complications that unresolved peptides can have on PC determinations can be minimized.
Comparisons of the XIC-based PC results in both TFA and FA mobile phases for the Peptide CSH C18, 130Å, 1.7 µm column and Peptide BEH C18, 130Å, 1.7 µm column (Figures 7 and 8) demonstrate the usefulness of the controlled charge applied to the CSH particle surface. These two C18 RP chemistries are comparable in both particle size and pore size, and are based on the same bridged ethyl and silica hybrid particle (BEH). Of note in this comparison is that both columns provide high PC separations for a broad range of peptide sizes and charge. With TFA as an ion pair, the CSH-based column had 10% greater PC in comparison to the BEH-based column; and, with FA as an ion-pair, the improvement in PC for the CSH Column was 20%. The improved loading of peptides on the CSH particle relative to the BEH particle provides for this increased PC.5
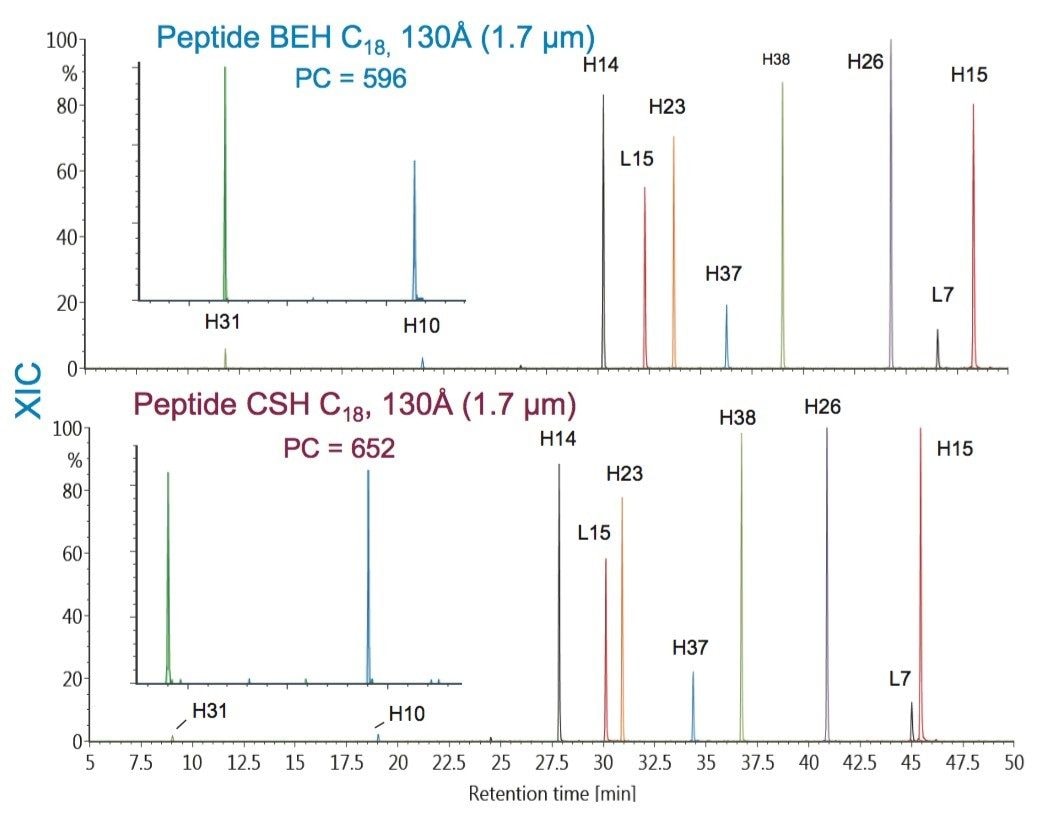
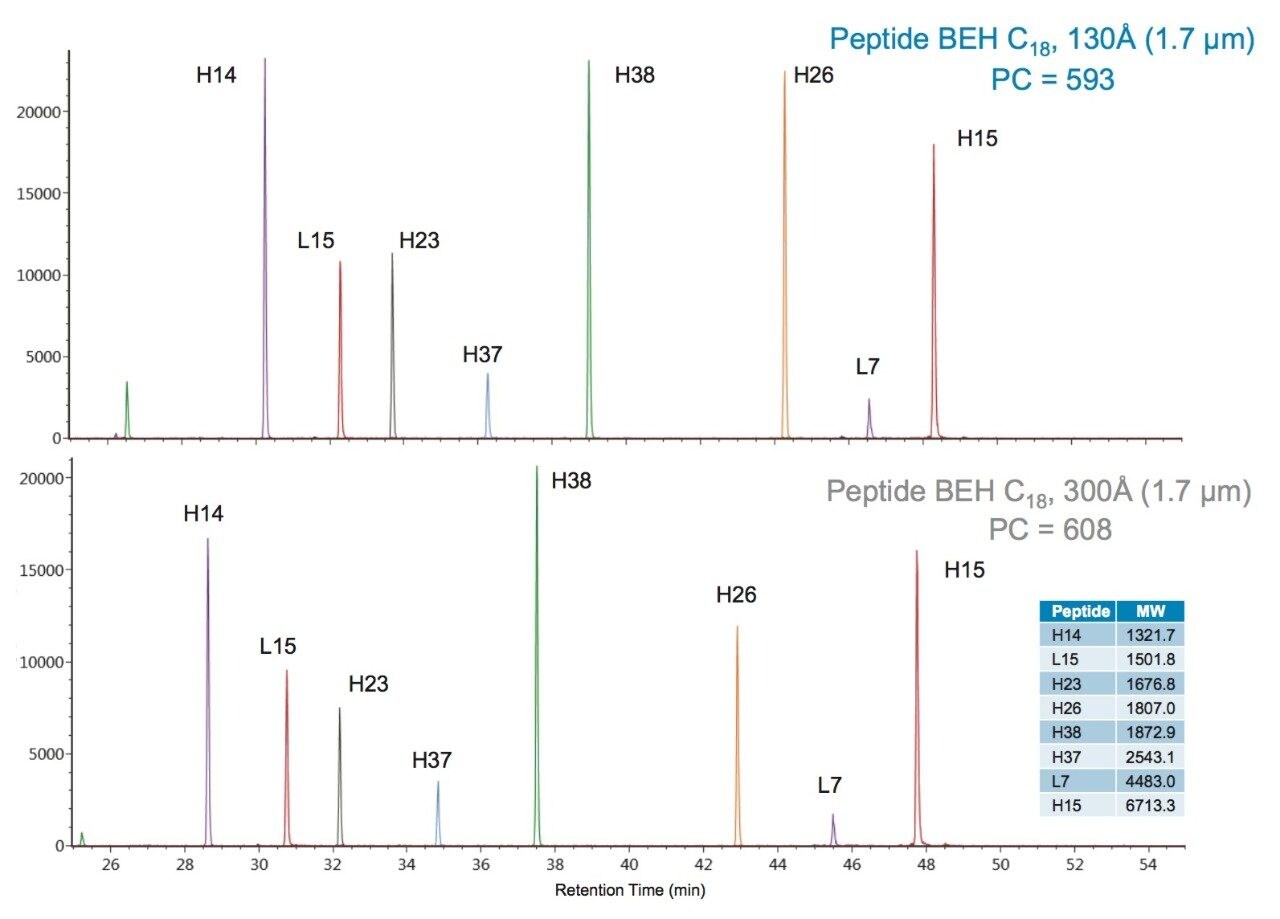
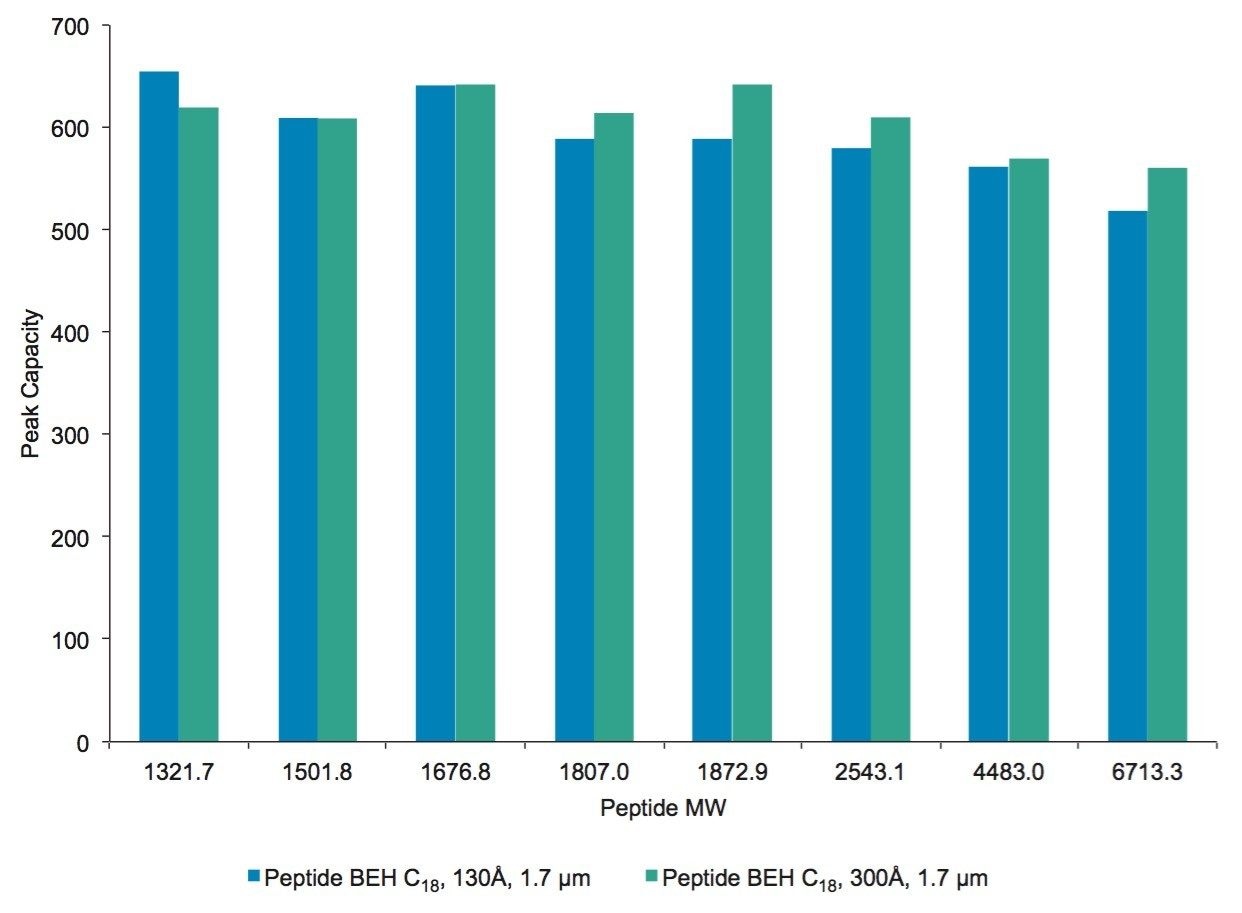
To further evaluate the effect of pore size on peptide separations, XICs for the Peptide BEH C18, 130Å, 1.7 µm and the Peptide BEH C18, 300Å, 1.7 µm columns were compared. An overlay of these XICs is presented in Figure 9, and a chart of the results is presented in Figure 10. In this comparison, peptides with a molecular weight greater than 1000 Da were considered.
In comparing the selected PC results of the two columns (Figure 10), it was observed that peptides with molecular weights greater than 1.8 kDa benefited from the 300Å pore size column with a corresponding modestly higher PC. This improvement in chromatography for the larger mAb-derived peptides may result from a combination of reduced restricted diffusion effects and the increased accessible surface area afforded by the 300Å pore-size phase.
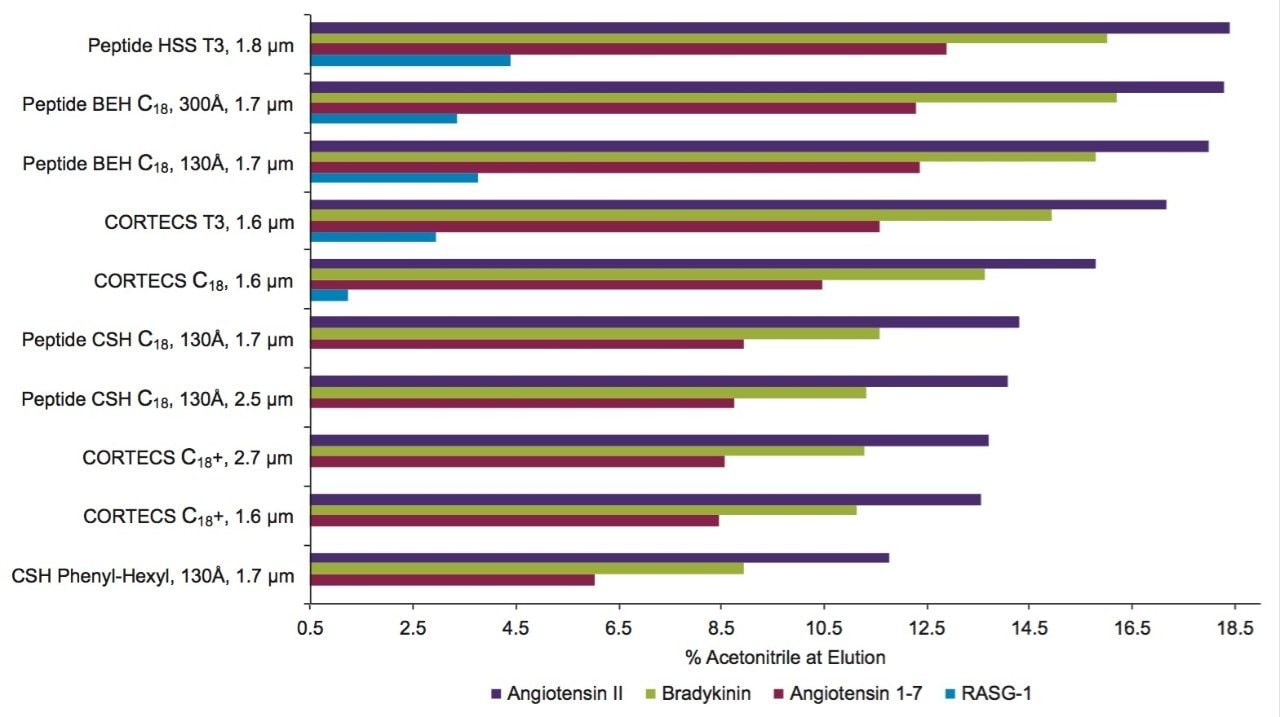
The percentage of the amino acid sequence that can be monitored during the peptide mapping of a therapeutic protein (i.e., coverage) standard is an important method development consideration. It has been proposed in USP General Chapter <1055> “Biotechnology-Derived Articles – Peptide Mapping” that 95% or greater sequence coverage be the target for a validated peptide map. Therefore, a RP column that provides greater peptide retention can be used to advantage if the protein digest results in numerous hydrophilic peptides, particularly if some of those peptides must be quantified in order to monitor a critical quality attribute (CQA).
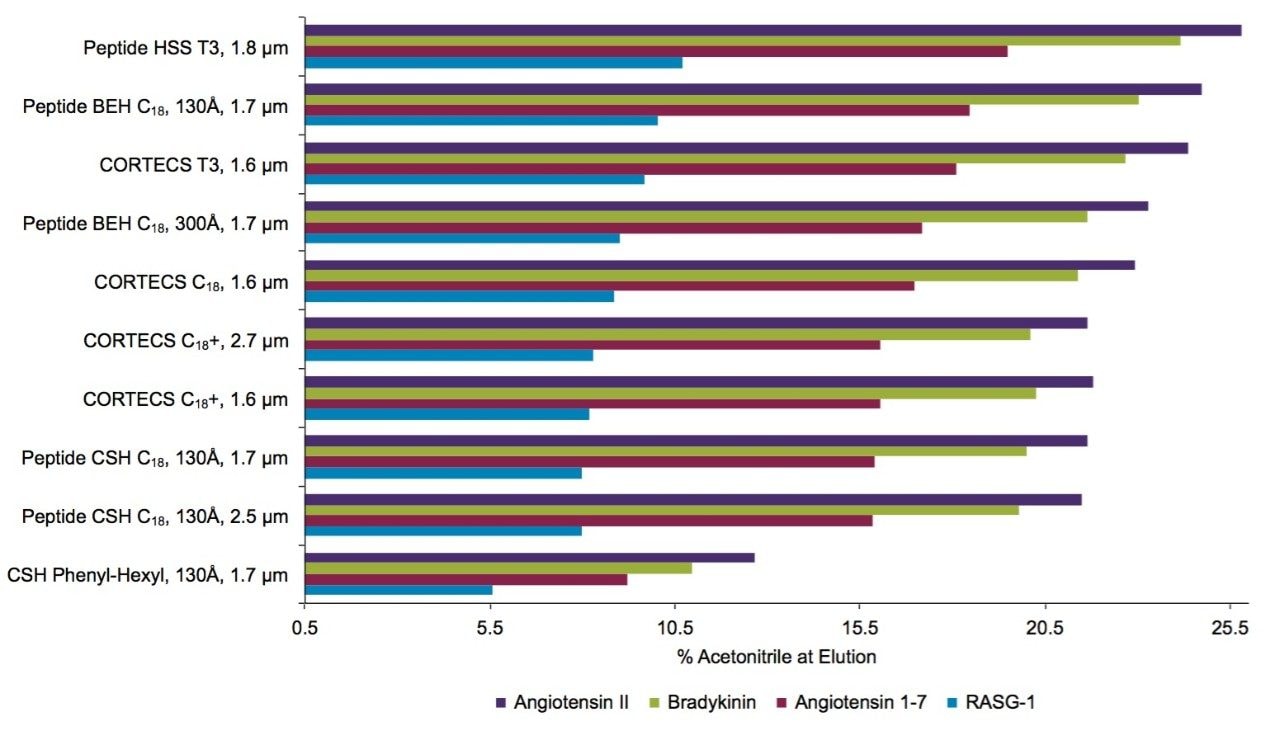
For this evaluation, we calculated the concentration of acetonitrile at which the four least hydrophobic peptides in the MassPREP Peptide Standard Mixture eluted from each column of interest. These data are shown for both 0.1% TFA (Figure 11) and a 0.1% FA (Figure 12) containing mobile phases. The Peptide HSS T3, 100Å column provided the highest retentivity of the columns tested in both TFA and FA. This phase is manufactured with an intermediate ligand density, an approach that has been shown to promote retention.6 All of the columns evaluated provided effective retention in TFA, with some retention of the RASG-1 peptide. However, the retentivity of the CSH Phenyl-Hexyl, 130Å Column was significantly lower than all of the other phases. Retention of the RASG-1 peptide is of note because despite its size (1000.49 Da) it has hydrophobicity indexes (HI, SSRCalc, v Q, ©2006–2015, Manitoba Centre for Proteomics & Systems Biology) of only 4.40 and 0.54 in TFA and FA for the C18, 100Å column. The hydrophobicity index is the predicted concentration of acetonitrile at which the peptide will have a retention factor (k’) of 10.7 In evaluating the retention in FA, the Peptide HSS T3, 100Å is again the most retentive phase, while the Peptide BEH C18, 130Å, Peptide BEH C18, 300Å, and CORTECS T3, 100Å Columns also provide adequate retention for RASG-1 under these conditions. For the other columns tested, RASG-1 is not adequately retained for analysis.
When developing a peptide mapping method for the analysis of a therapeutic protein, the selection of an appropriate reversed-phase column with appropriate retentivity requires one to consider separation conditions (e.g., mobile phase and temperature). However, another important consideration is the nature of the protein digest. In particular, if the proteolytic enzyme selected yields a large number of smaller hydrophilic peptides, the use of a more retentive phase can be advantageous. However, if the proteolytic digest yields, larger more hydrophobic peptides, then using a more retentive phase may not provide a significant benefit
Selectivity differences among different phases can also be used to an advantage to improve a peptide mapping separation method, namely by manipulating the resolution of a critical pair of closely eluting peptides. It is important to note, however, that increasing the selectivity for a given critical pair of peptides in these complex peptide mapping separations may result in a loss of selectivity between other pairs. Selectivity differences can be observed in many of the data presented, and may be the result of differences such as ligand type, ligand density, particle surface charge, and particle pore size. Of these variables, a change in ligand type can have the most significant impact on selectivity. As an example, Figure 1 compares the separation of the MassPREP Peptide Standard Mixture as obtained with several C18 phases. The separation achieved on the CSH Phenyl-Hexyl, 130Å Phase shows a change in elution order between Peak 8 (enolase T37) and Peak 9 (melittin). The greater relative retention of enolase T37 on the CSH Phenyl-Hexyl Phase is likely the result of the large number of aromatic amino acid residues (tyrosine, tryptophan, and phenylalanine). In particular, an amino acid motif of sequential phenylalanines contained in the T37 peptide can form strong π−π interactions with the phenyl ligand of that phase.
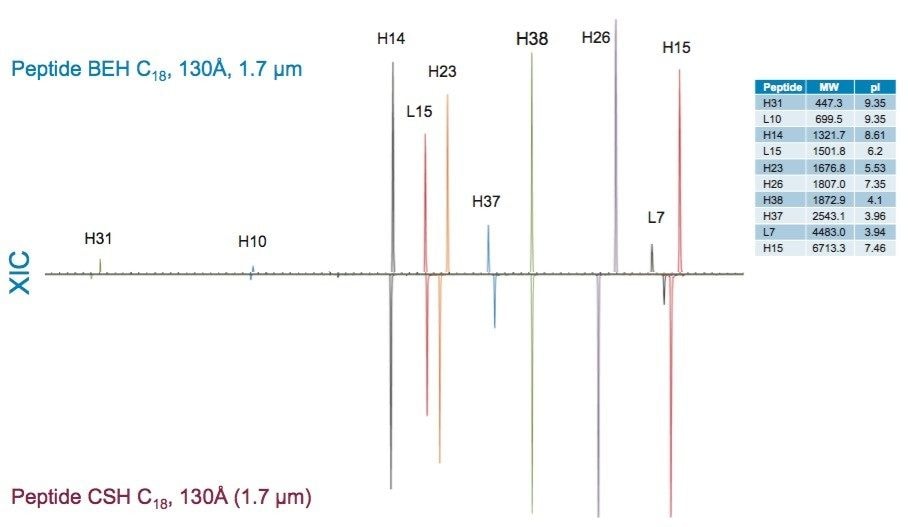
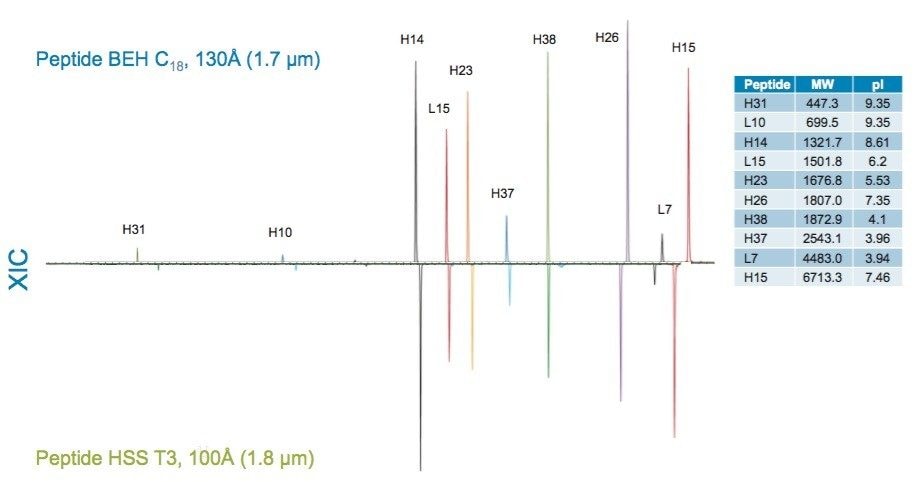
More subtle differences in selectivity are observed for phases with the same ligand; however, changes in ligand density, in addition to the characteristics of the base particle such as surface charge and pore size, can influence selectivity. Examples of these differences can be observed in comparisons of the XIC results for selected peptides from the trypsin-digested peptide maps of reduced and alkylated NIST mAb. For these comparisons, the Peptide BEH C18, 130Å column was compared to the Peptide BEH C18, 300Å, Peptide CSH C18, 130Å, and Peptide HSS T3, 100Å Columns. To better visualize these selectivity differences, the time axes of the chromatograms have been aligned on a selected peptide (H38).
The primary differences between the Peptide BEH C18, 130Å and the Peptide BEH C18, 300Å phases is pore size and surface area (185 m2/g and 90 m2/g). The ligand (C18) and ligand densities (3.1 µmole/m2) are comparable. As a result, as peptide size increases, a selectivity difference that is contributed to by a size exclusion effect is observed. This effect is more pronounced for the two largest peptides, L7 and H15, which are predicted to have significantly restricted pore access with a 130Å pore-size media.
Comparisons of the Peptide BEH C18, 130Å, 1.7 µm separation to the separations obtained for like particle size Peptide CSH C18, 130Å and Peptide HSS T3, 100Å Columns are shown in Figures 14 and 15. The differences in selectivity observed between the Peptide BEH C18, 130Å and Peptide CSH C18, 130Å Columns may be due to several factors. The primary difference between these two particles is the applied positive charge on the surface of the Peptide CSH C18 phase, though there are also slight differences in C18-ligand density with the Peptide CSH C18 phase being approximately 25% lower (2.3 versus 3.1 µmole/m2). Most notably, it is the significant surface charge difference between these two phases that can provide a potentially advantageous selectivity difference in the separation of peptides, particularly those that have substantial differences in net charge.
More subtle differences in selectivity are observed in the comparison of the Peptide BEH C18,130Å and the Peptide HSS T3, 100Å columns. These columns differ significantly in base particle composition (BEH versus silica), ligand density (3.1 and 1.6 µmole/m2), pore size (130Å and 100Å), and surface area (185 m2/g and 230 m2/g). This range of differences makes it difficult to predict peptide selectivity changes, therefore an empirical comparison, like the one shown here, is typically warranted.

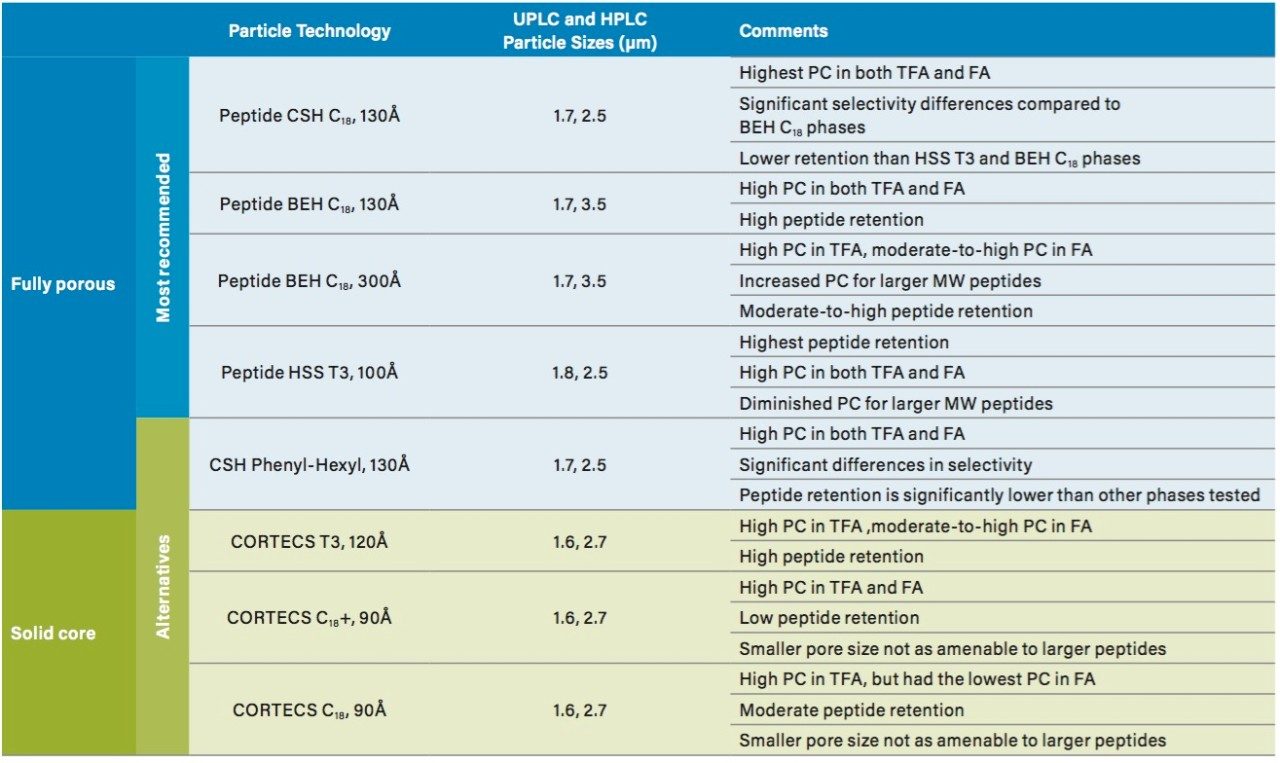
A chromatographic peptide mapping method must provide adequate resolution and recovery of a broad set of peptides if it is to be used in the quality analysis of a biotherapeutic protein. The three principal attributes of a RP column that can be used to advantage for these complex separations are peak capacity, retention, and selectivity. Selecting a column for a peptide mapping separation is decidedly difficult, as there are a multitude of columns provided by Waters and numerous other manufacturers. In addition, one must also take into account the many varied properties of peptides, such as size and charge, and mobile-phase conditions (TFA or FA). In reality, there is no single column that will provide an optimal separation for every peptide pair under any condition. As a result, column screening may be a necessary approach. All of the Waters RP columns listed in Table 4, along with a brief description of their attributes, are potentially capable of producing an effective peptide mapping separation and could be screened during method development. However, screening all of these columns along with selected columns from other manufacturers is not feasible. Therefore, a subset of four of these Waters RP particle technologies that generally provide peak capacity, retention, and selectivity differences to enable the successful development of RP peptide mapping separations have been identified as peptide chemistries (Table 4). These peptide column particles are also quality tested to provide additional assurance of performance reproducibility for peptide separations.
720005924, June 2017