This study uses a multi-omics approach to study zebrafish plasma to provide a greater understanding of the mechanisms involved in heart tissue regeneration following the operation.
Zebrafish (Danio rerio) have long been used as model systems for research involving biomedical and preclinical studies. One particular avenue of research has focused on using the zebrafish model for regenerative medicine investigations.1 Previous studies involving the excision of heart tissue from zebrafish have shown tissue to regenerate in only a few days following amputation. Detailed analysis using genomic and proteomic approaches have identified a number of potential candidates that may be implicated in the regeneration process. This study uses a multi-omics approach to study zebrafish plasma, rather than the excised tissue, to provide a greater understanding of the mechanisms involved in heart tissue regeneration following the operation. Lipidomic and proteomic data have been collected using a label-free LC-HDMSE (LC-DIA-IM-MS) approach, providing qualitative and quantitative information within a single acquisition. This method of data acquisition provides high sensitivity compared with traditional data dependent techniques and is therefore of particular use for situations were sample availability is limited. Interrogation of the data using a combination of pathway tools have shown high density lipid (HDL) transport, platelet activation, signaling, and aggregation pathways to be implicated following post amputation of heart tissue.
Two groups of male zebrafish were used for the study, consisting of sham operated (anaesthetized with no heart tissue excised) and amputation operated (anaesthetized with heart tissue excised). Subsequently, plasma samples (20 µL) were taken three days after anaesthetization/operation. The method by which plasma was collected has been detailed previously.2
For proteomic analyses, 7 µL aliquots of plasma per fish were prepared with 1% RapiGest SF Surfactant prior to reduction, alkylation, and overnight digestion with trypsin.
The remaining plasma was prepared for lipidomic analysis by mixing 1:3 v/v with acetonitrile:isopropanol (50:50) and stored at -20°C for 20 min. Protein precipitation was completed by centrifuging for 10 min. The supernatant was collected and diluted 1:1 with water in preparation for LC-MS analysis.
|
LC system: |
ACQUITY UPLC M-Class |
|
Column(s): |
5 μm Symmetry C18 180 μm x 20 mm 2G trap 1.8 μm HSS T3 C18 75 μm x 150 mm NanoEase analytical |
|
Column temp.: |
35 °C |
|
Flow rate: |
300 nL/min |
|
Mobile phase: |
Water (0.1% formic acid) (A) and acetonitrile (0.1% formic acid) (B) |
|
Gradient: |
5% to 40% B in 90 min |
|
Injection volume: |
1 μL |
|
LC system: |
ACQUITY UPLC I-Class |
|
Column: |
1.7 μm CSH C18 2.1 mm x 100 mm reversed phase analytical |
|
Column temp.: |
55 °C |
|
Flow rate: |
400 μL/min |
|
Mobile phase: |
10 mM ammonium formate with 0.1% formic acid/acetonitrile:water (60:40) (A) and 10 mM ammonium formate with 0.1% formic acid/isopropanol:acetonitrile (90:10) (B) |
|
Gradient: |
40% to 99% B in 18 min |
|
Injection volume: |
1 μL |
|
MS system: |
SYNAPT G2-Si |
|
Ionization mode: |
ESI (+) at 3.2 kV |
|
Cone voltage: |
30 V |
|
Acquisition mode: |
HDMSE 50 m/z to 2000 m/z both functions (low and elevated energy) |
|
Acquisition rate: |
Low and elevated energy functions at 0.5s |
|
Collision energy: |
5 eV (low energy function) and from 19 eV to 45 eV (elevated energy function) |
|
Resolution: |
25,000 FWHM |
|
IMS T-wave velocity: |
700 m/s |
|
IMS T-Wave pulse height: |
40 V |
|
MS system: |
SYNAPT G2-S |
|
Ionization mode: |
ESI (+) at 2.0 kV; ESI (-) at 1.0 kV |
|
Cone voltage: |
30 V |
|
Acquisition mode: |
MSE 100 m/z to 2000 m/z both functions (low and elevated energy) |
|
Acquisition rate: |
Low and elevated energy functions at 0.2 s |
|
Collision energy: |
4 eV (low energy function) and from 15 eV to 40 eV (elevated energy function) |
|
Resolution: |
10,000 FWHM |
Progenesis QI
Progenesis QI for Proteomics
EZInfo
Spotfire
Reactome
The LC-MS peptide and lipid data were processed and searched using Progenesis QI and Progenesis QI for Proteomics. Progenesis enabled normalized label-free quantification to be achieved along with peptide/compound identifications. Additional multi-variate statistical analysis of the data was performed with EZInfo. Curated datasets were submitted for pathway analysis using Reactome4,5 and PANTHER Classification System.6
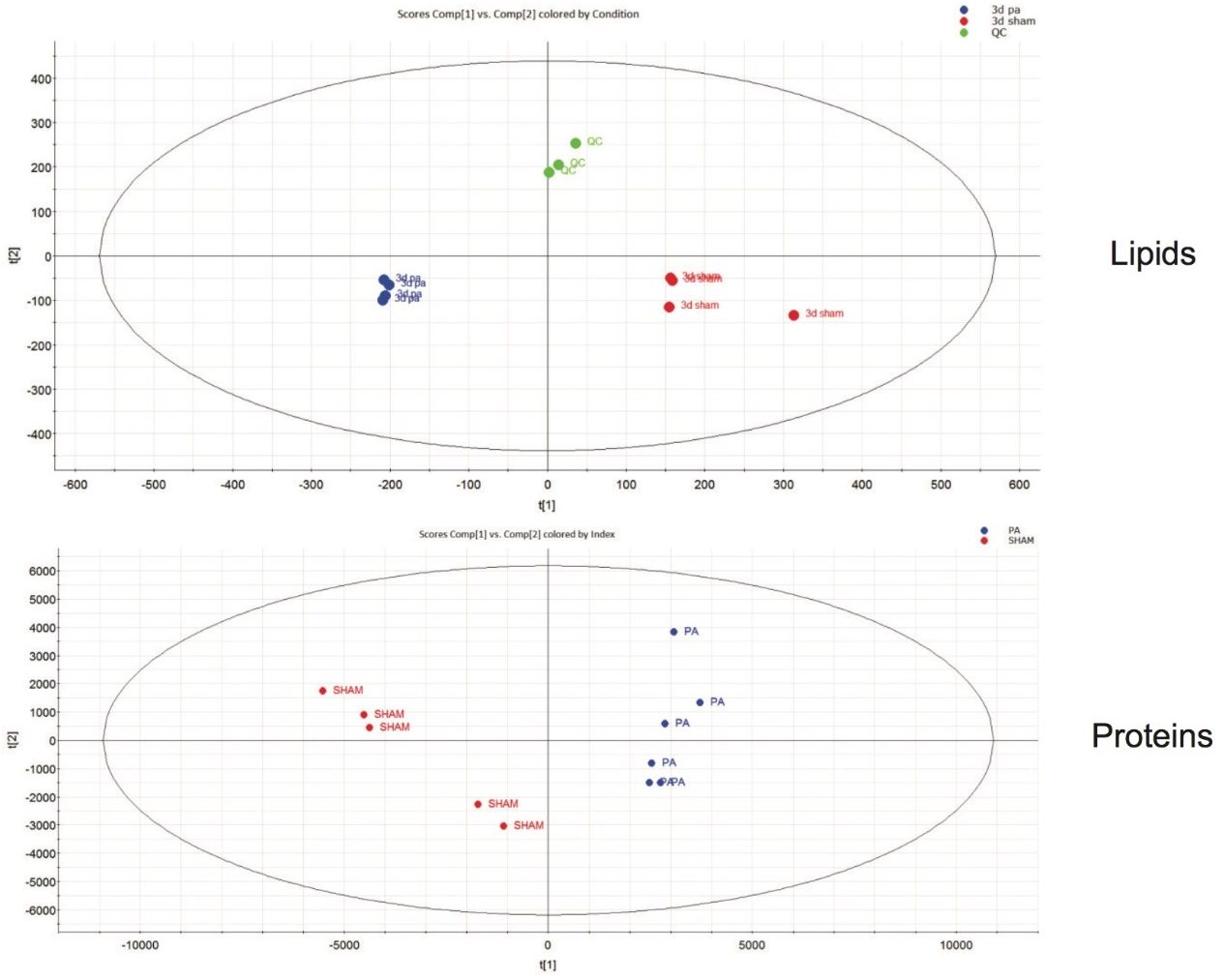
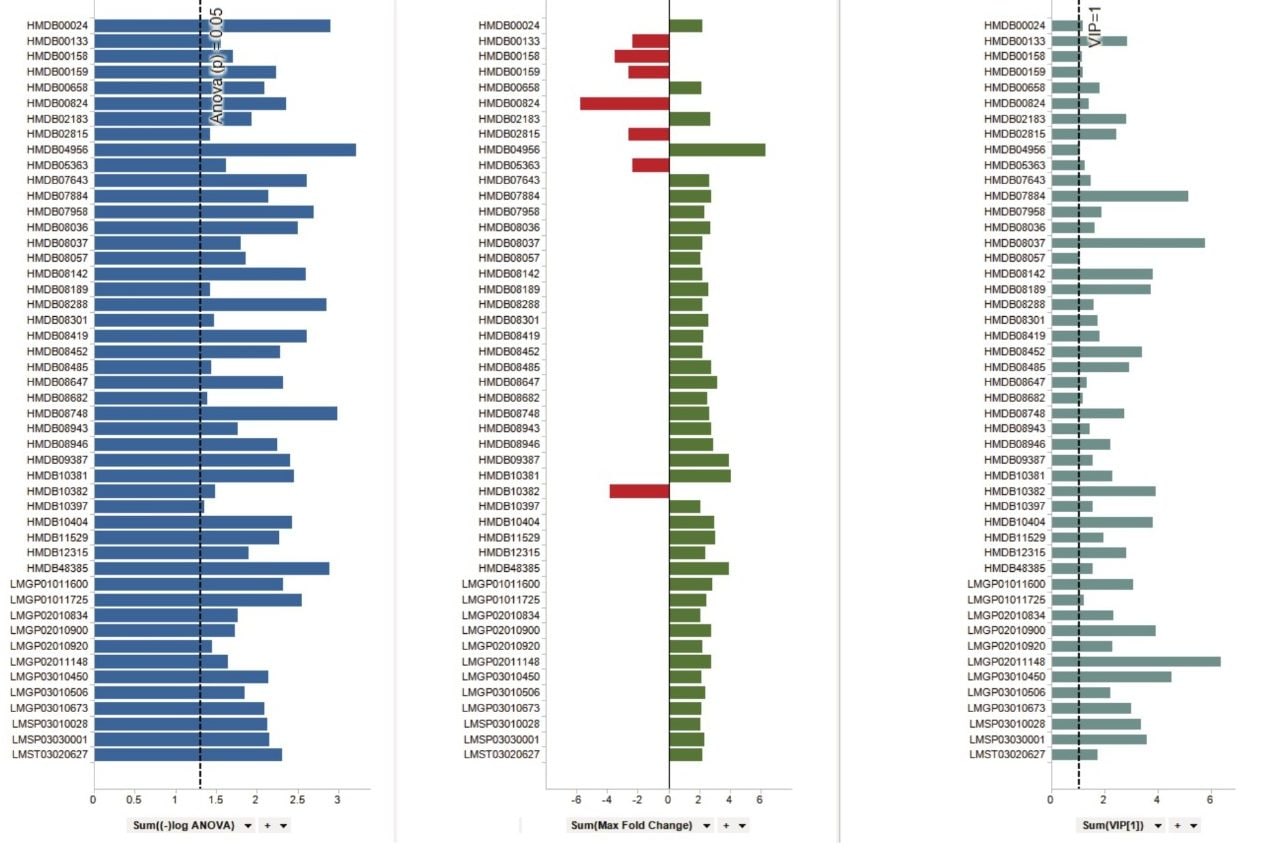
Statistical analysis of the LC-MS data by means of unsupervised principle component analysis (PCA) for the protein and lipid extracts show clear separation (PC1 and PC2 components) between sham operated and amputated groups (Figure 1). A variety of column chemistries were employed to ensure extensive coverage of the lipidome profile and subsequent processing, using Progenesis QI, resulted in excess of 30,000 features being peak picked. An S-plot constructed from the unsupervised PCA scores with additional filtering of the data ensured only features with a maximum fold change >2, ANOVA (p) ≤0.05 and variable importance in the projection (VIP) values >1 were included for further analysis and database searching using a combination of the Human Metabolite Database (HMDB) and LipidMaps. Example tentative identifications curated against these criteria are shown in Figure 2.
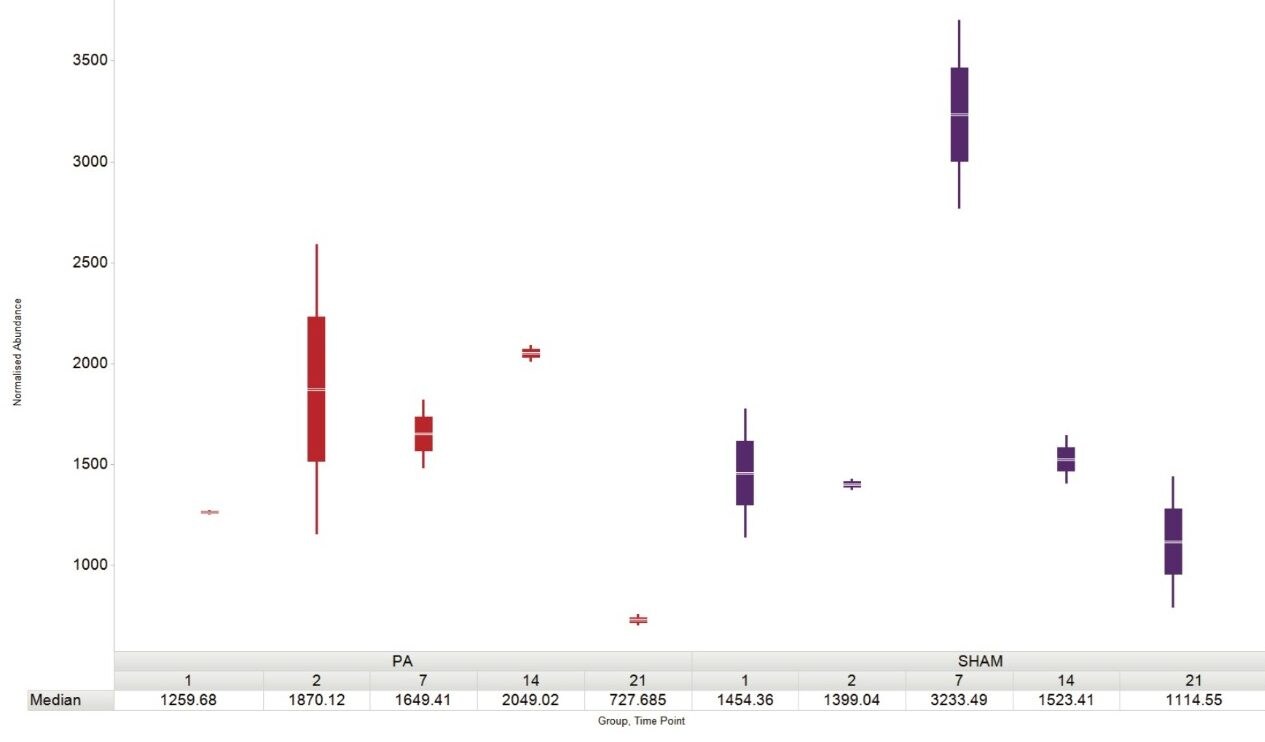
The lipid classes identified in the plasma sample extracts which showed differential expression included phosphatidylcholines (PC), triglycerides (TG), lysophosphotidylcholines (LPC), ceramides, diglycerides (DG), phosphatidylserines (PS), and phosphatidylethanolamines (PE). Zebrafish heart tissue is known to fully regenerate within a monthly timeframe. Amputated subjects showed varying abundance plasma profiles for a number of identified lipids over the course of tissue regeneration when compared with their sham counterparts. Figure 3 provides a representative lipid example monitored over a 21 day period.
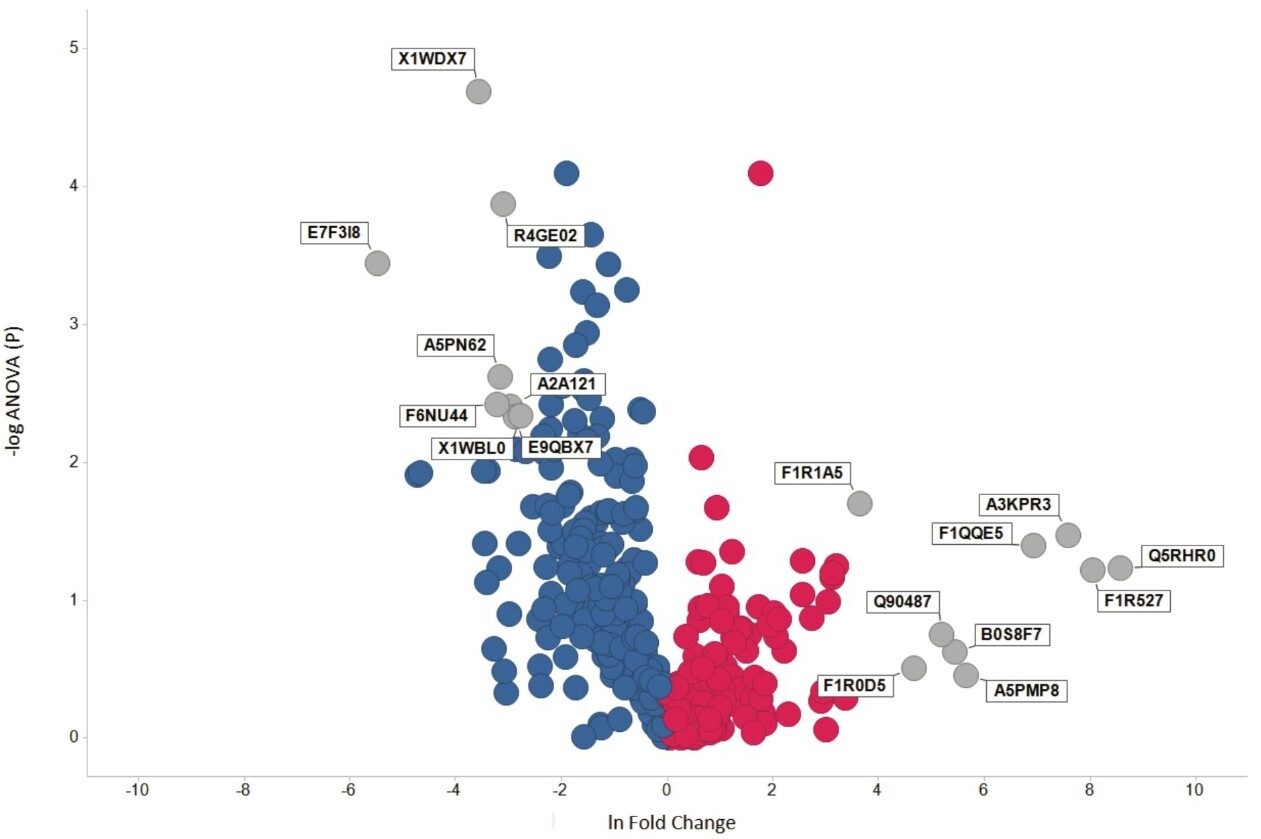
Over 440 proteins were identified and quantified based on a minimum of two peptides per protein and 1% false discovery rate (protein level). A volcano plot corresponding to the protein dataset shows individual identifications which represent changes in expression with statistical significance for amputated operated Zebrafish (Figure 4). Further filtering (ANOVA (p) ≤0.05; fold change >2) resulted in 18% of protein identifications showing significant expression.
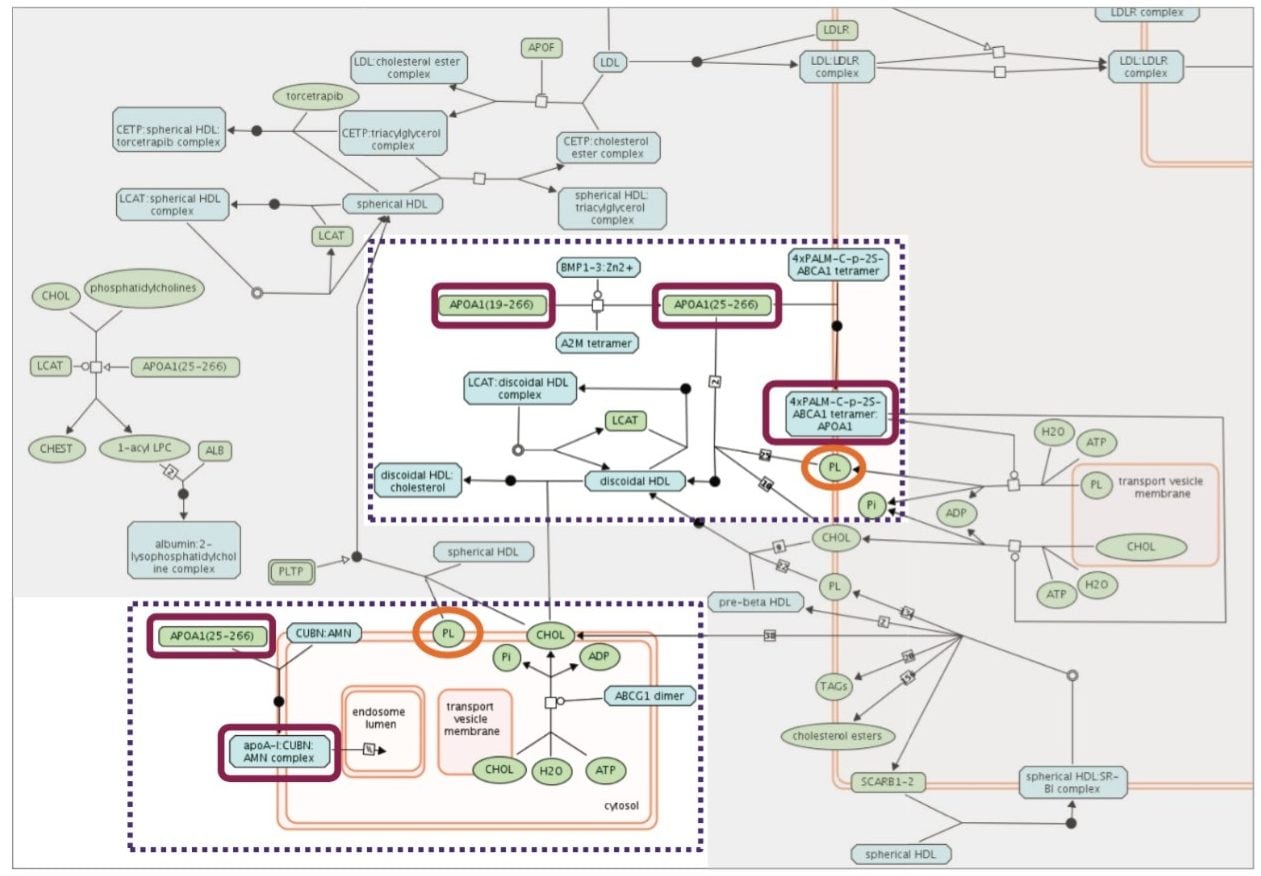
Statistically significant proteins and lipids exhibiting regulation probability values, which were identified in both sham and operated groups were interrogated using a combination of the PANTHER Classification System and Reactome. A variety of biological functions were matched to the data, including metabolic, cellular, regulation, and development processes. These functions were further supplemented with highly correlating pathways such as HDL transport/lipoprotein metabolism, platelet activation, and signaling. Figure 5 shows the identified protein and lipid entries which map to the HDL mediated lipid transport pathway. ATP binding cassette transporter A1 (ABCA1) has been shown to be a key component in providing cardioprotective, anti-inflammatory signaling through interaction with apolipoprotein A (apoA-I). Cascade effects are then initiated allowing cholesterol and phospholipids to be transported to apoA-I thereby generating precursors for HDL particles.7
720006008, May 2017