
This is an Application Brief and does not contain a detailed Experimental section.
This technology brief identifies and confirms the presence of sibutramine in finished herbal natural products. The TLC plate is interfaced with the Waters ACQUITY QDa Mass Detector where target zones are extracted to confirm the presence of sibutramine by mass detection.
The analysis of herbal natural products for undeclared synthetic drugs is becoming more important as adulteration of pharmaceutical drugs is detected. The HPTLC-MS System is able to separate and then confirm the presence of sibutramine in herbal natural products.
Diet foods, such as slimming tea and coffee or dietary food supplements, are widely marketed. Numerous preparations are available to consumers through several distribution channels and are particularly easy to obtain via the Internet. The U.S. FDA maintains a list of confirmed adulterated weight loss products through Public Notifications on their Medication Health Fraud webpage. The fraudulent addition of sibutramine has been recently detected in many tainted natural slimming pills, and is of serious health concern as it was removed from the U.S. market in 2010 due to unsafe health effects including heart attack. Therefore, the presence of sibutramine at any concentration is of interest to regulators and consumers.
Thin layer chromatography (TLC) is an analytical separation technique where separation occurs on an open stationary phase layered on a support such as glass or plastic. High Performance TLC (HPTLC), a quantitative technique, is the semi- or fully automated sample application, plate development, and analysis. Absolute confirmation of contaminants separated by TLC is done using the ACQUITY QDa by directly interfacing the plate to the mass detector source inlet. A 200 mg capsule of Sheng Yuan Fang slimming product (herbal natural product) is weighed and dissolved in 10 mL methanol. Samples are homogenized for 30 seconds by vortex mixing and extracted in an ultrasonic bath for 10 minutes at room temperature. After centrifugation at 2750 RCF for 10 minutes at 25 °C, the supernatant is collected and used as test solution. Sibutramine reference material was purchased by Fluorochem (Derbyshire, UK) and prepared at a concentration of 0.785 mg/mL in methanol. 5 µL of sample, and 2 µL of standard were spotted onto an HPTLC Si 60 F254, 20 x 10 cm plate by the Automatic TLC Sampler 4 and developed in 9:1 toluene/methanol solution. The plate is derivatized then visualized under white light.
Target zones are directly eluted using the TLC-MS Interface 2 with oval elution head into the ACQUITY QDa at a flow rate of 0.5 mL/min with 0.1% formic acid in acetonitrile. The ACQUITY QDa is operated in positive electrospray ionization mode using the default settings: 0.8 kV capillary voltage, 15 V cone voltage, and 600 °C desolvation temperature. A full scan spectrum with the range of 50–650 m/z is acquired at a sampling rate of 10.0 points/sec (continuum). Data processing and evaluation of mass spectra are performed with Empower Chromatography Data Software. Figure 1 shows the QDa results from the TLC plate elution (Figure 2) of sibutramine standard (lane B) and slimming product (lane C).

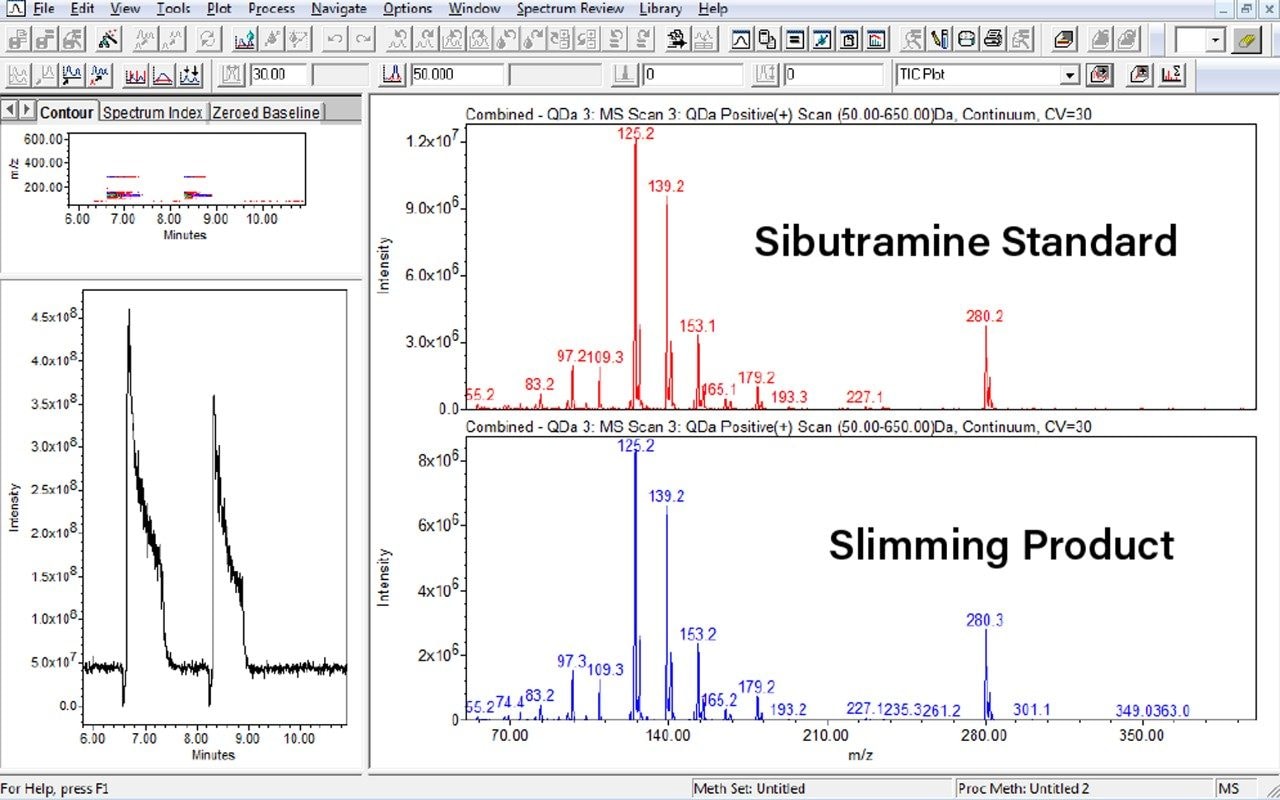
The ACQUITY QDa is an easy to use mass detector that can offer higher selectivity for complex samples than UV alone and give more confidence in analysis. It is especially useful in the analysis of compounds that have zero or poor UV absorbance where derivitization is necessary for sample analysis. Mass detection can bypass the extra derivitization steps and measure the sample directly.
720006046, June 2017