
In this application note, we further shortened the analysis time to 8.1 min, while meeting performance requirements using an ACQUITY H-Class UPLC System. The method performance characteristics of this UPLC method are presented.
Major isoflavones that are primarily found in soy (Glycine max), red colver (Trifolium pretense), and Kudzu (Pueraria lobata) are daidzein, glycitein, genistein, and their respective glucoside derivatives. The structures of 12 major isoflavones and apigenin, an internal standard (IS), are shown in Figure 1. Methods for the determination of isoflavones in dietary supplements have been established.1,2 These standard methods are based on reversed-phase high performance liquid chromatography (RP-HPLC) and ultraviolet and visible light (UV-Vis) spectroscopy detection. Because of the structural similarity and the large number of isoflavones, the method chromatographic run times are more than 70 minutes long. Recently, we have transferred an isoflavones standard method1 onto an ACQUITY Arc UHPLC System.3 The run time of the new method was reduced to 18 minutes per injection cycle. We also demonstrated the benefits of a small but powerful mass spectrometer, the ACQUITY QDa Mass Detector.4,5 With the ACQUITY QDa Detector, the time spent in the method transfer and development was greatly reduced, and the risk of interference was significantly lowered. In this application note, we further shortened the analysis time while meeting performance requirements using an ACQUITY H-Class UPLC System. The method performance characteristics of this UPLC method were presented.
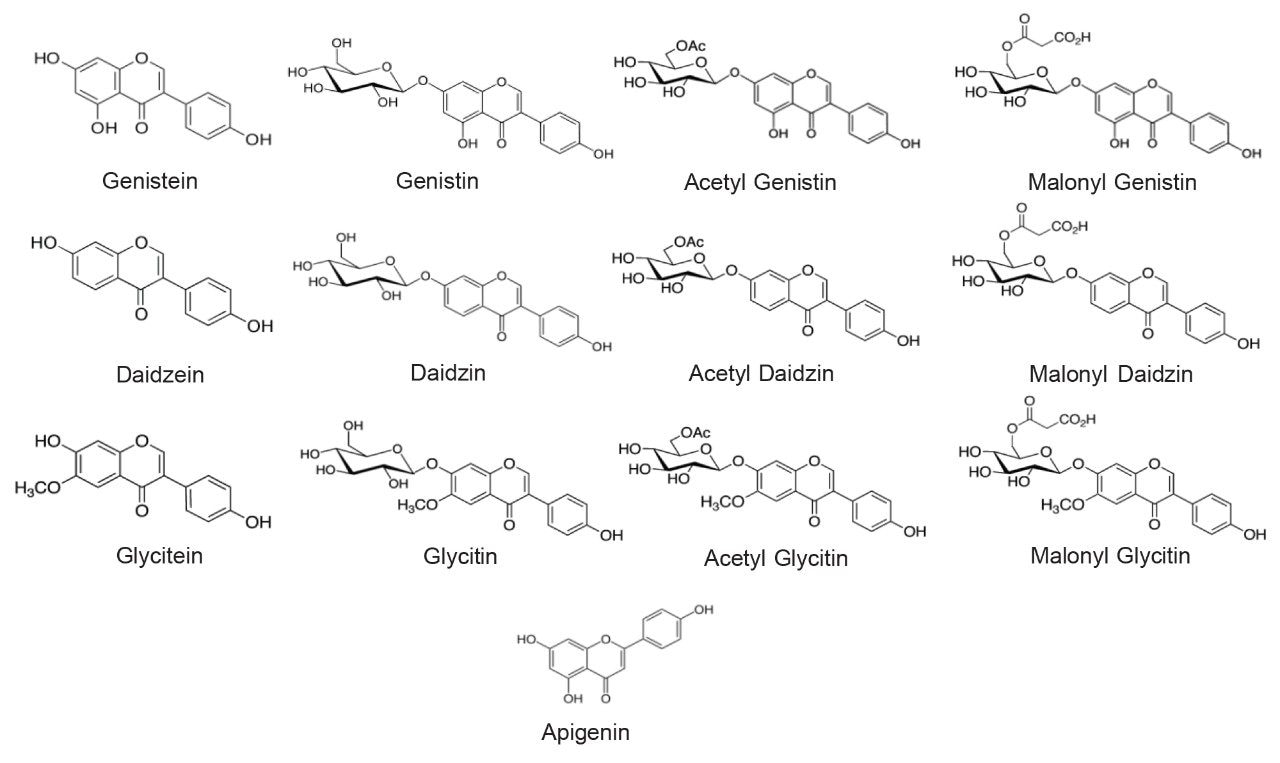
The standards, daidzin, glycitin, genistin, daidzein, glycitein, genistein, apigenin, were purchased from ChromaDex (Irvine, CA) and INDOFINE Chemical (Hillsborough Township, NJ). Defatted powdered Soy RS was purchased from US Pharmacopeia (Rockville, MD). NIST SRM 3238 was purchased from NIST (Gaithersburg, MD). Isoflavone dietary supplement samples from major brands were purchased from online retail stores.
The standard and sample solutions were prepared the same way as in the USP isoflavone method.1 Sample solutions were further diluted with acetonitrile (ACN) water mixture (2/3 by volume) to various levels to fit the calibration range. The concentration of IS (apigenin) was always kept at 4 ppm.
|
System: |
ACQUITY UPLC H-Class with ACQUITY PDA and ACQUITY QDa Detector (Performance) |
|
Software: |
Empower 3 |
|
Column: |
CORTECS UPLC C18 2.1 x 75 mm, 1.6 μm |
|
Column temp.: |
30 °C |
|
Mobile phase A: |
Water with 0.1% formic acid |
|
Mobile phase B: |
ACN with 0.1% formic acid |
|
Inj vol.: |
0.7 μL |
|
Flow rate: |
0.55 mL/min |
|
Run time: |
8.10 min |
|
UV detection: |
260 nm |
|
UV resolution: |
1.2 nm |
|
Time (min) |
Flow rate (mL/min) |
%A |
Curve |
|
|
1 |
Initial |
0.55 |
90 |
6 |
|
2 |
6.48 |
0.55 |
70 |
6 |
|
3 |
6.53 |
0.55 |
10 |
6 |
|
4 |
6.84 |
0.55 |
10 |
6 |
|
5 |
6.93 |
0.55 |
90 |
6 |
|
6 |
8.1 |
0.55 |
90 |
6 |
|
Polarity: |
ES+ |
|
Capillary (kV): |
0.8 |
|
Cone (V): |
15 |
|
Probe temp.: |
600 °C |
The USP method (isoflavones powder extract)1 was transferred to an ACQUITY UPLC H-Class System with a CORTECS UPLC C18 Column (1.6 µm, 2.1 x 75 mm). The CORTECS columns are packed with solid-core particles, which provide higher separation efficiency and lower back pressure than fully porous particles of equivalent particle size. The USP method’s HPLC gradient elution program was converted to a new UPLC gradient elution program using a Waters ACQUITY UPLC Column Calculator.3 The column properties of the HPLC and the UPLC methods were entered in the Column Calculator software to generate an equivalent UPLC elution program. This UPLC elution program was then used as the starting point and was optimized in the method development phase. The mobile phase additive was changed from 0.05% phosphoric acid to 0.1% formic acid, which is a mass spectrometry (MS) friendly additive. The factory default ACQUITY QDa instrument parameters were used without any modification.
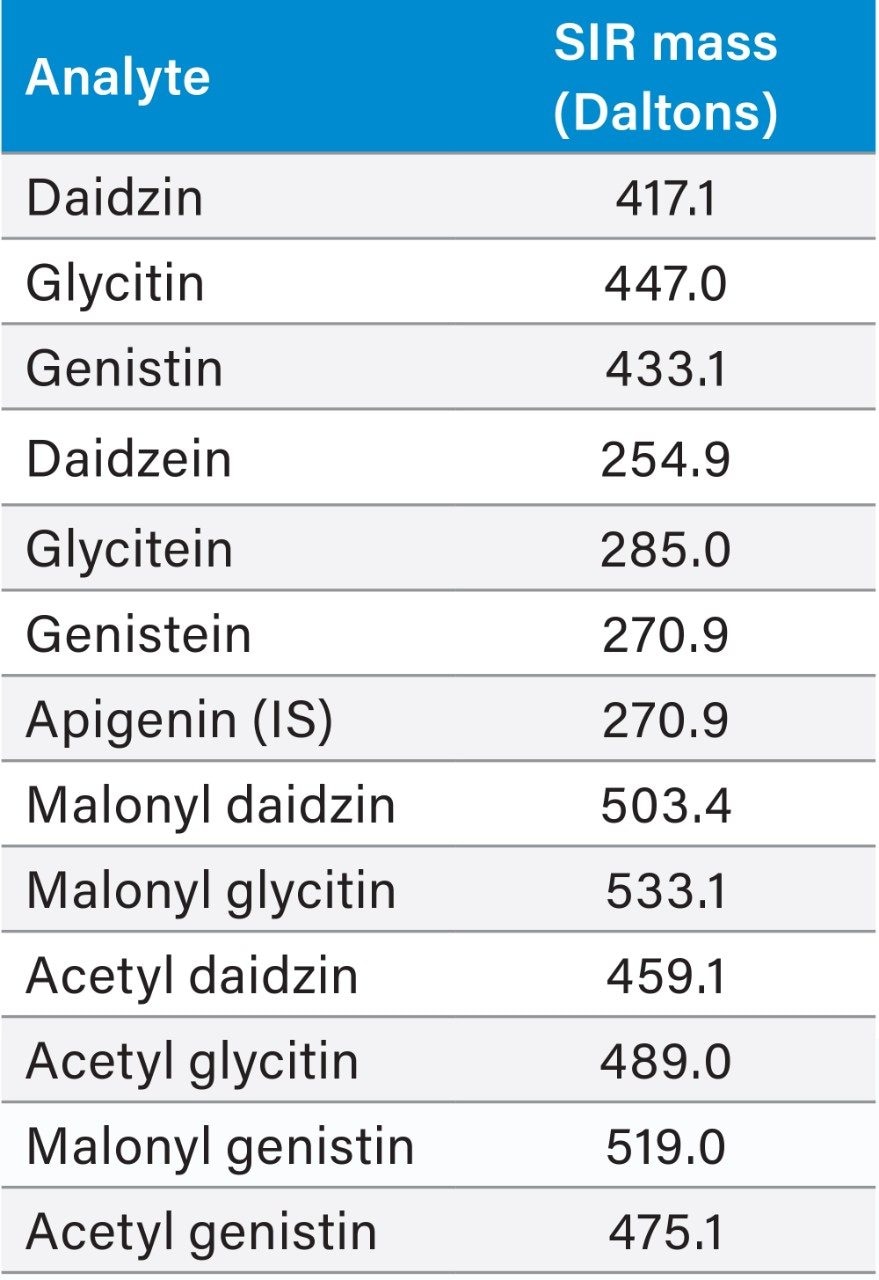
As the acetyl and the malonyl isoflavone standards are not commercially available, the peak assignment of these compounds relied on a reference material, the USP defatted powdered soy RS.1 By comparing the UV chromatograms of the unheated and the heated reference samples to their reference chromatograms, one can assign the peak IDs to the appropriate isoflavones. However, due to the change in LC conditions (HPLC to UPLC), and the close-eluting peaks, the UV peak patterns obtained in UPLC did not perfectly match those in the HPLC reference chromatograms. This issue was easily solved by using MS Single Ion Recording (SIR) traces (see Figure 2). Table 1 lists the isoflavone molecular ion masses. The Waters ACQUITY QDa Detector selectively detected these compounds in separate SIR channels, and avoided any possible interference from co-eluting compounds. The column temperature and the flow rate were optimized using the reference material. The optimization process was relatively quick since only one injection of the reference material generated enough information (peak RT and resolution) for one particular LC condition (column temperature or flow rate). Without the ACQUITY QDa Detector, usually several injections of standard solutions would have been needed for each LC condition in the method optimization process.
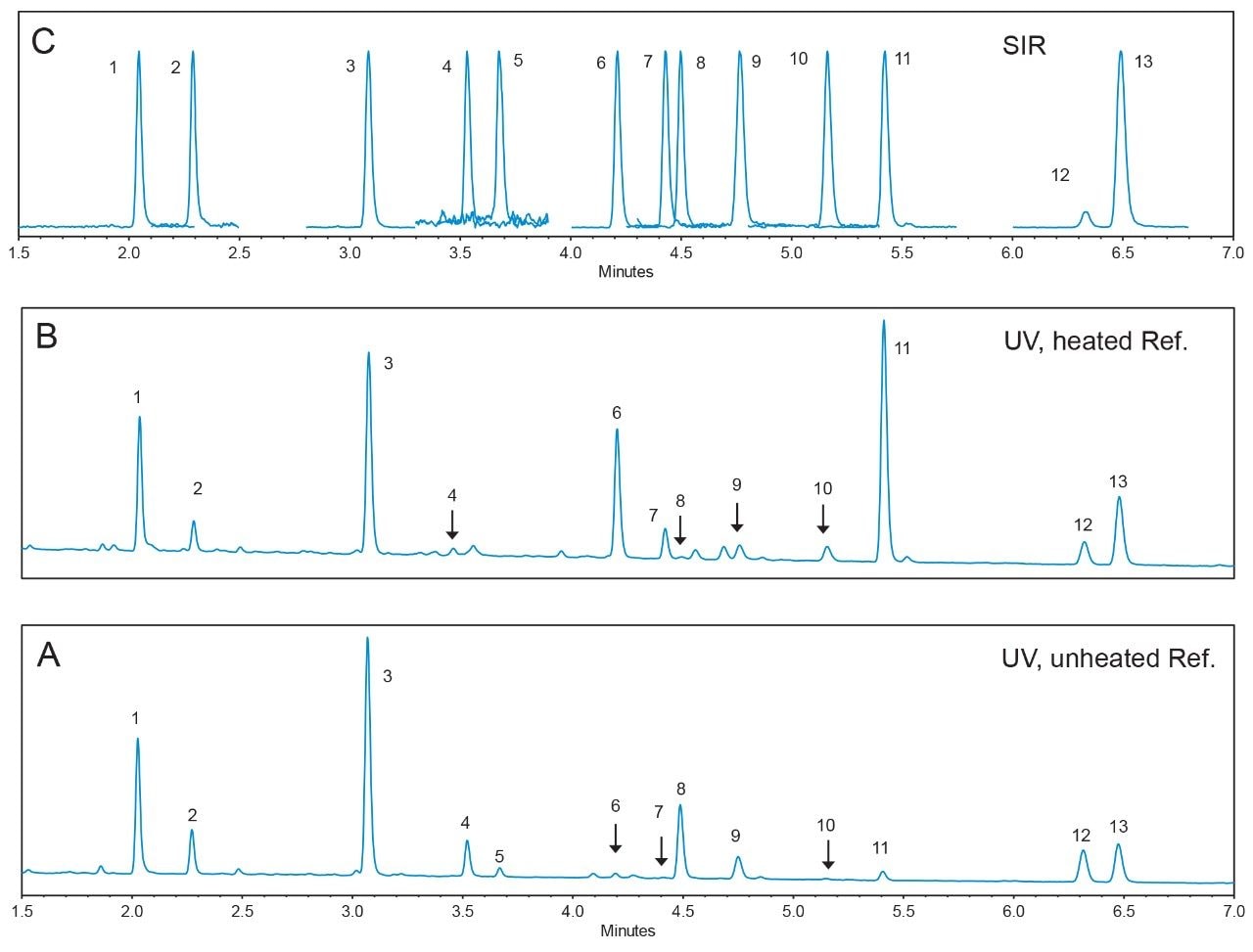
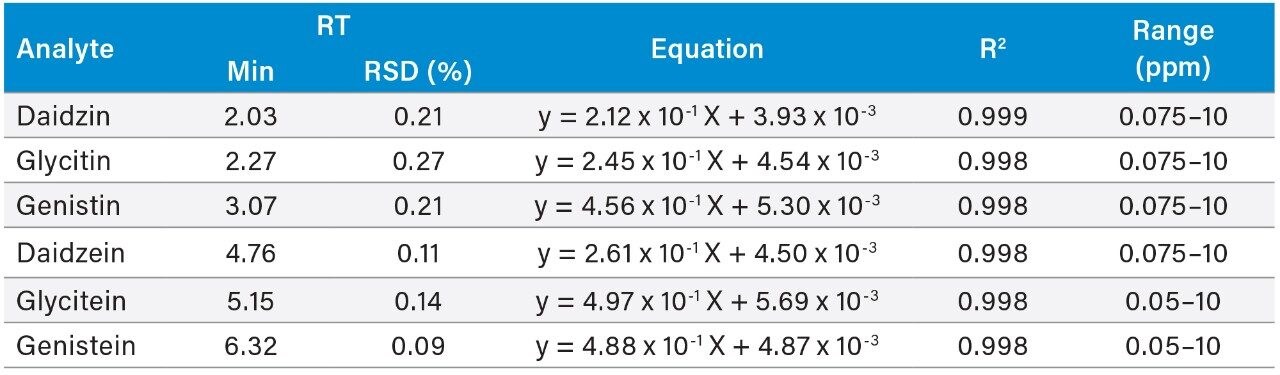
Table 2 shows the UV calibration results for the available standards. Apigenin was used as an IS for the calibration and the quantitation of all compounds. The square of the correlation coefficients (R2) between the responses (peak area ratio) and the standard concentrations (ppm) for all compounds were better than 0.998. The relative standard deviation (RSD) in retention time for all compounds were less than 0.27%.
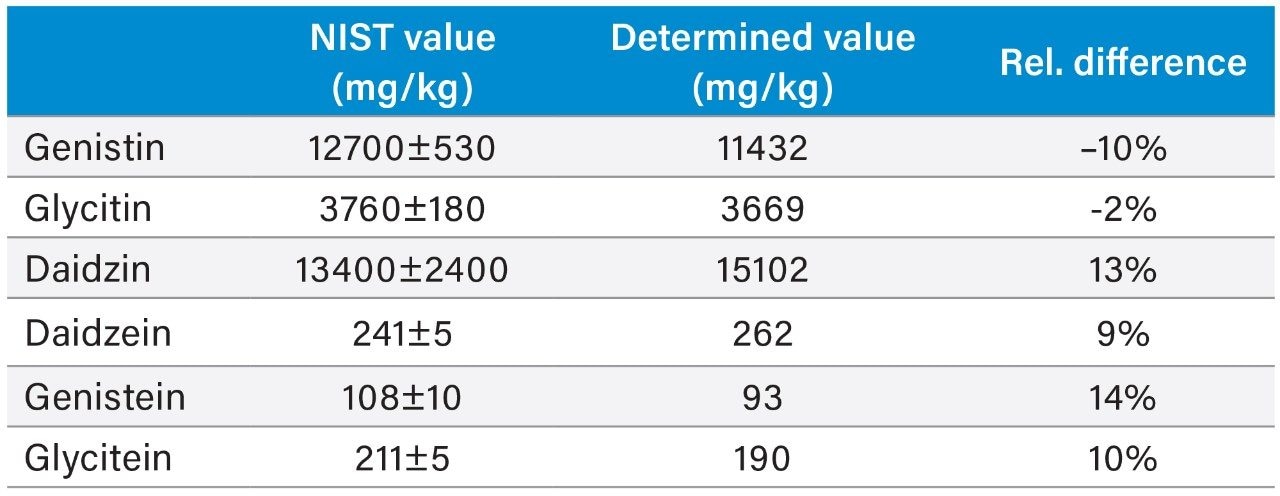
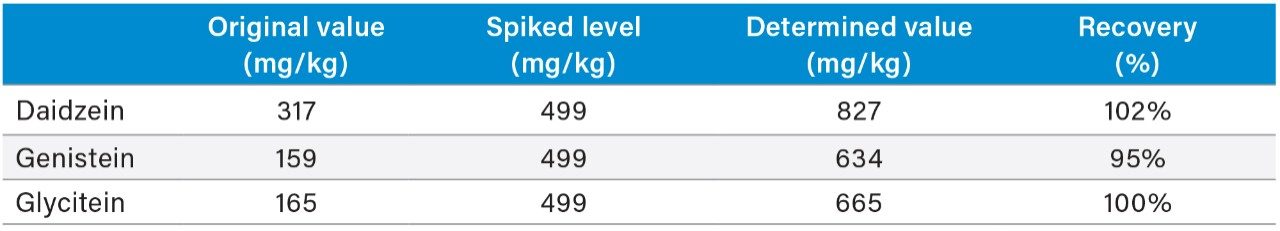
Table 3 shows the isoflavone results for the NIST 3238 SRM and a comparison to its certified and reference values. Relative difference of less than 14% was obtained for the genistin, glycitin, daidzin, genistein, and glycitein. The accuracy for the daidzein, genistein, and glycitein was also evaluated by a spiking experiment (Table 4). Recovery of 95% to 102% were obtained for these analytes.
The isoflavone contents in three isoflavone dietary supplement samples were measured by this 8-minute UPLC-UV method. The sample forms included tablets, capsules, and powder. The USP calibration and quantitation protocols1 were followed in the data processing. The conversion factors for the acetyl and malonyl derivatives that were specified in the USP HPLC-UV method were used in this analysis. Table 5 shows the determined individual and total isoflavone contents, as well as the total isoflavone contents on labels. For an easy comparison, the label values were converted to the same concentration unit (mg/kg). Two of the three samples (C and D) showed good agreement between the determined values and their label values, while one sample (B) had much less measured total isoflavone content than its label value. The reason for this low total isoflavone content in sample B compared to its label claim is unknown.
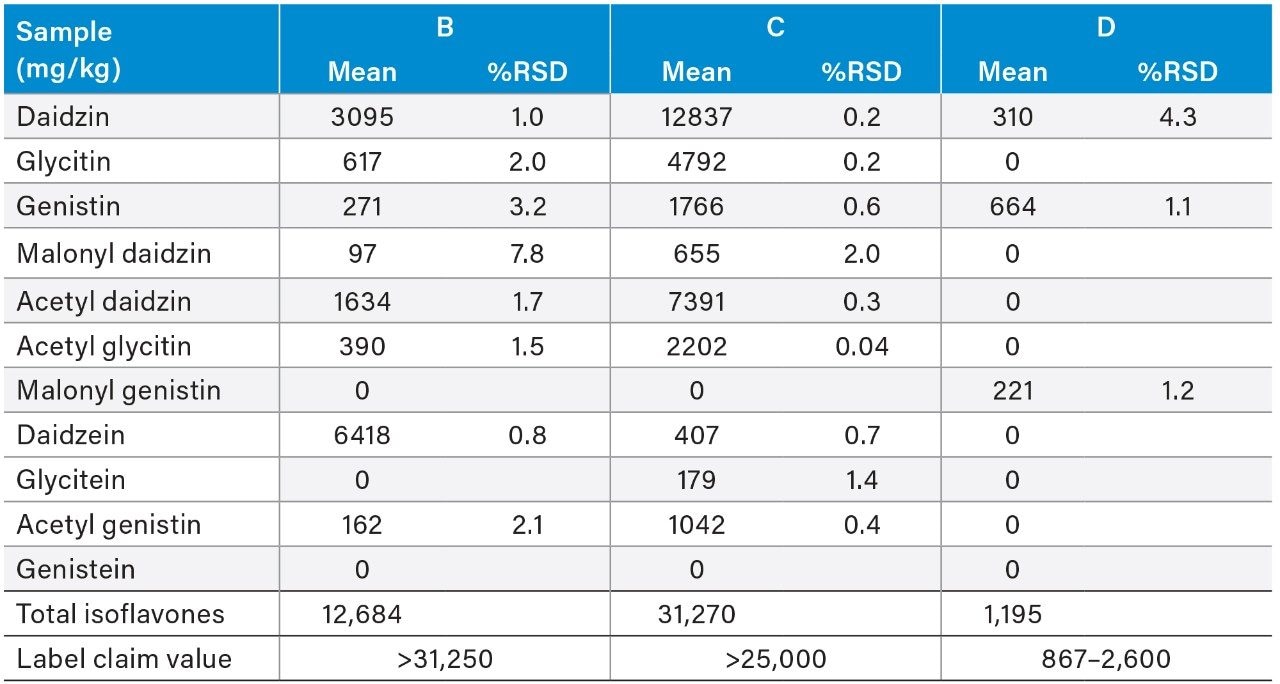
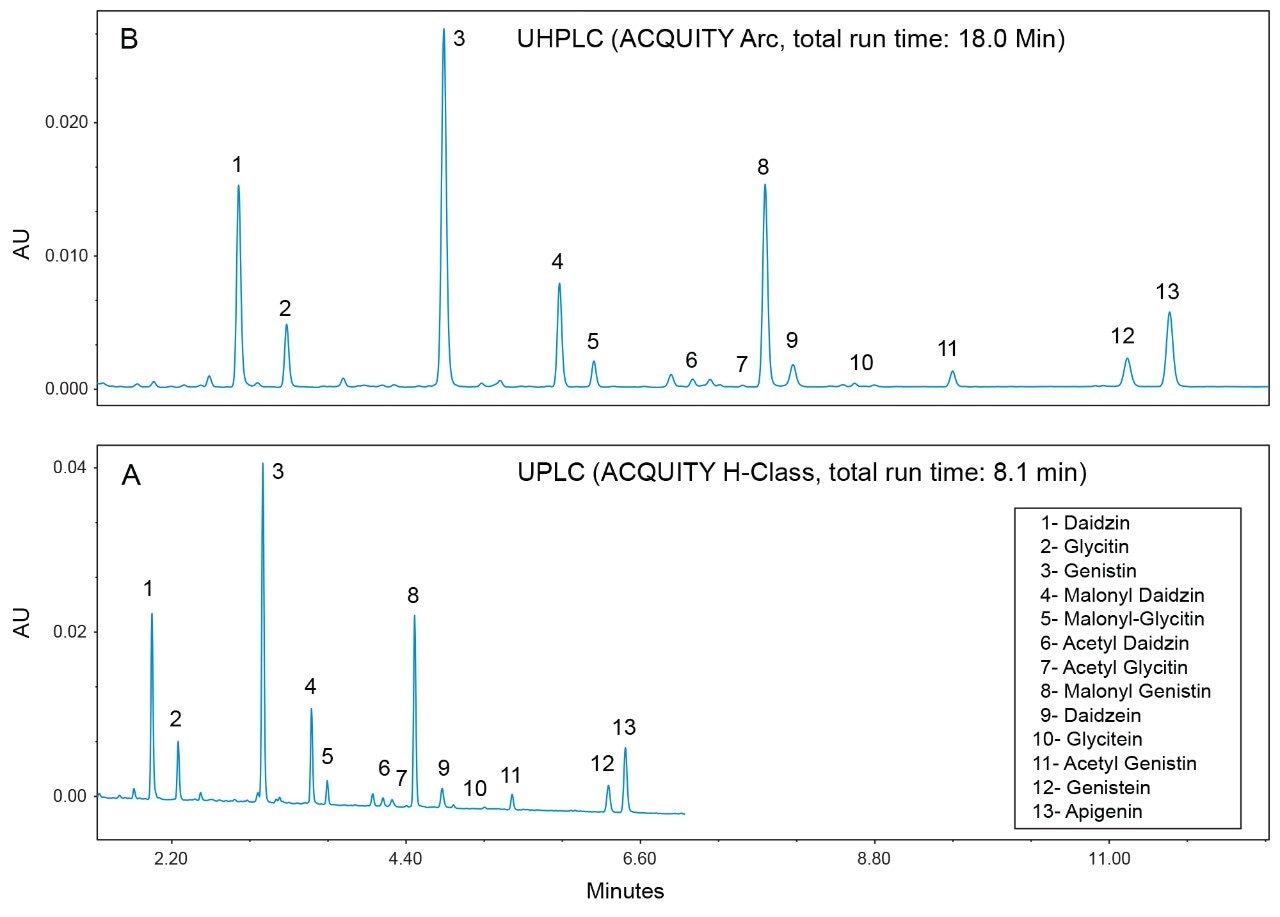
Figure 3 is a comparison of the UPLC and the UHPLC chromatograms of the same reference material (USP defatted soy RS). The total run time in the UPLC method was 8.1 min, which was less than a half of the total run time in the UHPLC method (18 min). The UPLC method can provide twice the sample analysis throughput than the UHPLC method. The solvent cost for UPLC was less than a quarter of that for UHPLC since the UPLC flow rate was about half that of UHPLC. Both of the UHPLC and the UPLC methods were much faster than the existing USP method, which is a 74 min HPLC method.
The MS detector was extremely useful during method transfer and development and in complex sample analyses. Compared to typical UV/Vis detection, the ACQUITY QDa Detector provided excellent detection selectivity. This high selectivity offered high confidence in peak assignment, which was extremely useful when pure standards were not available and interference was common.
The USP isoflavone method was successfully transferred to an ACQUITY UPLC H-Cass System with an ACQUITY PDA Detector. The total analysis time per injection was 8.1 minutes, which was less than the half of the UHPLC method (18 min),5 and significantly shorter than the HPLC methods (74 minute).1,2 The UPLC method provides 9 times higher analysis throughput, and more than 90% cost savings in solvents than the HPLC methods. ACQUITY QDa Mass Detector offered excellent detection selectivity, which was a great asset in the method transfer and development, as well as in the isoflavone analysis for unknown and challenging samples where potential interference from co-eluting compounds can be high. Analysis of isoflavones in three dietary supplements showed compliance with the label claims for two of the samples. One sample showed low total isoflavone content comparing to its label claim.
Optional section to showcase related work by customers, Waters colleagues, or collaborators not formally referenced (a short synopsis may be used to introduce), or web links to relevant additional information such as product videos, peer-reviewed papers, posters, articles, etc.
720006065, July 2017