For forensic toxicology use only.
This application note details the extraction of THC-OH, THC-COOH, and THC from plasma samples using a novel SPE sorbent, Oasis PRiME HLB, in a μElution format for forensic toxicology applications.
Amongst forensic toxicology laboratories, cannabis is one of the most frequently encountered drugs.1 For most forensic laboratories providing toxicological analyses, the confirmation, and quantification of Δ9-tetrahydrocannabinol (THC) and its primary metabolites in whole blood or plasma is high on the list of requests from clients and enforcement agencies, such as district attorneys and/or police departments (city, county, or state levels).2 While whole blood is often used in postmortem forensic cases, whole blood/plasma ratios can be quite inconsistent, particularly for COOH-THC.3 Therefore, for pharmacokinetic studies involving medicinal uses or behavioral effects, plasma is often a preferred matrix.4,5 The ability to efficiently extract and quickly analyze samples submitted by such agencies is seen as a definite benefit to the laboratory. In this application, the efficient extraction and accurate quantification of THC and two major metabolites, THC-COOH and THC-OH, is achieved using a novel solid-phase extraction (SPE) sorbent, Oasis PRiME HLB, coupled with fast analysis using LC-MS/MS. This method offers a great solution for laboratories looking to incorporate this type of analysis into their methodology portfolio.
Sample preparation is an important consideration for forensic toxicology laboratories. Specifically, there is a desire to use the simplest possible method which yields the maximum cleanup. THC and its metabolites, in particular, can be prone to non-specific binding during sample preparation and manipulation, so any method that minimizes sample manipulation, evaporation, and reconstitution is desirable. This application employs Waters’ newly developed sample preparation sorbent, Oasis PRiME HLB, which is designed to provide several key advantages over traditional SPE sorbents. These include the ability to eliminate sorbent preconditioning and equilibration, creating a faster workflow compared to traditional SPE products. It also has the ability to remove more matrix interferences, particularly phospholipids, resulting in cleaner extracts and reducing the risk of short column lifetimes or MS source fouling. Compared to liquid-liquid extraction (LLE) methodologies, this method avoids the use of toxic, LC incompatible solvents and does not require evaporation and reconstitution of the extracts. In addition, phospholipids are generally not removed by LLE, so the extracts produced from this method are cleaner in that respect, with less risk of ion suppression from residual phospholipids.
This application details the extraction and analysis of THC and its major metabolites, 11-hydroxy Δ-9-THC (THC-OH) and 11-nor-9-Carboxy-Δ-9-THC (THC-COOH)6 from plasma using an Oasis PRiME HLB µElution Plate, followed by UPLC-MS/MS analysis. The SPE procedure is simple, fast and very efficient, with concentration occurring through the device and elution in LC compatible solvents, allowing for direct injection without subsequent evaporation and reconstitution of samples. Analysis is rapid and highly consistent, with all analytes eluting in less than 3 minutes. Recoveries were excellent (all over 75% with RSDs less than 6%) and matrix effects (ME) were minimal for all compounds (all MEs less than 20%). Quantitative results were highly reproducible. Quality control results were within 10% of expected concentrations and average RSDs within 5%.
All standards and stable isotope labeled internal standards were purchased from Cerilliant (Round Rock, TX, USA). Stock standards at 100 µg/mL were prepared in 40% methanol (THC, THC-OH, and THC-COOH). A working internal standard (I.S.) solution, consisting of 100 ng/mL THC-D3, THC-OH-D3, and THC-COOH-D3 was also prepared in 40% methanol. Individual calibrator and quality control standard solutions were prepared daily in 40% methanol.
100 µL of each working calibrator or QC standard and 100 µL internal standard were added to 1800 µL of plasma to make calibration curves and QC samples. Calibrator concentrations ranged from 0.1–100 ng/mL for all analytes. Quality control samples were prepared at 0.375, 1.75, 7.5, and 37.5 ng/mL in plasma.
200 µL 0.1% FA in ACN was added to 100 µL plasma to precipitate out the protein in a micro centrifuge tube. Then the mixture was vortexed for 5 seconds and centrifuged for 5 minutes at 7000 rcf. The supernatant was then diluted with 400 µL water prior to loading.
The entire pre-treated sample was directly loaded on to the Oasis PRiME HLB µElution Plate (p/n 186008052) without conditioning or equilibration. All wells were then washed with 2 x 250 µL aliquots of 25:75 methanol:water. All the wells were then eluted with 2 x 25 µL aliquots of 90:10 ACN:MeOH and diluted with 50 µL of water prior to analysis. 5 µL was injected onto the UPLC-MS/MS system. The SPE extraction procedure is summarized in Figure 1.

Analyte recovery was calculated according to the following equation:
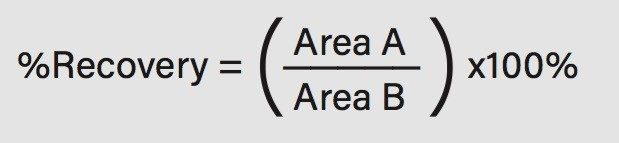
Where A equals the peak area of an extracted sample and B equals the peak area of an extracted blank matrix sample in which the compounds were added post-extraction.
Matrix effects were calculated according to the following equation:

The peak area in the presence of matrix refers to the peak area of an extracted matrix sample in which the compounds were added post-extraction. The peak area in the absence of matrix refers to analytes in a neat solvent solution.
|
UPLC system: |
ACQUITY UPLC I-Class |
|
Column: |
ACQUITY UPLC BEH C18, 1.7 μm, 2.1 x 50 mm (p/n 186002350) |
|
Column temp.: |
40 °C |
|
Sample temp.: |
10 °C |
|
Mobile phase A (MPA): |
Water with 0.1% formic acid |
|
Mobile phase B (MPB): |
ACN with 0.1% formic acid |
|
Strong wash solvent: |
70:30 ACN:water with 2% formic acid |
|
Weak wash solvent: |
10% ACN |
|
Injection volume: |
5 μL |
|
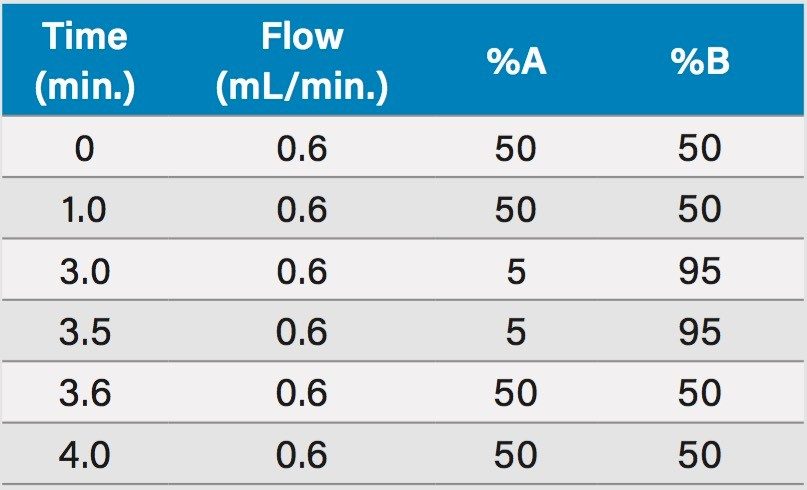
The gradient ramp is shown in Table 1. |
|
MS system: |
Xevo TQ-S Mass Spectrometer |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
2.0 kV |
|
Cone voltage: |
Optimized for each analyte |
|
Desolvation gas: |
1000 L/hr |
|
Cone gas: |
150 L/hr |
|
Desolvation temp.: |
500 °C |
|
Source temp.: |
150 °C |
Data were acquired and analyzed using MassLynx Software (v4.1). Quantification was performed using TargetLynx.
Figure 2 shows UPLC chromatograms of the three cannabinoids from an extracted calibrator at 1 ng/mL. All compounds eluted within 3 minutes and all peak widths were under 3 seconds at 5% of baseline. All peaks were symmetrical with peak asymmetries between 0.95–1.15.
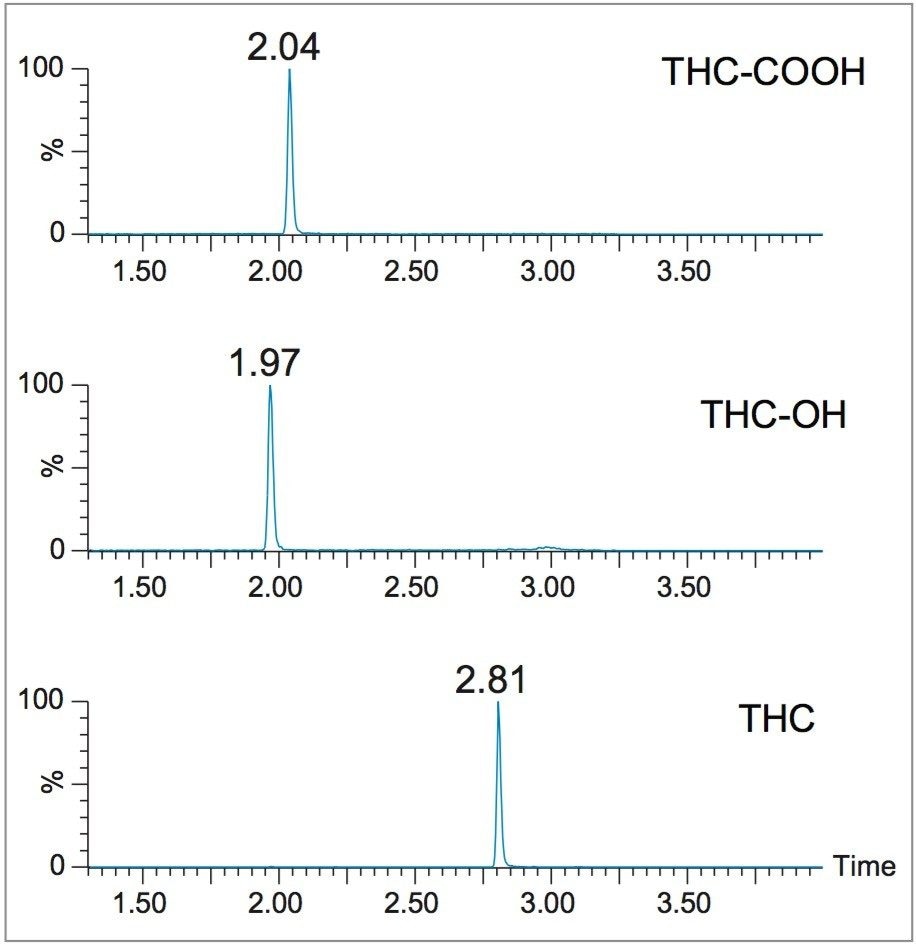
Figure 2. Chromatograms of THC-OH, THC-COOH, and THC from extracted plasma samples.
This separation was run on the ACQUITY UPLC I Class System – Xevo TQ-S, ACQUITY UPLC BEH C18 , 1.7 µm, 2.1 x 50 mm Column (p/n 186002350), with system back pressure around 6500 psi. The concentrations are 1 ng/mL for all compounds.
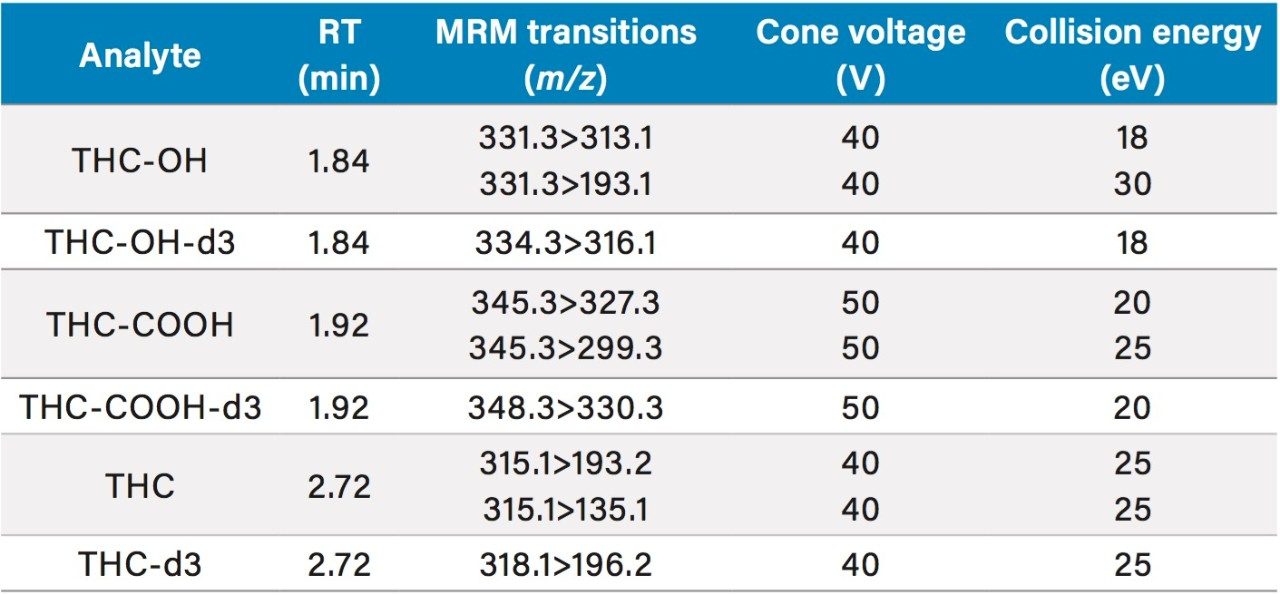
Table 2 lists the UPLC retention times and individualized MS parameters of the cannabinoids and their stable isotope labeled internal standards, including MRM transitions, cone voltages, and collision energies. Two MRM transitions were used for each compound, a primary (listed first) and a confirmatory transition (listed second).
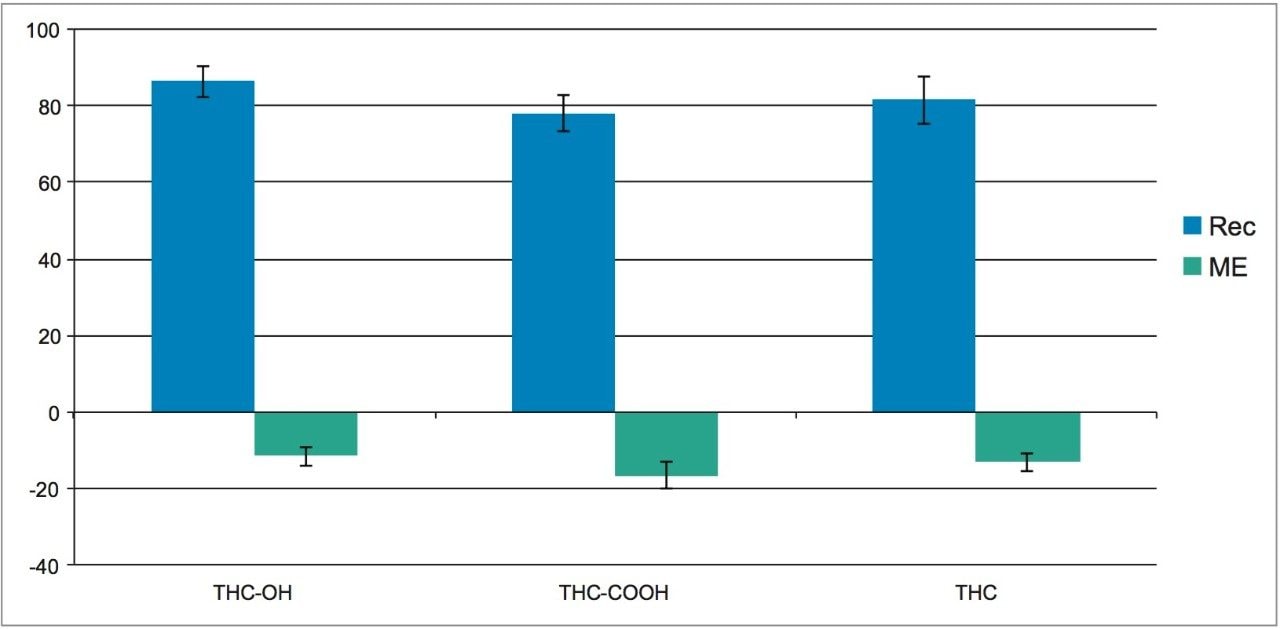
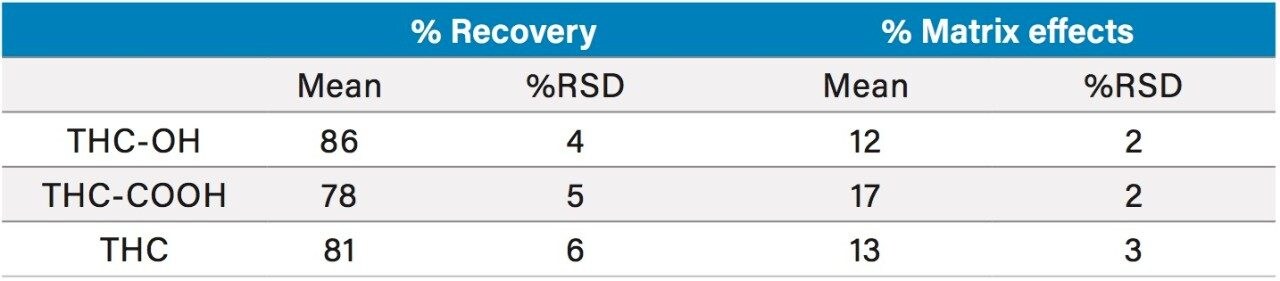
Extraction recoveries were high and consistent. As Figure 3 shows, recovery for all analytes was around 80% with all %RSDs under 6%, demonstrating the high reproducibility of Oasis PRiME HLB. Matrix effects were low, at less than 20% for all compounds with standard deviations <3%. Once again, the low standard deviations (6% or less) demonstrate the consistency of extraction and cleanup seen with Oasis PRiME HLB. All recovery and matrix effect data are summarized in Table 3.
One of the key attributes of Oasis PRiME HLB is its ability to deliver cleaner extracts than other sample preparation methods. One way that this is achieved is by removing endogenous phospholipids. Figure 4 shows the profile of phospholipids remaining in plasma extracted with Oasis PRiME HLB using different elution solvents. A total of 18 different elution solvents were evaluated to compare the amount of phospholipids remaining, including different percentages of MeOH in water, ACN in water, and ACN/MeOH combinations. The figure shows that ACN does not seem to elute as many phospholipids as MeOH. Overall, >80% ACN in water or >75/25 ACN/MeOH as elution solvent for Oasis PRiME HLB are recommended for the cleanest possible eluates. For this application, a combination of 90:10 ACN:MeOH resulted in high, consistent analyte recovery with low residual phospholipids.
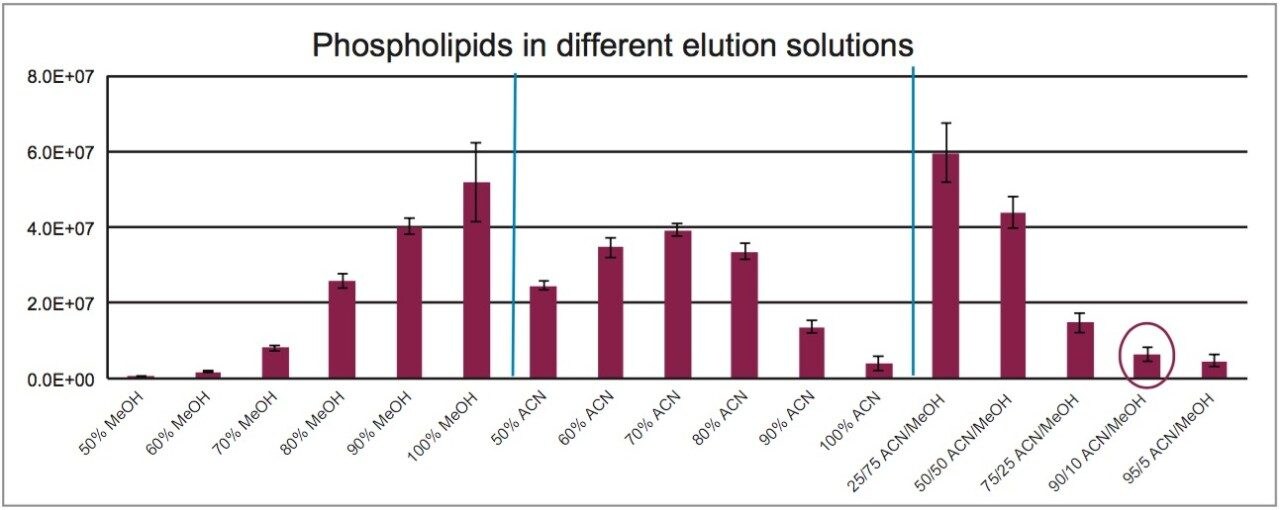
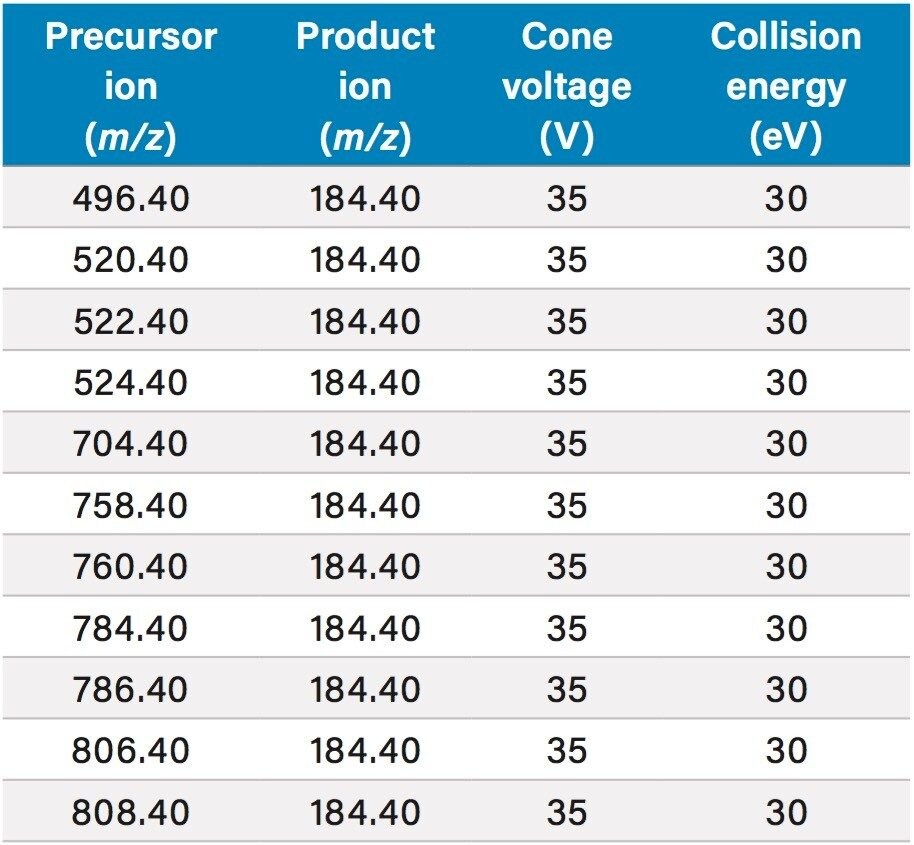
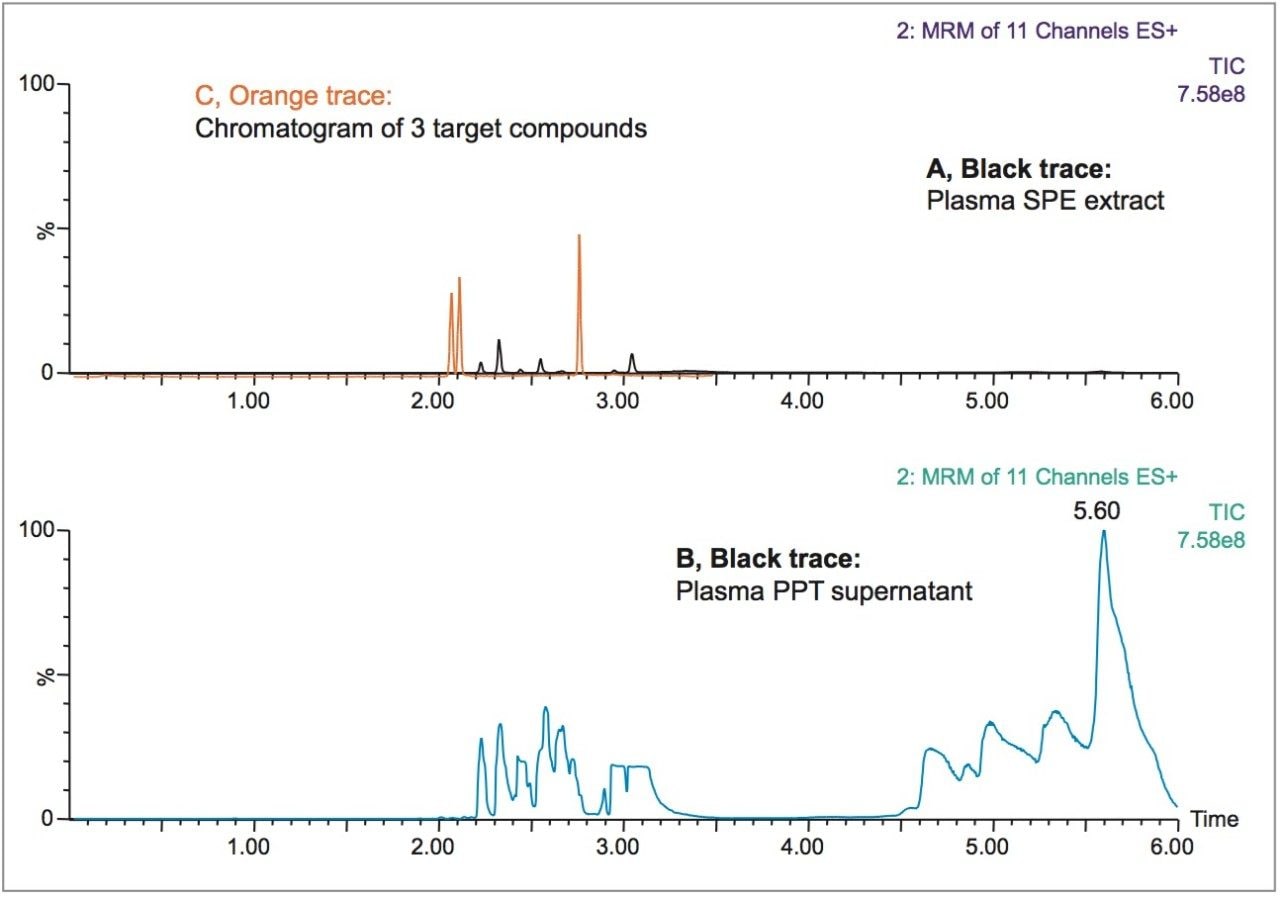
Figure 5 shows chromatograms of combined phospholipid traces from an Oasis PRiME HLB extract (A) and an identical sample subject to protein precipitation (B). Compared with protein precipitation (PPT), Oasis PRiME HLB extract removes over 99% of phospholipids, resulting in a much cleaner extraction. This can translate to reduced matrix effects, longer column lifetimes, and less mass spectrometer source maintenance.
The chromatography of the three target compounds is also shown (C), demonstrating the potential interference of phospholipids if they were not removed during the extraction.
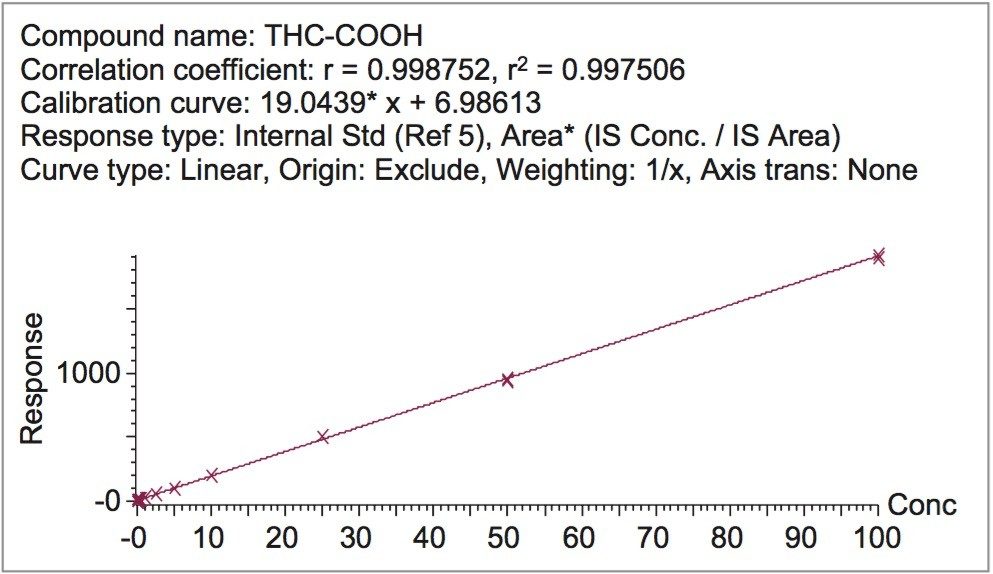
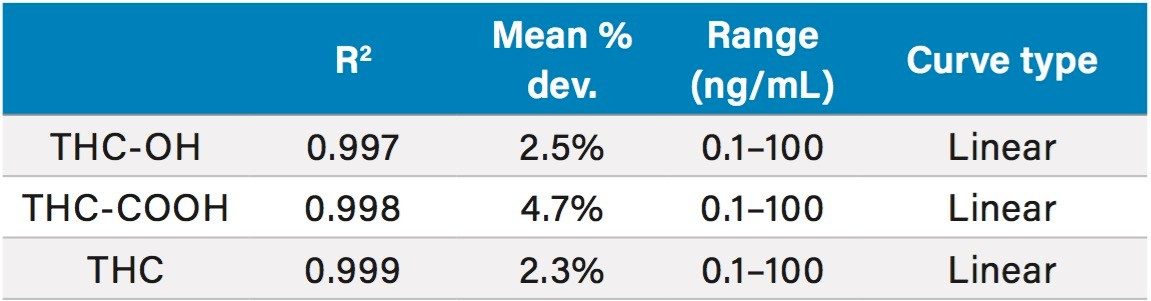
Calibration and quality control samples were prepared as previously described in the materials and methods section. Calibration ranges were from 0.1–100 ng/mL for all tested compounds. Quality control samples were prepared at four concentrations as appropriate for the calibration ranges. All compounds had linear responses over the entire calibration range with R2 values of 0.99 or greater with 1/x weighting with average deviations <5%. Figure 6 shows the calibration curve of THC-COOH and Table 5 summarizes the data from these curves for all the compounds. Lower limits of quantification (LLOQ) were 0.1 ng/mL for all compounds. In each case, all FDA recommendations for accuracy, precision, linearity, and analytical sensitivity were met for validated methods.
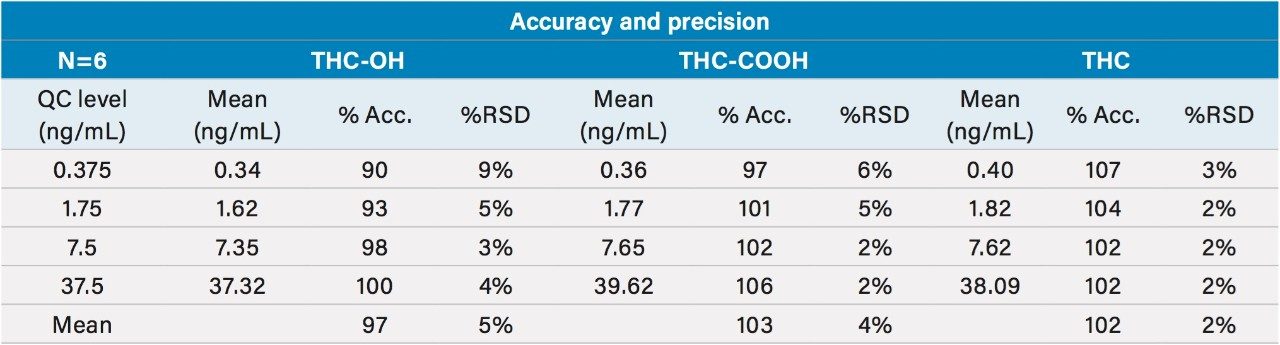
Quality control samples prepared at 0.375, 1.75, 7.5, and 37.5 ng/mL were accurate and precise. All results were within 10% of expected values with average RSDs between 2–5% (N=6). This data can be seen in Table 6 and it demonstrates that the method is linear, accurate, and precise over a calibration range that includes the entire scope of expected values of samples. The method was also proved to be both selective and sensitive enough to routinely measure THC in plasma well below a 2–3 ng/mL cut off level.3 This was exemplified by the excellent accuracy and precision at the 0.375 ng/mL QC sample level, where calculated concentrations of all six replicates were within an average of 10% of expected.
This application note details the extraction of THC-OH, THC-COOH, and THC from plasma samples using a novel SPE sorbent, Oasis PRiME HLB, in a µElution format for forensic toxicology applications. The unique nature of this sorbent enabled the elimination of conditioning and equilibration steps, simplifying the extraction procedure, and speeding up the sample preparation workflow. In addition, the µElution format enabled the direct injection of extracts without evaporation or reconstitution, saving time and minimizing the risk of nonspecific binding. One key attribute of this sorbent is its ability to remove phospholipids resulting in a much cleaner final sample. As mentioned previously and shown in Figure 5, 99% of phospholipids were eliminated from extracted samples, some of which would have co-eluted with the target analytes in this assay.
Recoveries were very consistent, (80% with RSDs < 6%). Matrix effects were less than 20% for all compounds. Linearity, accuracy, precision, and analytical sensitivities were excellent for all compounds. All accuracies were within 10% of target concentrations with average RSDs between 2–5%, demonstrating the high reproducibility of the combination of this sorbent and the UPLC-MS/MS method. In conclusion, Oasis PRiME HLB has been successfully used to achieve consistent recoveries with low matrix effects as well as accurate quantification at 4 orders of magnitude from plasma samples.
720005820, October 2016