
This application note demonstrates a sensitive LC-MS/MS method for the analysis of 23 common PAAs with very easy sample preparation. The Xevo TQ-S micro provides sensitive detection levels to satisfy the EU 2020/1245 regulations.
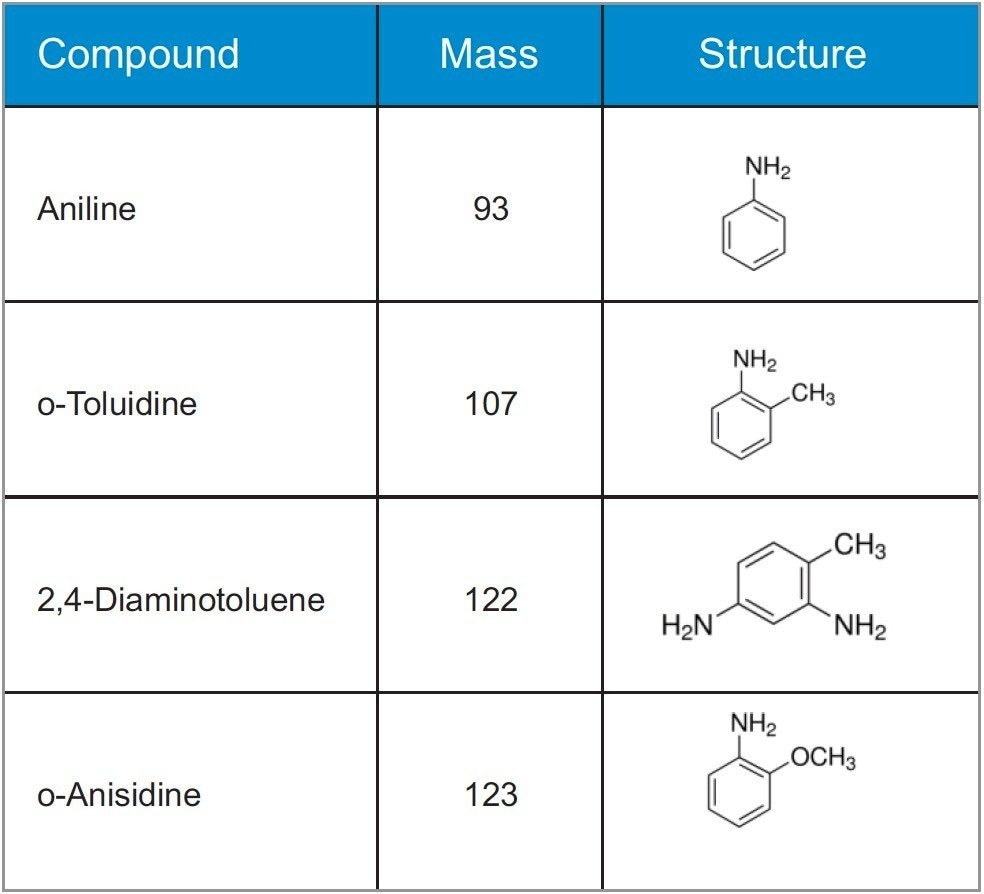
Primary Aromatic Amines (PAAs) are a class of compounds of which the simplest form is aniline (Figure 1). PAAs are substances that are used, for example, in the production of certain colorants, so-called azo pigments, notably in the color range yellow – orange – red. Whereas a large number of PAAs are safe for human health, some PAAs are known human carcinogens. For kitchenware, paper napkins, baker’s bags with colorful print, and other printed items that come in contact with food, some PAAs may pose a health risk, if they are transferred to the food.
Because of the potential health risks, specific migration limits (SMLs) are put in place. The longstanding EU 10/2011 regulation1 has recently been amended with EU 2020/1245.2 Whereas previously, in relation to PAAs, ‘Plastic materials and articles shall not release primary aromatic amines, excluding those appearing in Table 1 of Annex I, in a detectable quantity into food or food simulant. The detection limit is 0.01 mg of substance per kg of food or food simulant. The detection limit applies to the sum of primary aromatic amines released’. The recent EU 2020/1245 update has identified that PAAs listed in Annex I of 10/2011/EU (for example 1,3 phenylenediamine) must not be detectable above 0.002 mg/kg in food or food simulant and individual PAAs listed in entry 43 of Appendix 8 of 1907/2006/EC3 shall not be detectable above 0.002 mg/kg in food or food simulant. Furthermore, the EU 2020/1245 amendment stipulates for other PAAs (not listed in Annex I of the regulation nor Appendix 8 of 1907/2006) should be assessed for risk, but to avoid potential combined toxicity, their total migration is set to a maximum of 0.01 mg/kg food or food simulant.
The provisions state that for primary aromatic amine migration from polyamide kitchenware, only one migration test will be carried out, if this first extract is compliant within limits. However, if this first simulant extract exceeds the permitted limits, then two subsequent migration studies are required.4 This PAAs migration testing is conducted with simulant B, 3% (w/v) acetic acid, as it has been demonstrated that this simulant represents the worst case for the migration of PAAs from polyamide kitchenware.5
PAAs are small, basic compounds, which are ionized with low pH. As a result of their basic properties and the 3% acetic acidic sample solvent, some PAAs don’t focus well on the head of the column, resulting in poor peak shape and/or loss of retention. In order to improve chromatographic retention ion-pairing reagents are often used.4 Unfortunately these reagents have a negative impact on the electrospray sensitivity and are to be avoided where possible.
In this application note we describe a LC-MS/MS method for the analysis of 23 common PAAs in kitchenware after migration, using a Waters ACQUITY UPLC I-Class System coupled to a Xevo TQ-S micro Mass Spectrometer. The described method does not use an ion-pair reagent to improve chromatographic retention.
|
UPLC system: |
ACQUITY UPLC I-Class |
|
Sample manager: |
Flow-Through Needle |
|
Column: |
ACQUITY UPLC HSS T3, 1.8 μm, 2.1 x 100 mm |
|
Mobile phase A: |
Water |
|
Mobile phase B: |
Methanol |
|
Column temp.: |
45 °C |
|
Sample temp.: |
10 °C |
|
Flow rate: |
0.4 mL/min |
|
Run time: |
15 min |
|
Injection volume: |
20 μL |
|
0 min |
5% B |
|
10 min |
100% B |
|
12 min |
5% B |
|
12.01 min |
5% B |
|
15 min |
5% B |
|
MS system: |
Xevo TQ-S micro |
|
Ionization mode: |
ESI + |
|
Capillary voltage: |
2 kV |
|
Desolvation temp.: |
600 °C |
|
Desolvation gas flow: |
1200 L/hr |
|
Source temp.: |
150 °C |
|
Acquisition: |
Multiple Reaction Monitoring (MRM) |
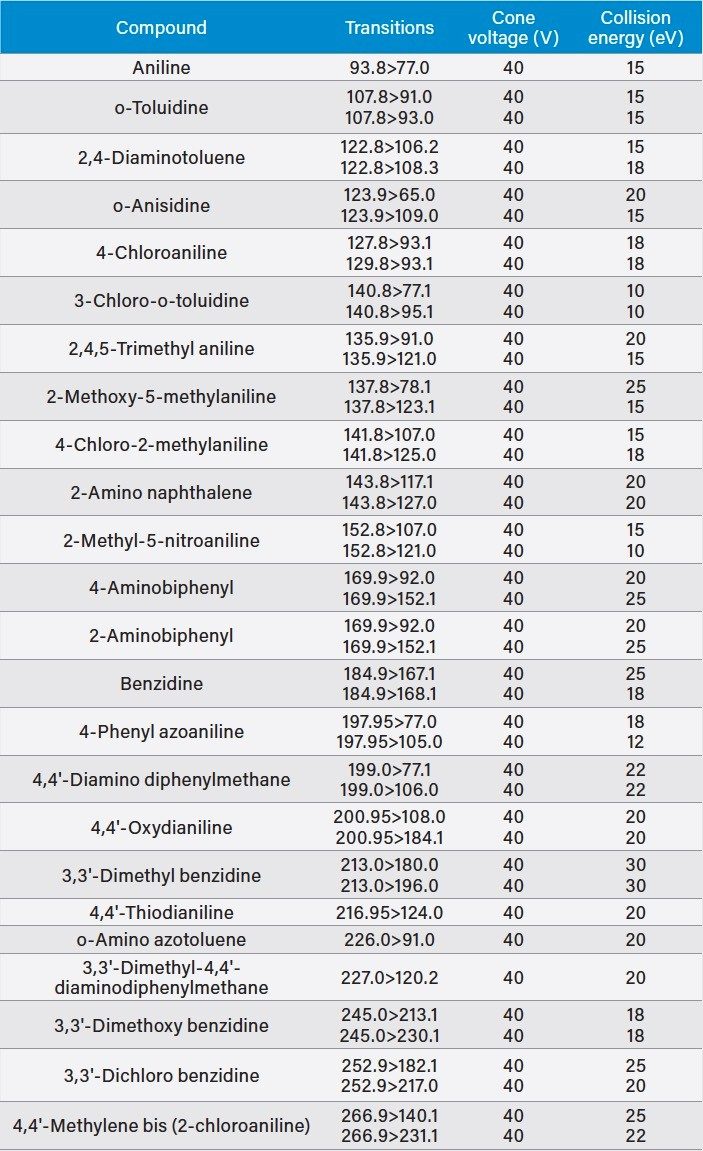
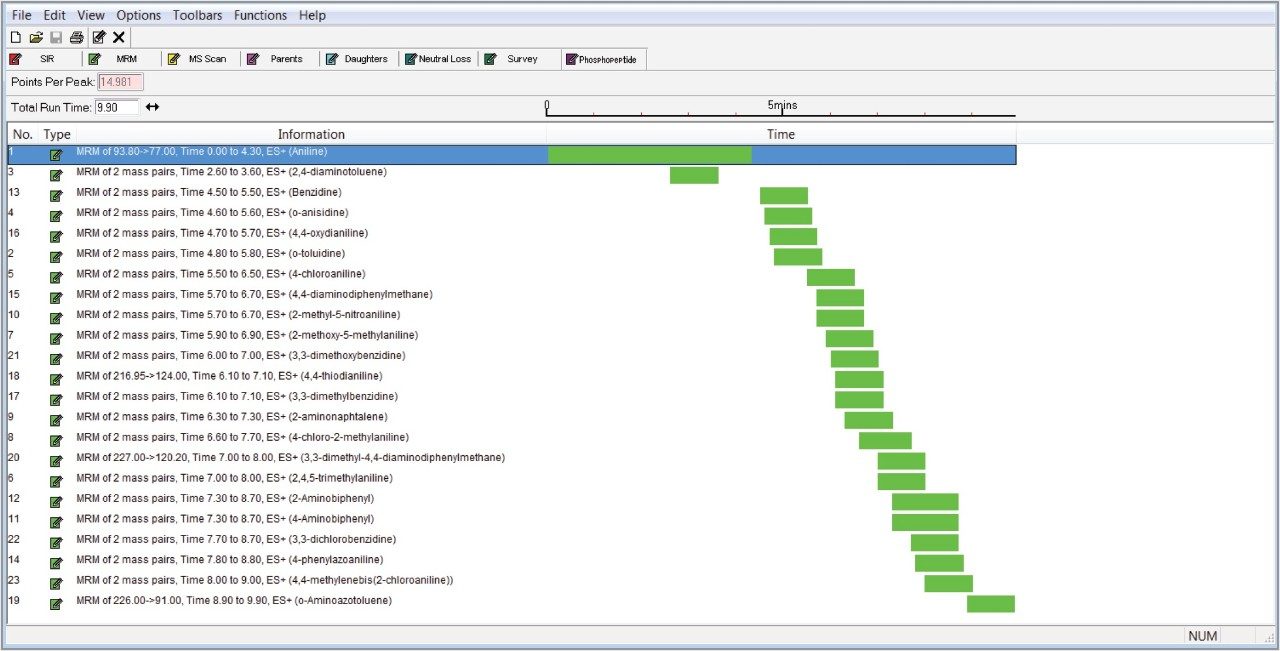
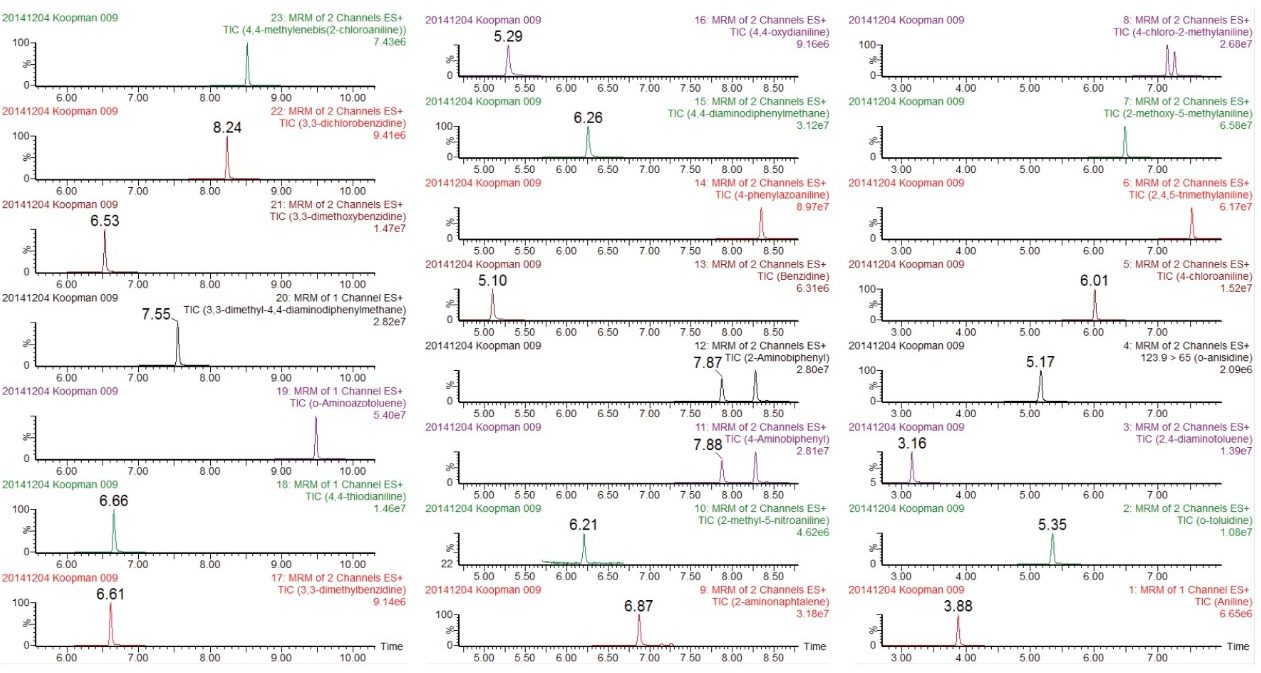
Two MRM transitions were used, unless otherwise stated. The dwell times were chosen automatically using the built-in points-per-peak calculator in the MS method. The data were acquired using MassLynx v4.1 Software, and processed using TargetLynx XS Application Manager. Table 1 summarizes all MRM transitions. Figure 2 shows the retention time windows of the MRM method.
A mixed standard solution containing all PAAs at a concentration of 100 µg/mL was used. The working standards were further diluted with the 3% acetic acid food stimulant solution. For the solvent calibration a dilution series starting at 100 ng/mL down to a level of 0.78 ng/mL was made.
Nine polyamide kitchenware utensils were extracted with a 3% acetic acid solution according to the procedure described in the EU 10/2011 guidelines.1
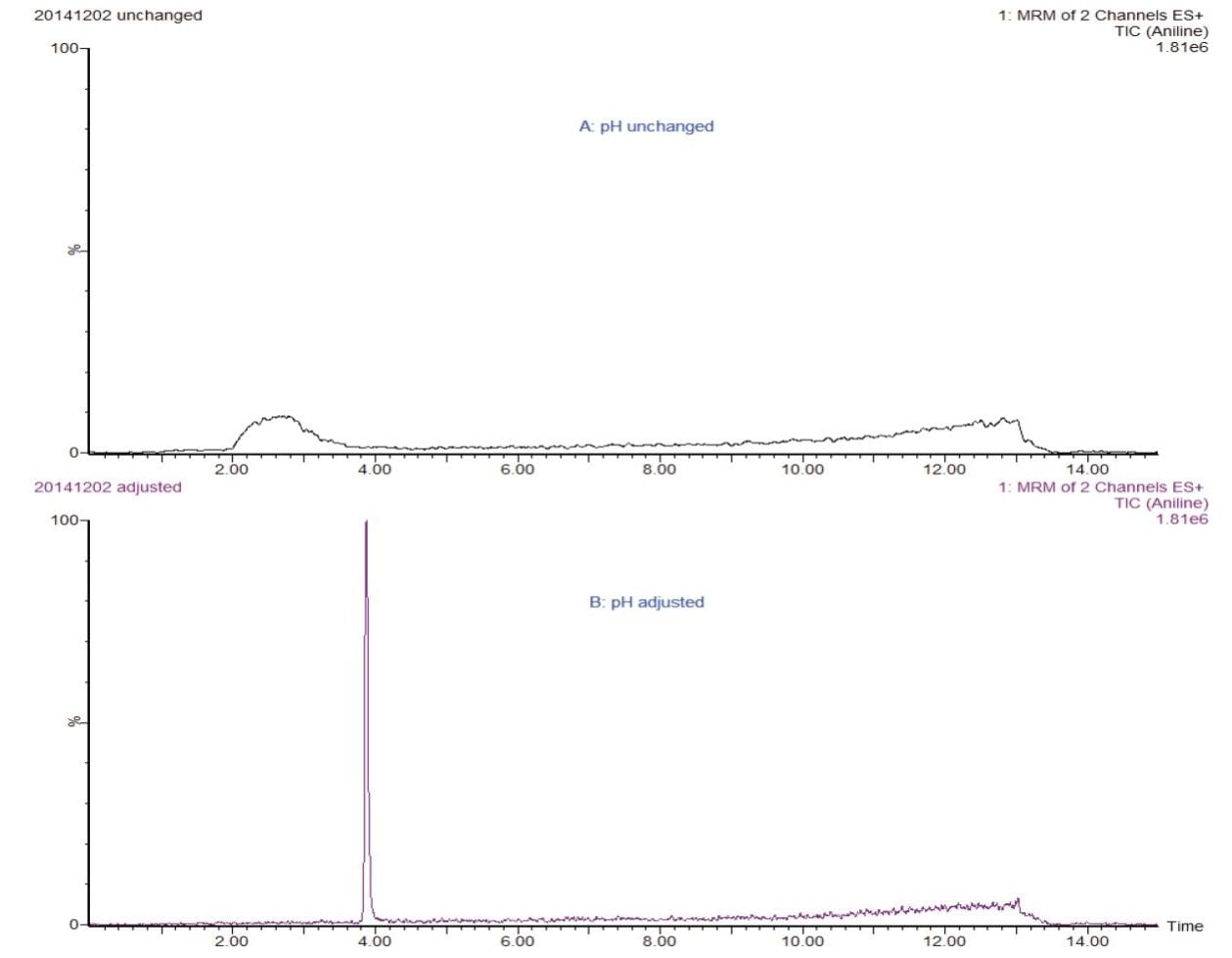
Because of the basic properties of PAAs, and the fact that acetic acid is used as a migration stimulant, some PAAs don’t focus well on the head of the column, resulting in poor peak shape and/or loss of retention. Aniline elutes early and is therefore prone to this effect. As a result, some literature references cite the use of ion-pair reagents.4 Adding ammmonium hydroxide to the 3% acetic acid samples prior to injection, the pH of the sample is increased and the polar and weakly basic PAAs such as aniline will be in their neutral form. A volume of 10 µL of a 25% NH4OH solution was added to 1 mL of sample. This approach resulted in more robust results and is therefore preferred over the use of ion-pair reagent. Figure 3 shows a chromatogram of aniline with an unchanged pH (top) and adjusted pH (bottom). The neutralization of the pH drastically improves the peak shape of aniline, without the need for ion-pairing reagent.
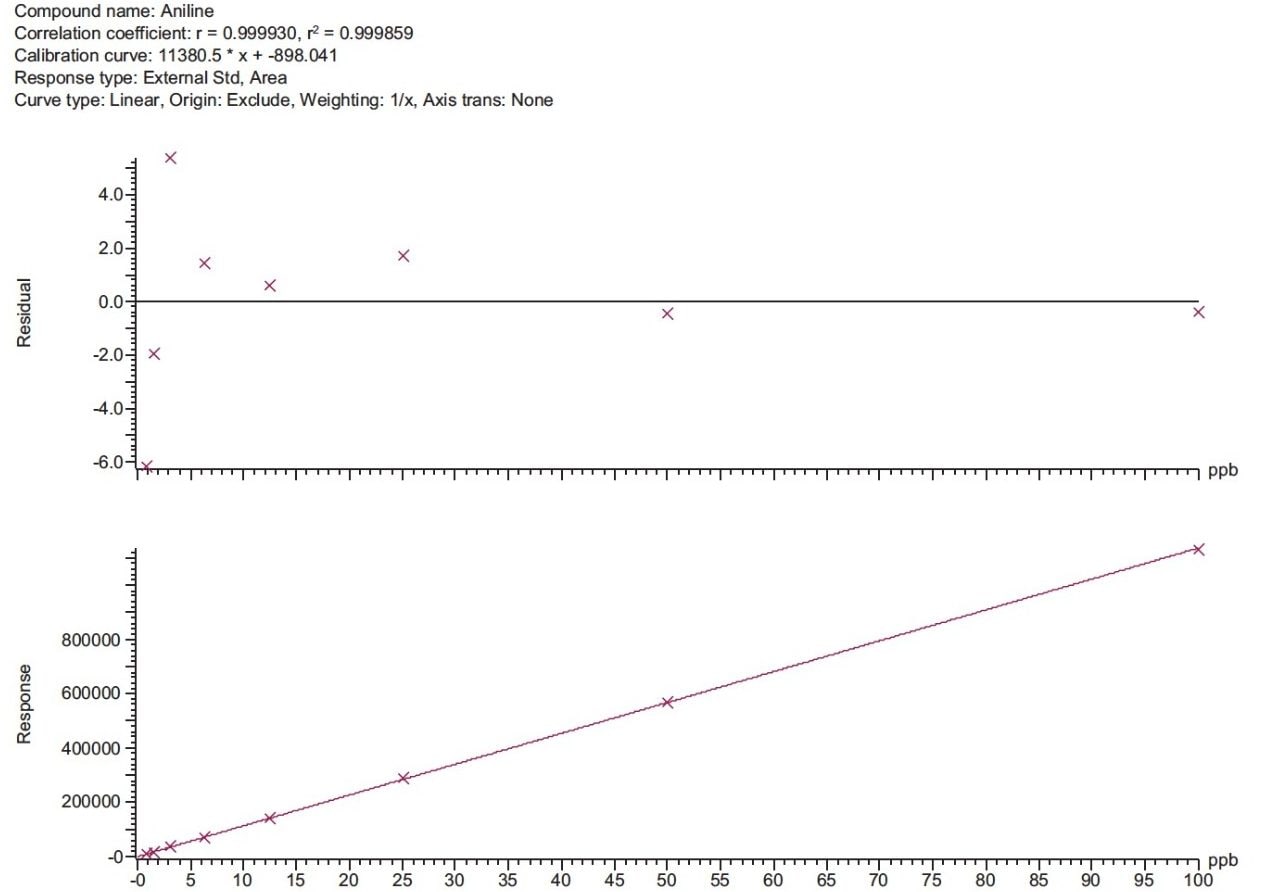
Calibration curves were prepared from 0.78 ng/mL to 100 ng/mL for all compounds. An example is given for aniline (Figure 5). For each calibration curve, a linear regression and a 1/X weighting was applied. All compounds show good linearity across the range of concentrations as well as excellent % residual values.
Acidified mobile phases aid in the protonation of compounds and therefore improve the sensitivity in positive ion electrospray. As no acid was added to the mobile phases, we investigated whether a post-column addition (PCA) with formic acid would be beneficial. Using the Xevo TQ-S micro’s built-in IntelliStart fluidics, a solution of 2% formic acid was infused at a constant flow rate of 20 µL/min into the UPLC flow exiting the column. As such the formic acid solution was diluted 20-fold with the mobile phase, resulting in a final concentration of 0.1% of formic acid going into the ESI source. Figure 6 shows how this PCA was configured in the acquisition method, while Figure 7 shows the chromatograms for a selection of PAAs with (top trace) and without (bottom trace) this post-column addition. For better interpretation, the intensity axes have been linked. As can be seen from the chromatograms, the sensitivity is significantly improved when formic acid is added to the eluent.
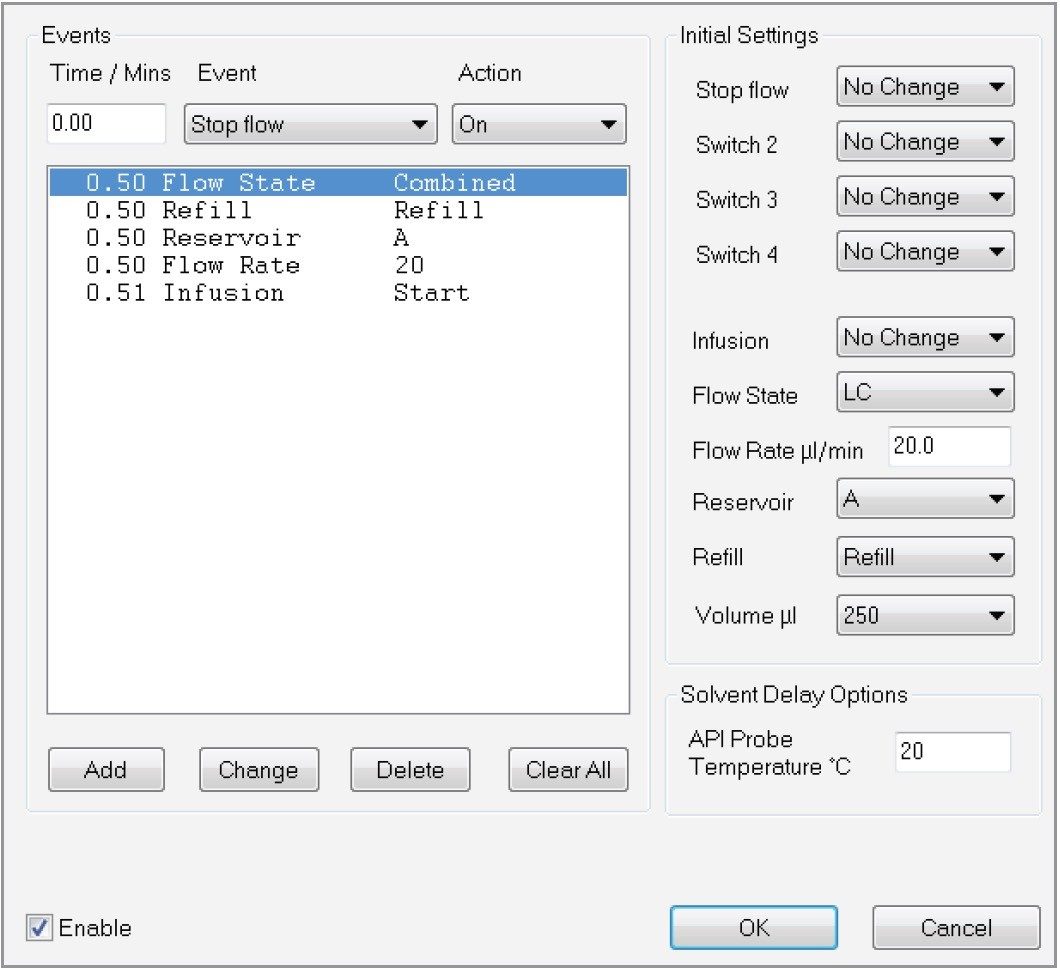
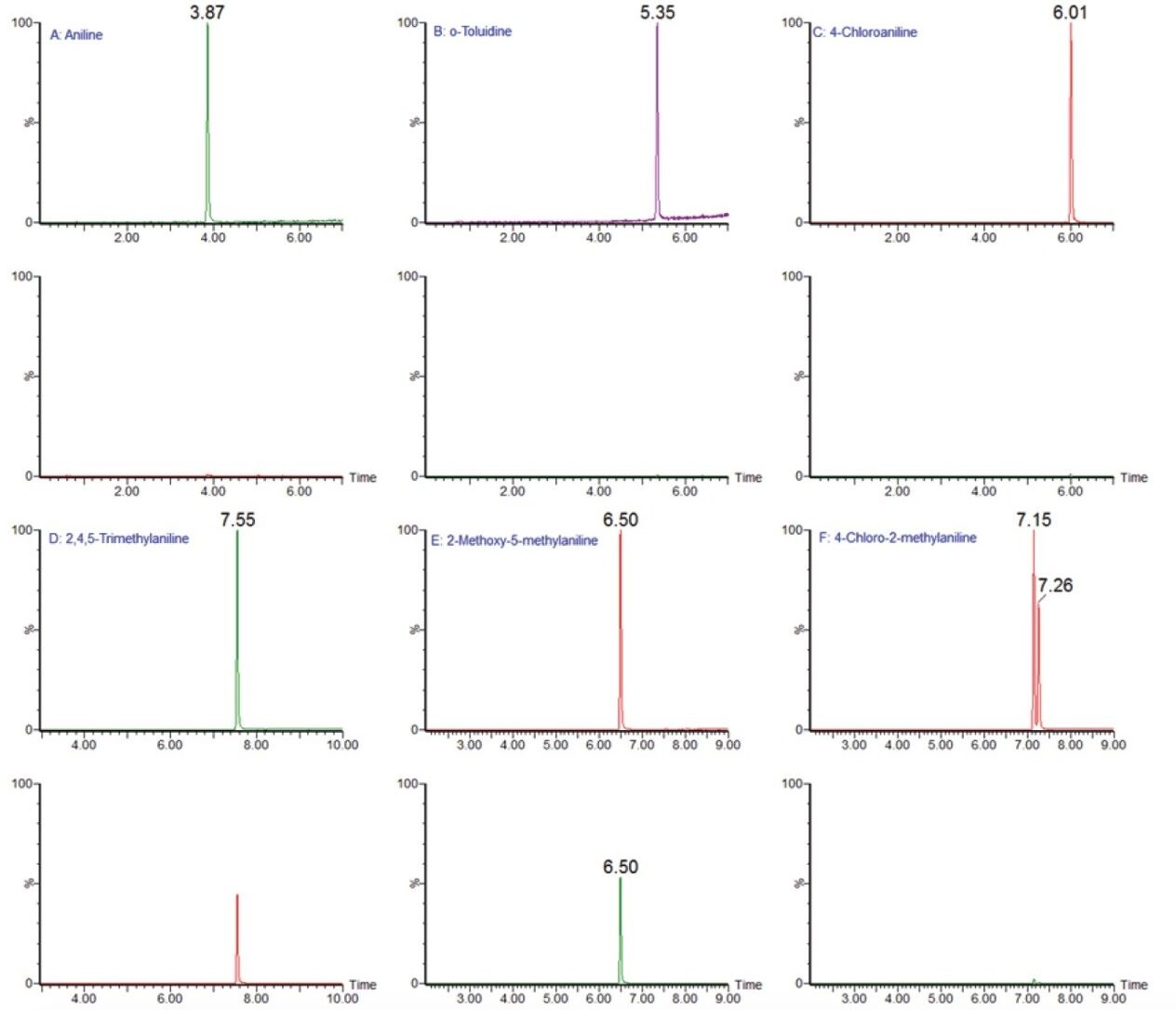
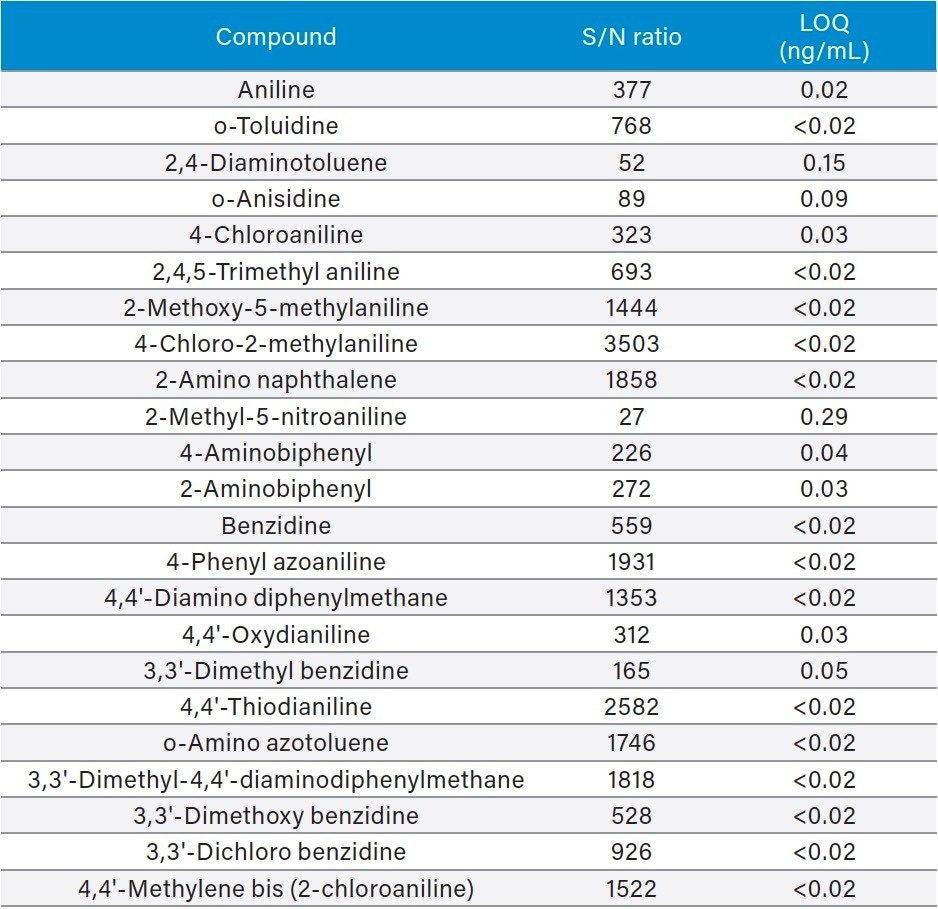
Table 2 summarizes the quantitation limits (LOQ) for all compounds using this PCA approach. The LOQ is defined as the concentration giving rise to a signal-to-noise (S/N) value of 10:1. For the calculation of S/N, raw data was used and the peak-to-peak algorithm was applied. An extrapolation was made in most cases, as the reported S/N values were still significantly high, even at the lowest reported standard level of 0.78 ng/mL. Calculated LOQs below 20 pg/mL are not mentioned specifically but are cut off at this level. The reported LOQ concentrations range between 20 pg/mL and 300 pg/mL.
Internal standards were not used in this method. Therefore it was investigated whether the food simulant extract leads to ion suppression. One of the samples was spiked to a final concentration of 10 ppb and this sample was compared with a standard dissolved in the same food stimulant solution. All spike recoveries were within 90% to 107%, indicating that matrix effects were low to non-existing for the 23 compounds under investigation.
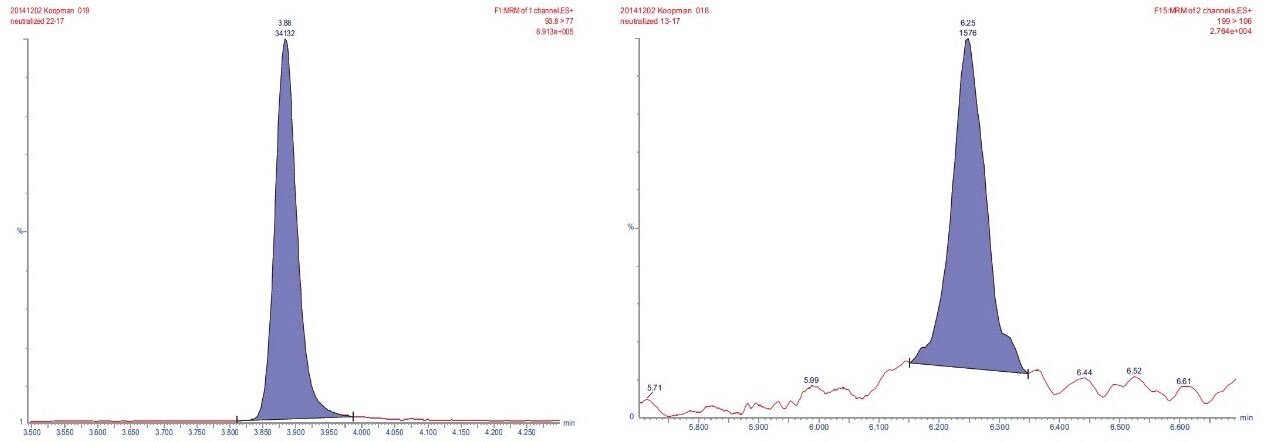
Using the external calibration curves, nine kitchenware samples were quantified. Except for aniline and 4,4'-diamino diphenylmethane found in all nine samples at levels between 0.4 to 1.1 ppb and 0.04 to 0.11 ppb, respectively, no other PAAs were detected. Figure 8 shows the chromatograms of aniline in the sample containing 0.4 ppb and of 4,4'-diamino diphenylmethane in the sample containing 0.04 ppb. As can be seen sensitivity was excellent at these sub ppb level.
We have demonstrated a sensitive method for 23 PAAs with very easy sample preparation. The addition of ammonium hydroxide as neutralizing agent, and a post-column addition of formic acid into the Xevo TQ-S micro via IntelliStart’s built-in fluidics – resulted in a very sensitive assay which could reach sub ppb levels. Linearity was observed over a large range and up to 100 ppb. The samples were all below detection limits except for aniline which was detected at 0.4 to 1.1 ppb, and 4,4'-diamino diphenylmethane which was detected at 0.04 to 0.11 ppb. The migration of all PAAs were compliant with the individual and summed SML (SML(T)) limits, as stipulated in Regulation EU 2020/1245.
720005781, Revised November 2020