
In this application note, a method transfer from another manufacturer’s HPLC System to an ACQUITY Arc system was performed using the Path 1 of the Arc Multi-flow path technology.
This flow path, which was designed for methods transfer, allows the ACQUITY Arc System to mimic the dwell volume of a typical HPLC system. The results produced retention times within 5% of the original method. Additional fine-tuning of retention times was accomplished using Gradient SmartStart Technology, which adjusts the initial hold of the method without requiring any changes to the gradient table. The combination of these two features enables methods transfer to an ACQUITY Arc System in just two injections for a simplified methods transfer approach, including the following:
Sugars and sugar alcohols are classes of carbohydrates that are natural constituents of foods and provide important nutritional benefits. Some sugars are added to processed foods in order to enhance flavor or to mimic fresh food products. With the increasing incidence of obesity and diabetes across the developed world, the need to monitor sugar intake has grown in recent years. Consequently, there are now requirements to provide accurate information about sugar content on food product labels in order to comply with increasingly stringent regulatory demands.
The analysis of these compounds is challenging because they lack chromophores within their compound structures, and because of the close similarity among the various molecules, many of which are simple isomers of one another. Structures and formulae are shown in Figure 1. Due to its separation power, accuracy and speed of analysis, HPLC has become the method of choice for the analysis of sugars.1 HPLC techniques typically employ RI or ELS detection. RI detection requires careful control of the mobile phase to avoid any changes during the analysis and therefore requires isocratic elution. With RI detection it is also difficult to change the mobile phase composition from one analysis to the next because the RI detector may require several hours to equilibrate when a different mobile phase composition is introduced. ELS detection is more robust for mobile phase composition changes, but ELS often does not meet the sensitivity demands for the detection of sugars in complex matrices.
An alternative gaining traction is the use of mass detection with electrospray ionization (ESI). Waters ACQUITY QDa Detector offers the opportunity to decrease detection limits as well as the ability to obtain mass spectral information on components in the sample. The combination of chromatographic retention time and mass information can provide improved selectivity for the profiling of sugars and sugar alcohols. The ACQUITY QDa Detector is the only mass detector that has been holistically designed to be incorporated with an LC system. It fits in the LC stack, occupying the same amount of space as a PDA detector. Extensive training is not required, so users already familiar with HPLC can quickly take advantage of the improved selectivity and sensitivity that mass detection affords. In this application note we describe the use of the ACQUITY QDa Detector coupled to the ACQUITY Arc System for the profiling of sugars in milk and infant formula.

A 50 mg/L stock of the seven saccharides listed above was prepared in 1:1 acetonitrile-water. This stock was further diluted to produce nine individual levels (0.5, 1, 2, 2.5, 5, 10, 20, 25, and 50 mg/L).
Samples of a non-fat dry milk powder, a dairy-based infant formula, a soy based infant formula, and a low fat milk were purchased. These were prepared based on the procedure described by Chavez-Servin et al2 as follows:
Add approximately 0.6 g sample to a 25 mL volumetric flask.
*Carrez 1 reagent: dissolve 0.36 g K4[Fe(CN6).3H20 in 10 mL water.
**Carrez 2 reagent: dissolve 0.72 g ZnSO4.7H2O in 10 mL water.
The supernatant was diluted with 1:1 water:acetonitrile, 1:500 for the analysis of sucrose and lactose, and 1:20 for the analysis of fructose, glucose, galactose and inositol. The initial dilution of 25 was included for the final quantification calculations.
|
LC system: |
ACQUITY Arc Path 1 |
|
Run time: |
30.0 min |
|
Column: |
XBridge BEH Amide XP 2.5 μm, 3.0 × 150 mm |
|
Column temp.: |
85 °C |
|
Mobile phase: |
90:5:5 Acetonitrilewater-2-propanol with 0.05 % diethylamine and 500 ppb guanidine hydrochloride |
|
Flow rate: |
0.8 mL/min |
|
Injection volume: |
1 μL |
|
MS system: |
ACQUITY QDa (Performance) |
|
Ionization mode: |
ESI Capillary |
|
Voltage: |
0.8 V |
|
Cone voltage: |
5.0 V |
|
Probe temp.: |
600 °C |
|
Acquisition rate: |
2 Hz |
|
Full scan: |
100 to 500 m/z |
|
Curve fit: |
Quadratic, 1/x weighting |
|
Smoothing: |
Mean, Level 7 |
|
SIR {M+Cl-}-: |
215.0 fructose, glucose, galactose, inositol 377.0 sucrose, lactose, maltose |
Figure 2 shows the ACQUITY Arc System with the ACQUITY QDa and PDA detectors. Although shown with PDA, UV detection was not used for this analysis. Figure 3 shows the SIR chromatograms of the seven saccharide standards at a 5 ppm level (standard 5) used in the study. The annotated m/z (215 and 377) represent the [m+ Cl]- adducts. Figure 4 shows the mass spectra for the analytes. Addition of guanidine hydrochloride to the mobile phase shifts the equilibrium to the chloride adducts, m/z 215 for the monosaccharides and inositol, and 377 m/z for the disaccharides. Note the baseline separation of galactose and glucose using this chemistry, and also that lactose eluted before maltose.
![SIR chromatograms of the seven saccharide standards used in the study, the annotated m/z represents the [m+ Cl]- adducts.](/content/dam/waters/en/app-notes/2016/720005767/720005767en-f3.jpg.82.resize/img.jpg)
![Mass spectral information extracted from SIRs of the seven saccharide standards. The annotated m/z represents m/z the [m+ Cl]- adducts.](/content/dam/waters/en/app-notes/2016/720005767/720005767en-f4.jpg.82.resize/img.jpg)
Figure 5 shows the SIR chromatograms of the saccharide standards at 5 ppm (standard 5) along with the dairy and soy based infant formulas at m/z 215. Note the absence of galactose in the soy based formula as would be expected. However, inositol is present in both formulations as it is an important nutrient in infant formula.3 Inositol is highlighted in Figure 6.
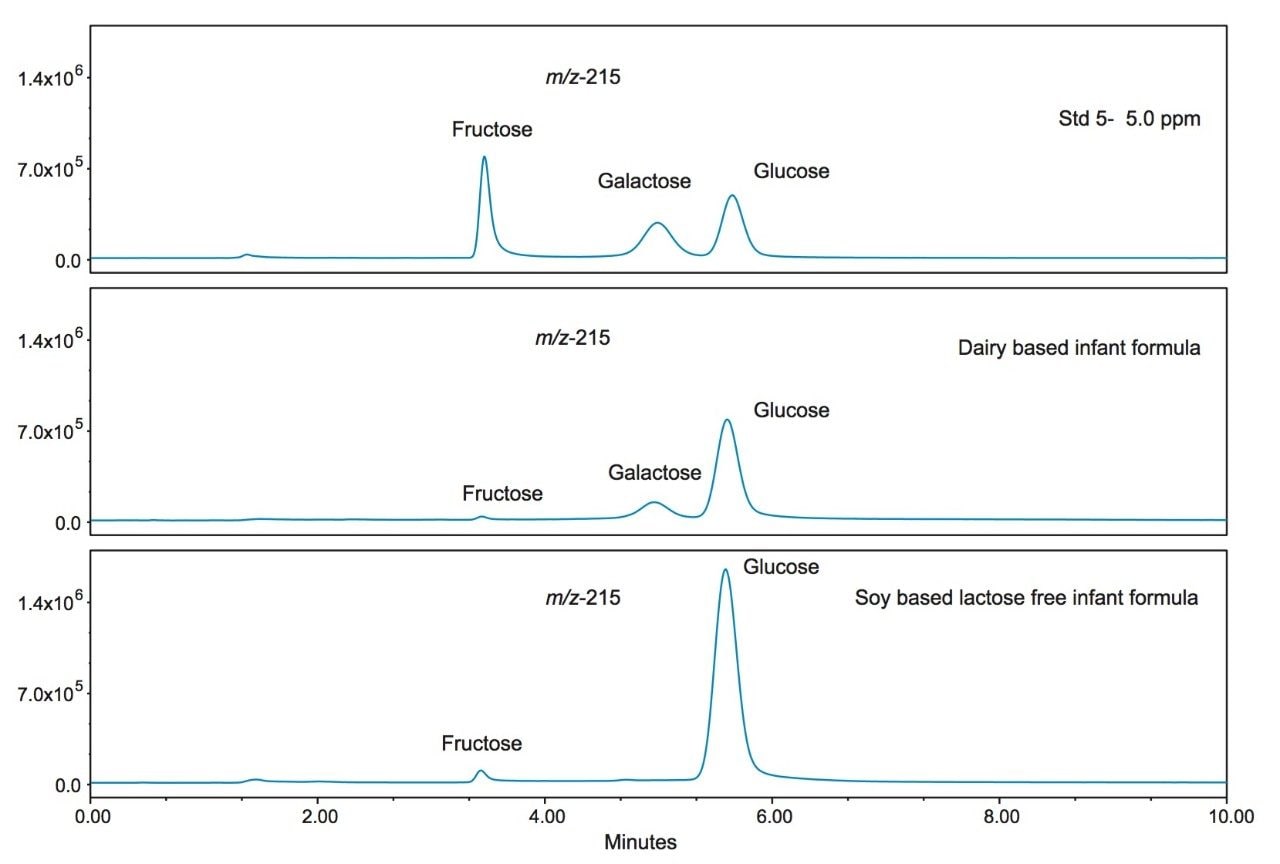
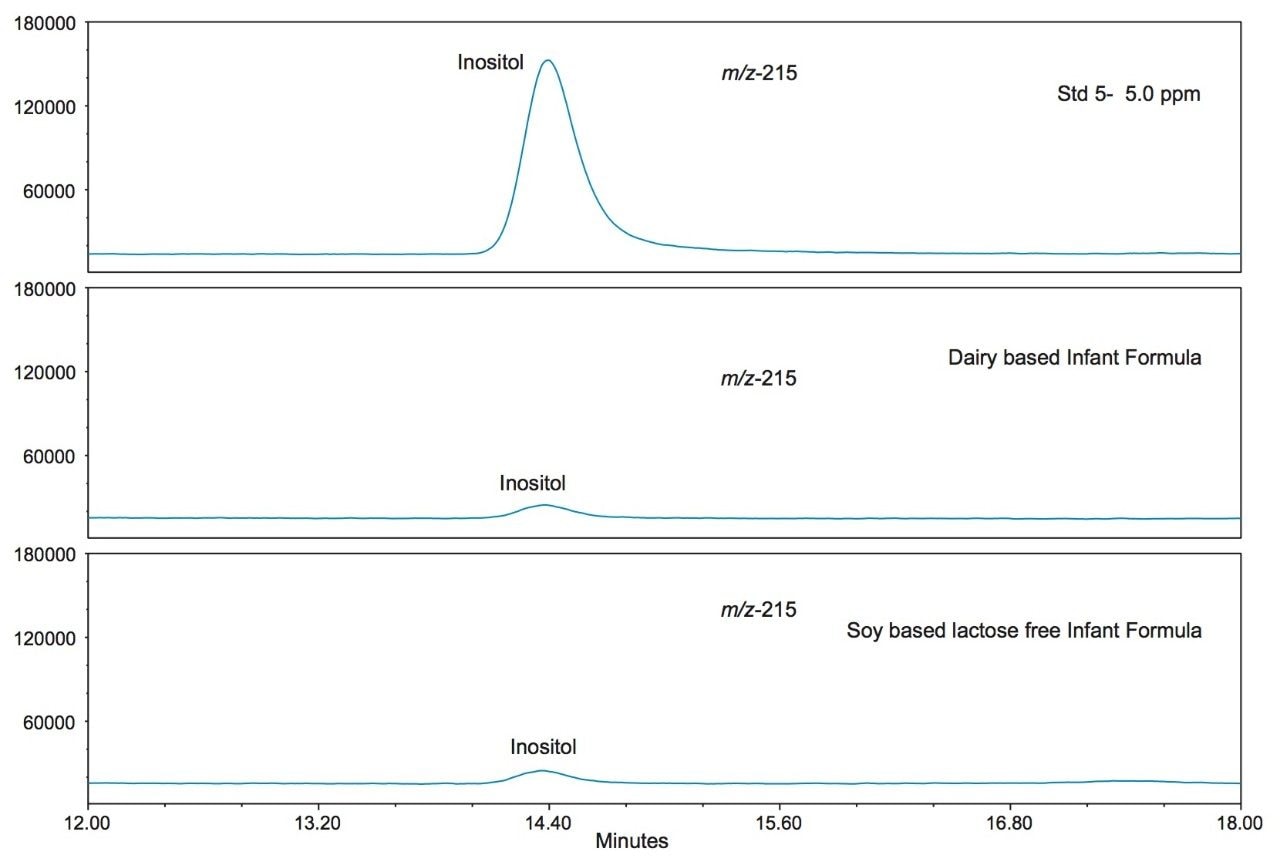
Figure 7 shows the SIR chromatograms of the saccharide standards at 5 ppm (standard 5), along with the dairy and soy-based infant formulas at m/z 377. Here we see the absence of lactose in the soy formulation. However sucrose and maltose are present, which are absent in the dairy formulation. This is also to be expected as these two sugars are derived from plant based sources and should not be found in a dairy matrix unless added artificially. Figure 8 shows the calibration curves for the analytes. The regression coefficient (R2) was >0.998 for all analytes.
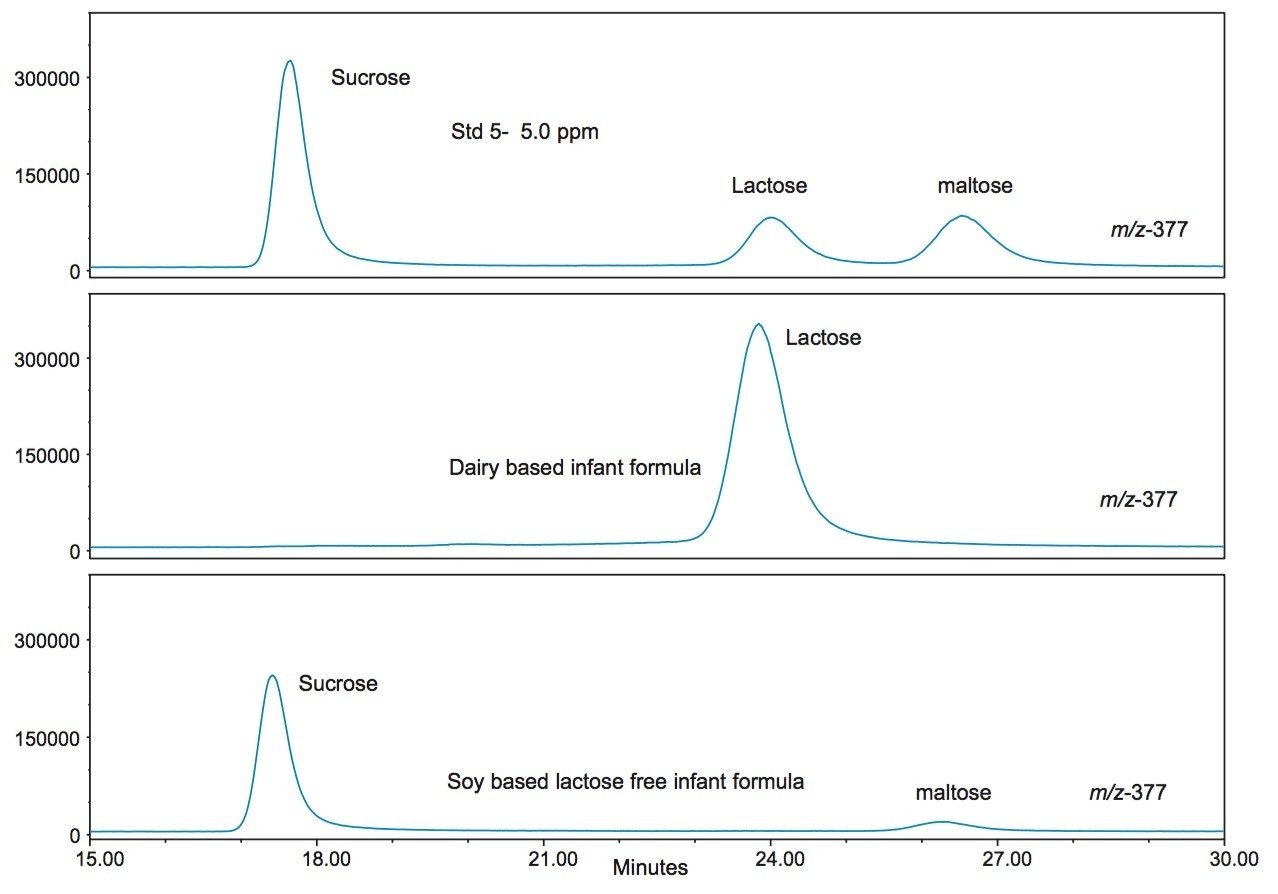
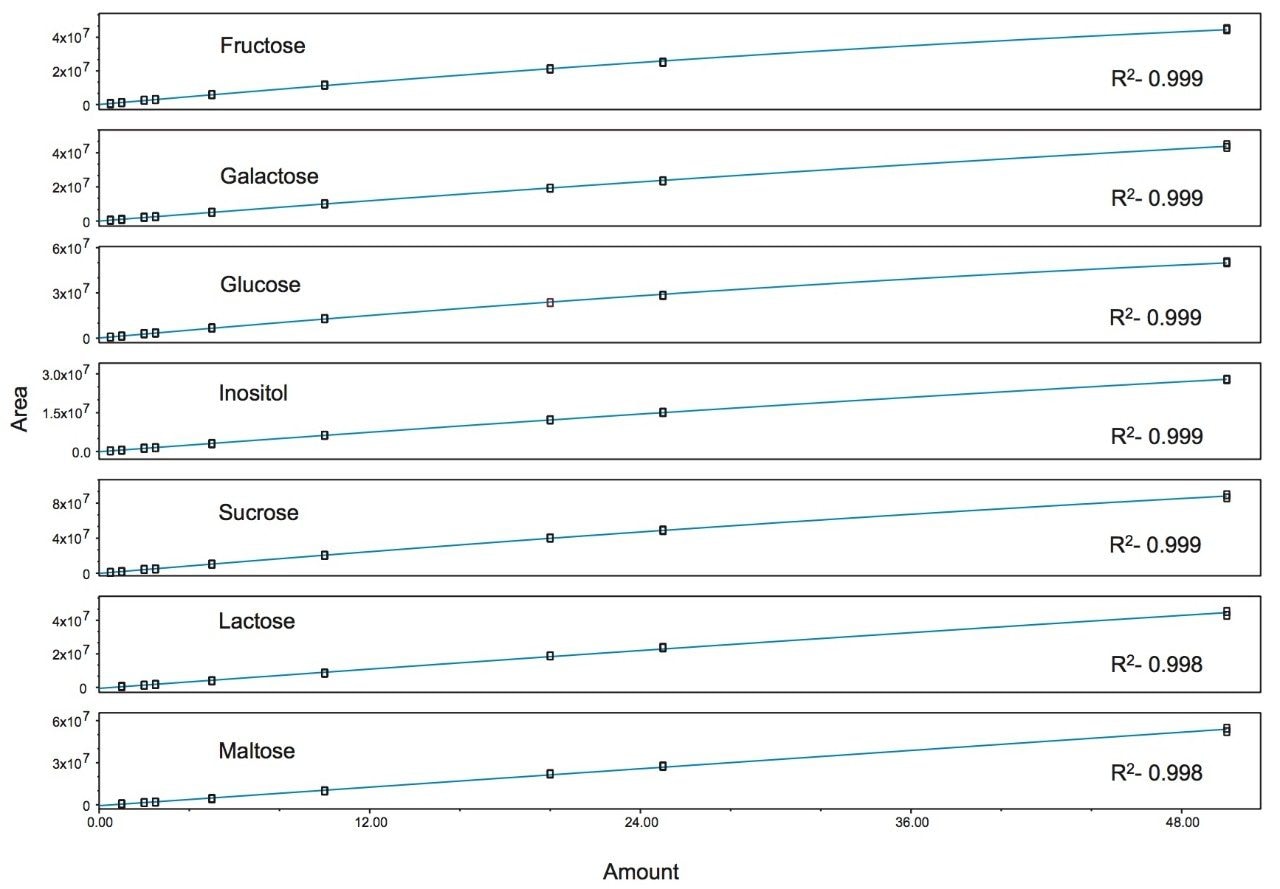
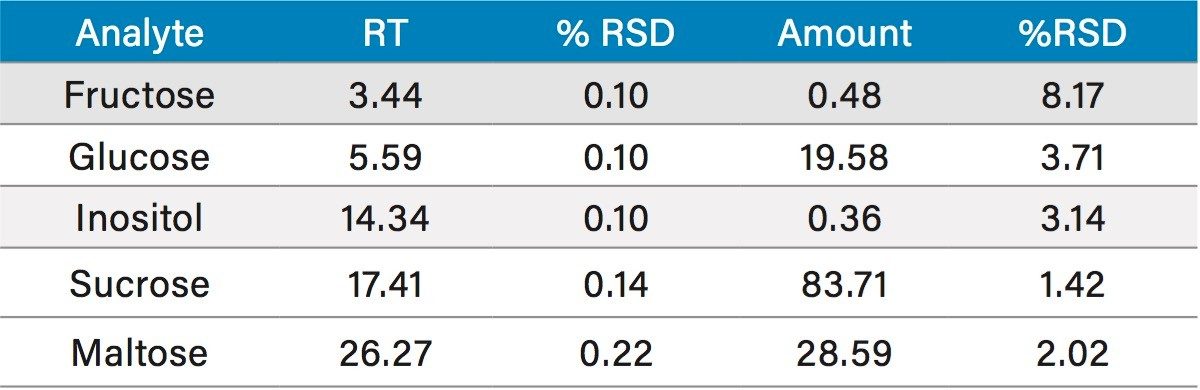
Table 1 lists the quantitated values for the saccharides studied. The values for inositol in the dairy and soy based infant formula generally agree with the values reported by Indyk.4 Likewise, the value for lactose in the dairy infant formula generally agrees with that reported by Ferreira.5 Table 2 lists reproducibility data for 7 injections of the soy based infant formula. RSD’s were <0.25% for retention time and 8.20% for amount.

The analysis of carbohydrates in dairy products can be challenging because of the mix of closely related UV transparent compounds. The combination of the ACQUITY Arc System, ACQUITY QDa Detector, and the XBridge BEH Amide Column offers scientists the advanced performance expected of ACQUITY separations, high resolution, sensitivity, and improved throughput, along with a complimentary mass detector to RI and ELS that provides the additional advantages of:
1. IMPLVO Ferreira, et al. Determination of Sugars and Some Other Compounds in Infant Formula, Follow Up Milks and Human Milk by HPLC-UV/RI. Carbohydr Polym. 37: (3) 225-229, 1998.
2. JL Chavez-Servin et al Analysis of Mono and Di-Saccarides in Milk Based Formulae by High Performance Liquid Chromatography with Refractive Index Detection. J Chrom A. 1043: (2) 211-215, 2004.
3. HE Indyk and DC Woolard. Determination of Free Myo-Inositol in Milk and Infant Formula by High Performance Liquid Chromatography. Analyst. 119: 397-402, 1994.
4. Ibid p 401.
5. Ferreira, Op. Cit. p 227.
720005767, July 2016