
In this study, ionKey/MS Systems, which integrates the UPLC analytical separation directly into the source of the MS, is used for the accurate, precise, and reproducible analysis of bradykinin. Use of the ionKey/MS System, mixed-mode SPE and higher m/z b or y ion MS fragments provides the level of selectivity and sensitivity necessary to accurately quantify bradykinin and distinguish subtle differences in concentrations.
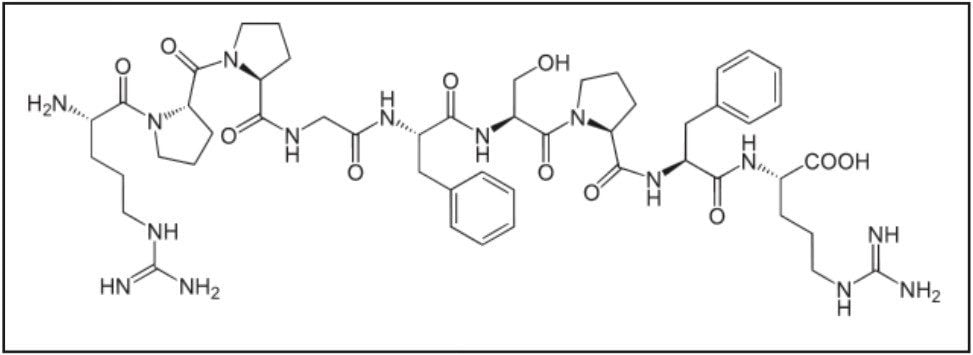
The need for robust and sensitive analysis of peptide species challenges both chromatographic separation and mass spectrometry. Peptides, in general, are often difficult to analyze by LC-MS/MS, as mass spectrometer (MS) sensitivity is low due to the formation of multiple precursors and poor or overly extensive fragmentation, making liquid chromatography (LC) and sample preparation even more critical. A previous application note (720004833EN) described in detail the development of a fast, flexible analytical scale, SPE-LC-MS/MS method for the quantification of the peptide bradykinin (Figure 1) in human plasma for use as a biomarker in the preclinical or discovery setting.1 Accurate quantification of bradykinin in plasma is particularly challenging because it is present in low pg/mL levels, is rapidly metabolized, and is also artificially produced during blood sampling and sample preparation via proteolytic processes.2
In this work, the LC-MS platform was updated to incorporate the use of the ionKey/MS System which integrates the UPLC analytical separation directly into the source of the MS (Figure 2). The iKey Separation Device (150 µm I.D.), shown in Figure 3, contains the fluidic channel, electronics, ESI interface, heater, eCord, and the chemistry to perform UPLC separations. Additionally, this technology offers significant increases in sensitivity compared to 2.1 mm I.D. chromatography, making it ideal for peptide analyses. Most bioanalytical LC-MS/MS assays often consume high volumes of both solvent and sample, thus increasing the cost of the assay and limiting the number of replicates that can be analyzed. In addition to the sensitivity increase the ionKey/MS System provides over the 2.1 mm diameter scale, it also reduces solvent and sample consumption and provides enough sample to perform multiple injections that may be required to meet incurred sample reanalysis (ISR) guidelines.


|
UPLC conditions |
|
|---|---|
|
LC system: |
ACQUITY UPLC M-Class with 2D Technology configured with optional trap and back flush elution |
|
Separation device: |
Key Peptide BEH C18 Separation Device, 300Å, 1.7 μm, 150 μm x 50 mm (p/n 186006969) |
|
Trap column: |
ACQUITY UPLC M-Class Symmetry C18, 5 μm, 300 μm x 50 mm (p/n 186007498) |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
0.1% formic acid in acetonitrile |
|
Loading solvent: |
99:1 mobile phase A:B, 25 μL/min for first two minutes, reverse valve |
|
Valve position: |
Initial position one (forward loading of trap), switch to position two at two minutes (back flush elute of trap onto the analytical column) |
|
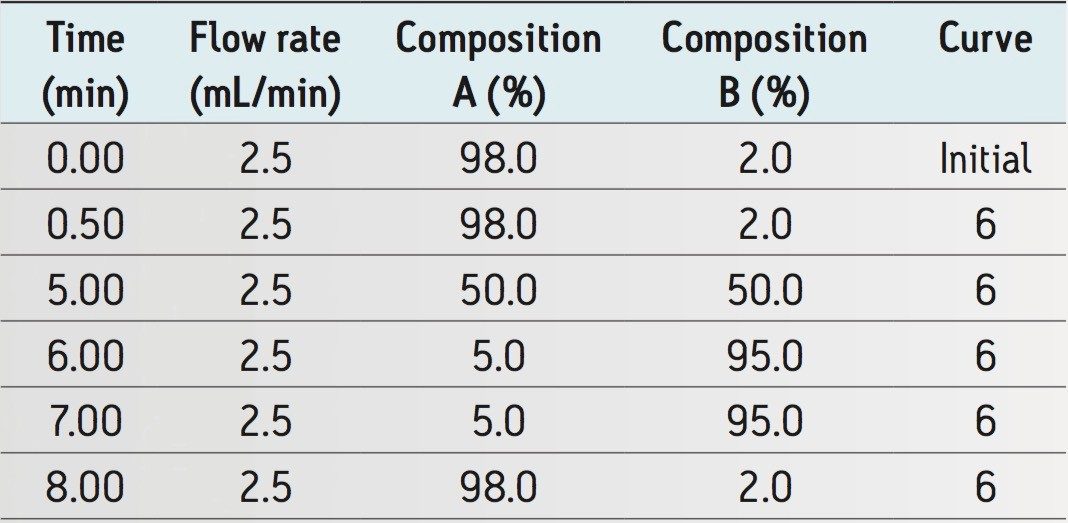
Analytical gradient: |
See Table 1 |
|
Elution flow rate: |
2.5 μL/min |
|
iKey temp.: |
75 °C |
|
Sample temp.: |
15 °C |
|
Injection vol.: |
10 μL |
|
Total run time: |
12.0 minutes |
|
Collection plates: |
Waters 1 mL collection plates (p/n 186002481) |
|
MS system: |
Xevo TQ-S Mass Spectrometer with ionKey Source and iKey Seperation Device |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
3.8 kV |
|
Source temp.: |
120 °C |
|
Cone gas flow: |
50 L/hr |
|
Collision cell pressure: |
3.83 x 10(-3) mbar |
|
Collision energy: |
Optimized by component, see Table 2 |
|
Cone voltage: |
Optimized by component, see Table 2 |
|
Chromatography software: |
MassLynx 4.1 |
|
Quantification software: |
TargetLynx |
This study utilizes specifically designed blood collection techniques to inhibit bradykinin formation ex vivo , takes advantage of mixed mode solid-phase extraction (SPE) and use of the novel and highly efficient ionKey/MS System for selective, sensitive, and robust chromatographic separation, and quantification of the nonopeptide bradykinin. The sensitivity increase that ionKey/MS System provides over the 2.1 mm diameter scale method for bradykinin enables a 2x reduction in plasma and a 7–10x increase in signal-to-noise (S:N). As a result, we can accurately and precisely quantify 2.5 pg/mL of bradykinin above the basal level.
Human plasma was obtained from one male donor whose blood was collected in BD P100, P700, P800, and blood collection tubes containing only K2EDTA. The various BD P blood collection tubes contain various mixtures of proprietary stabilizers/inhibitors that immediately solubilize during blood collection, and enable preservation of human plasma proteins and peptides.
10 μL of the internal standard (IS), [Lys-des-Arg9]-bradykinin (5 ng/mL) was added to 100 μL of human plasma and mixed. The samples were then diluted 1:1 with 5% NH4OH in water and mixed.
Pretreated plasma samples were extracted according to the protocol in Figure 4. All solutions are made up by volume. All extraction steps were applied to all wells of the Oasis WCX 96-well μElution Plate that contained samples.

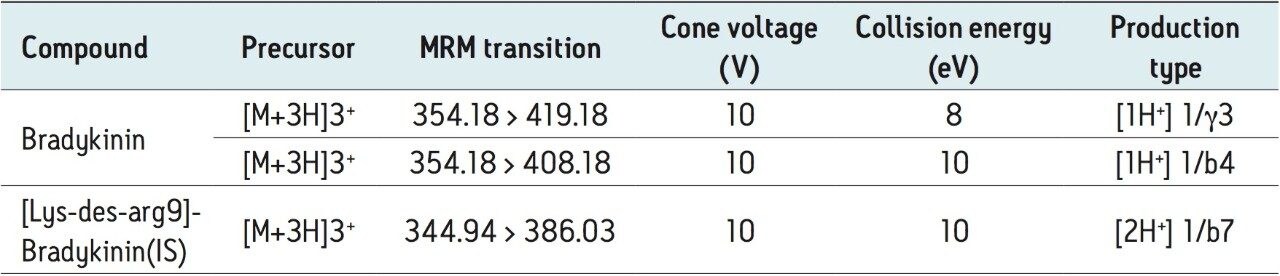
The 3+ precursors of bradykinin (m/z 354.18) and IS (344.94) were used for quantitation. The fragment at m/z 419.18 y31+ was chosen as the primary fragment for bradykinin quantitative analysis, while the m/z 408.18 b41+ fragment was used for confirmatory purposes. For the IS, the fragment at m/z 386.03 b72+ was chosen. Optimal MS conditions are shown in Table 2. Although many peptides produce intense fragments below m/z 200, these ions (often immonium ions) result in high background in extracted samples due to their lack of specificity. In this assay, the use of highly specific b or y ion fragments with m/z values higher than their precursors yielded significantly improved specificity, facilitating the use of simpler LC and SPE methodologies.
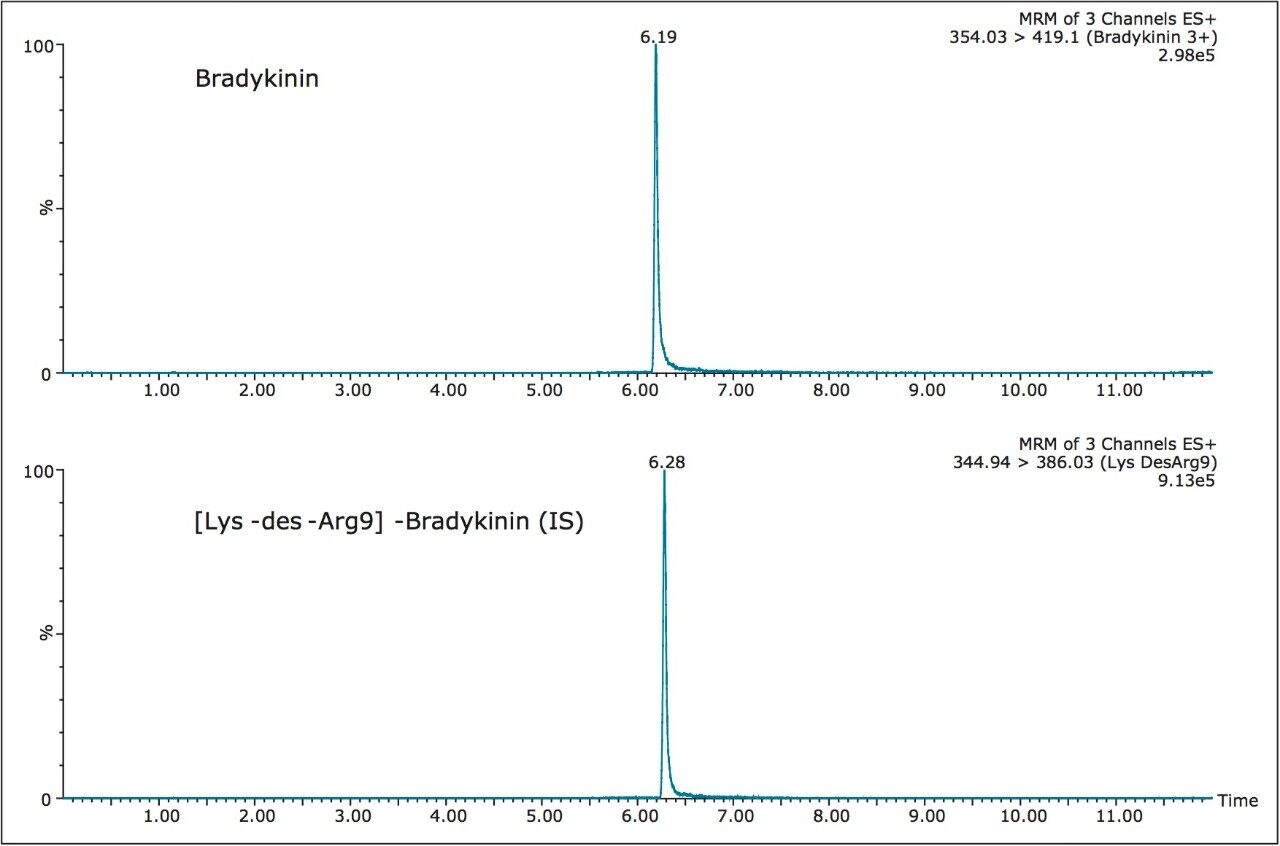
Chromatographic separation of bradykinin and its IS was achieved using the novel microfluidic chromatographic iKey Separation Device. The iKey Separation Device has a channel with UPLC-grade, sub-2-µm particles that permits operation at high pressure and results in highly efficient LC separations. By integrating microscale LC components into a single platform design, problems associated with capillary connections, including manual variability, leaks, and excessive dead volume are avoided. Use of the iKey Peptide BEH C18 Separation Device, 300Å, 1.7 µm, 150 µm x 50 mm (p/n 186006969) provided excellent peak shape, increased peak height, and improved S:N compared to the analytical scale (2.1 mm I.D.) LC-MS analysis. Representative chromatograms of bradykinin and the IS using the iKey Separation Device are shown in Figure 5. The use of multidimensional chromatography, specifically a trap and back-elute strategy, provided further sample cleanup and facilitated the loading of 10 μL of the high organic SPE eluate (required to maintain solubility of the peptides) without experiencing analyte break through. Additionally, the ability to inject the larger sample volumes typical for analytical scale LC analysis (e.g. 10 μL) on the iKey Separation Device can provide the substantial gains in sensitivity that are often required to accurately and reliably detect low pg/mL levels of peptide and protein in complex matrices.
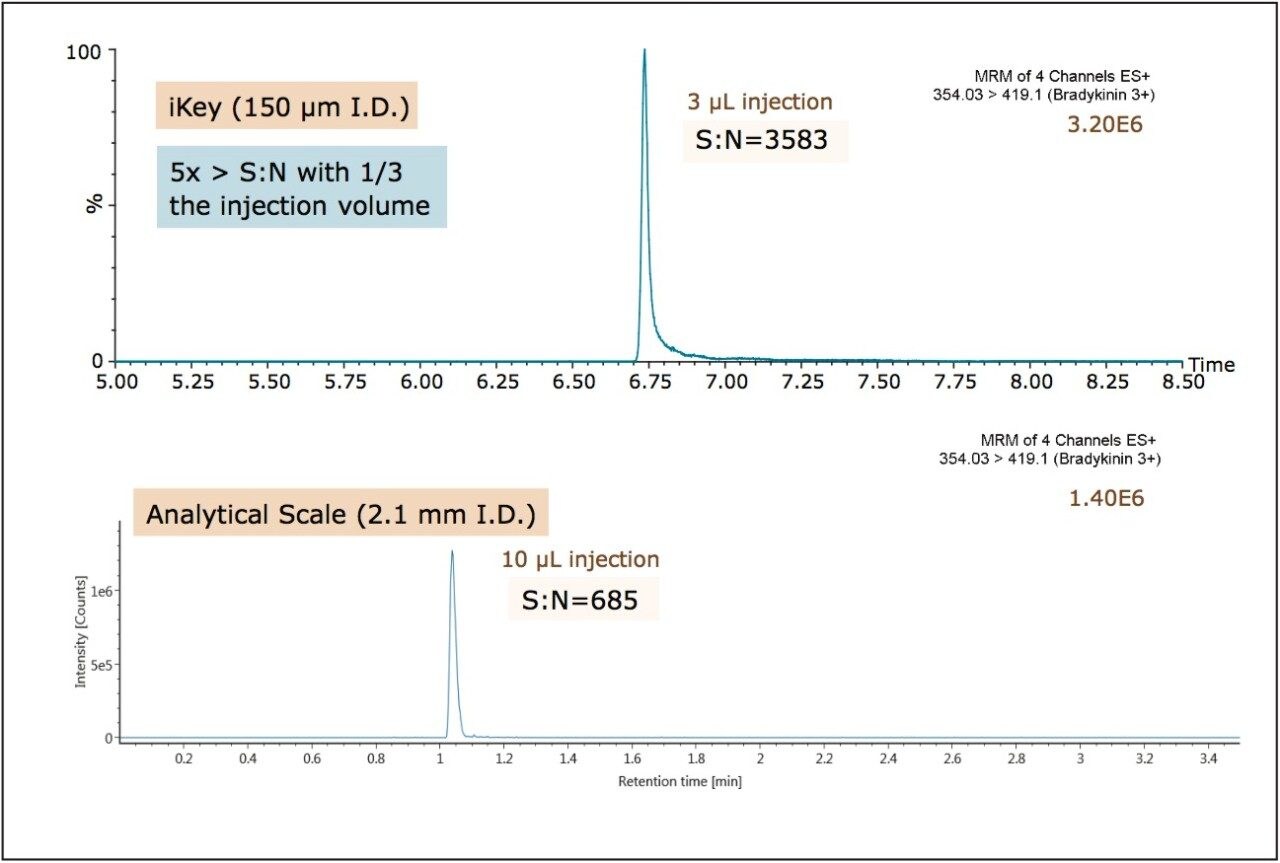
Use of the ionKey/MS System facilitated the development of a highly efficient LC separation of bradykinin in plasma with significant improvement in sensitivity and S:N over the analytical scale LC-MS using 2.1 mm I.D. chromatography. Initially, samples were extracted using the protocol described in the previous application note (720004833EN). Briefly, 200 μL of plasma was extracted followed by a 1:1 dilution of the eluate with water. A 3 μL injection of this sample on the iKey Separation Device provided a 5x improvement in S:N compared to a 10 μL injection of the same sample analyzed at the 2.1 mm scale, and is shown in Figure 6. The improvement in ionization efficiency and subsequent increase in sensitivity afforded by the iKey Separation Device facilitated this lower injection volume. The ability to obtain comparable or improved sensitivity with smaller injection volumes (1–3 μL) using the ionKey/MS System makes this technology ideal when sample is limited or when multiple injections are required to meet ISR guidelines.
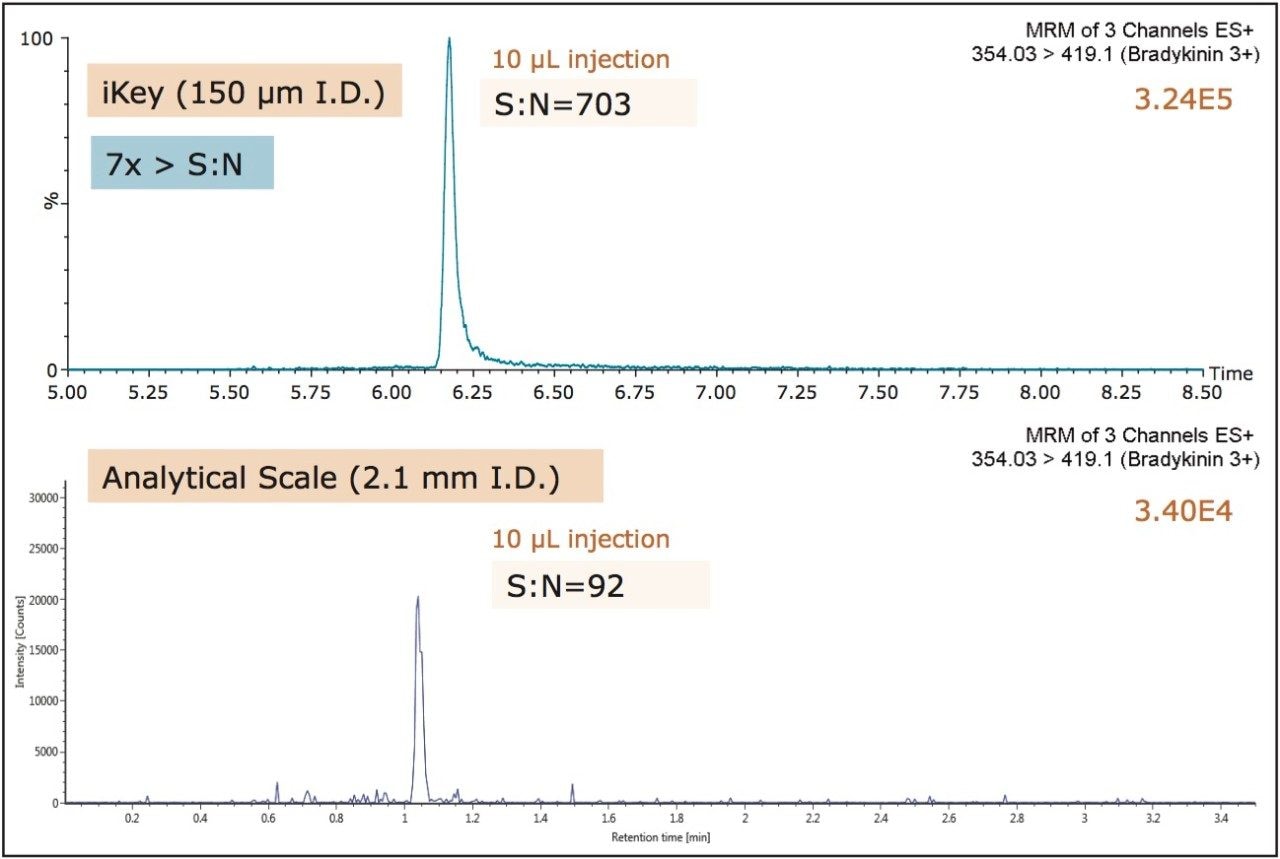
Method optimization resulted in the reduction of the required plasma sample by half and an increase in eluate dilution to 1:2, both of which minimized matrix interferences. A comparison of a 10 μL injection of extracted plasma (using the optimized method) the ionKey/MS System and a traditional analytical flow system (ACQUITY UPLC and Xevo TQ-S with UNIFI) resulted in a 10x increase in signal and 7x increase in S:N with the ionKey/MS System. This improvement is illustrated in Figure 7, with a comparison of endogenous levels of bradykinin. Ultimately, the use of the 150 µm iKey Separation Device enabled the development of a low flow quantitative MRM method for bradykinin that achieved a detection limit of 2.5 pg/mL with only 100 μL of plasma.
The development and optimization of the SPE method was described in detail in the previous application note (720004833EN) and was employed for this study. Use of the Oasis WCX SPE, provided both reversed-phase and ion-exchange modes of retention, enabling greater sample cleanup, selectivity, and ultimate sensitivity for this peptide. Additionally, the Oasis WCX 96-well µElution Plate (p/n 186002499) can be processed manually in under 30 minutes and is compatible with most liquid-handling robotic systems for automation to meet sample throughput requirements. This format also provides the ability to elute in very small sample volumes of only 50 μL, minimizing the potential for peptide losses that might occur during evaporation due to adsorption to the walls of collection plates and/or chemical instability.
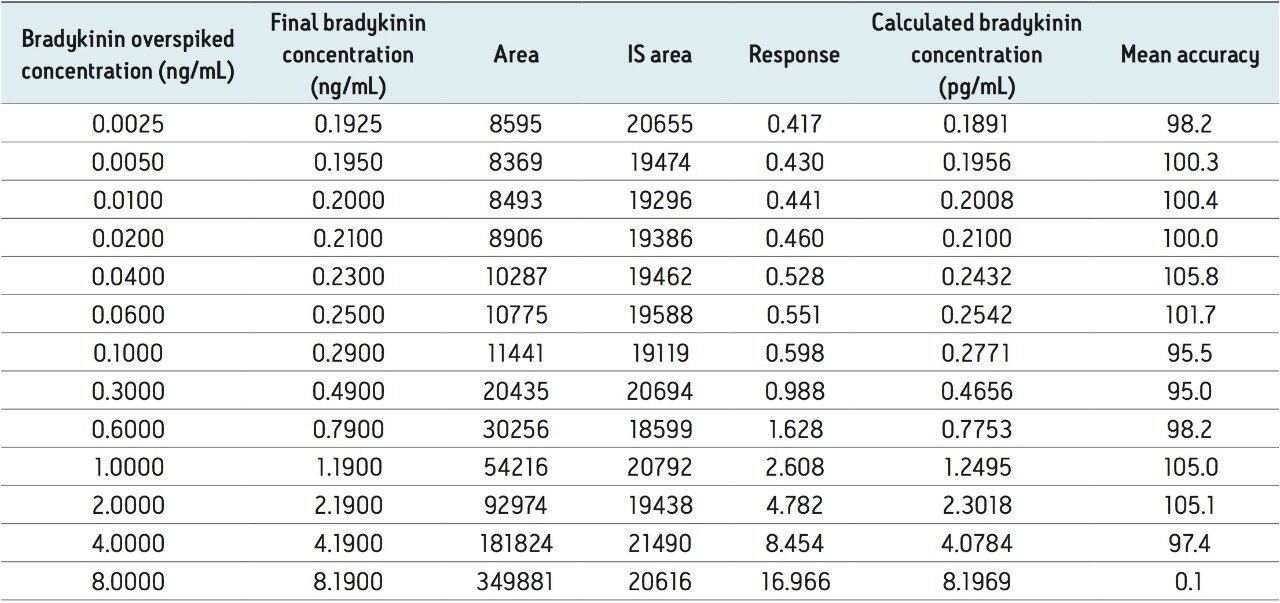
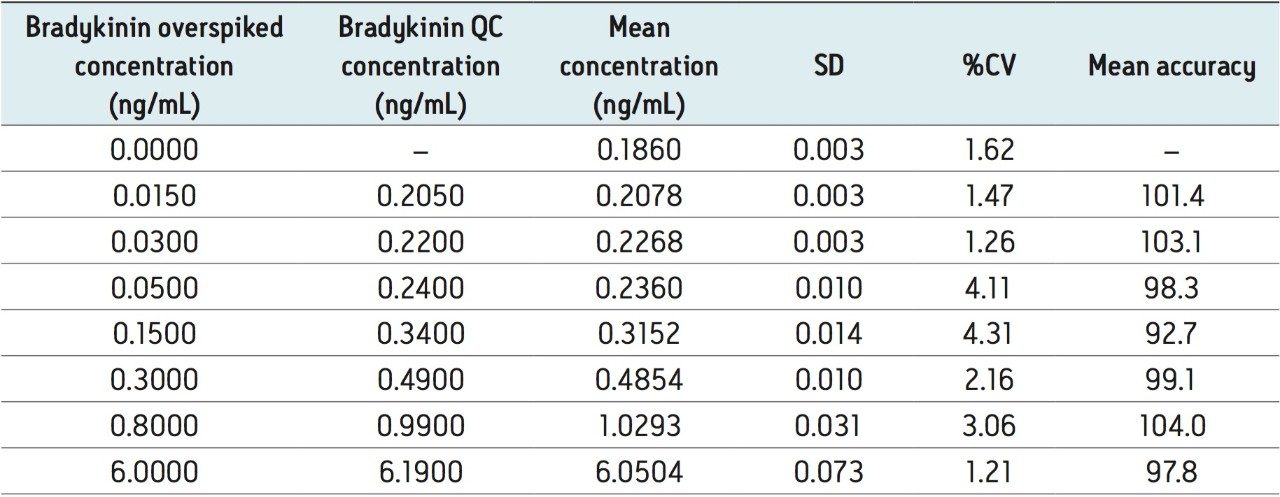
To generate standard curves, human plasma (derived from blood collected in BD P100 tubes) was fortified with bradykinin at the following final concentrations: 2.5, 5, 10, 20, 40, 60, 100, 600, 1,000, 2,000, 4,000, and 8,000 pg/mL. Each standard level was prepared in duplicate. Quality control (QC) samples were prepared from the same plasma at 15, 30, 50, 150, 300, 800, and 6,000 pg/mL. QC samples at each level were prepared in triplicate. [Lys-des-Arg9]-Bradykinin (final concentration of 0.5 ng/mL) was used as the internal standard (IS). Peak area ratios (PARs) of the analyte peak area to the IS peak were calculated. The calibration curve was constructed using PARs of the calibration samples by applying a one/concentration (1/x) weighted linear regression model. All QC sample concentrations were then calculated from their PARS against the calibration curve. Due to the presence of endogenous bradykinin, standard addition was used. The mean basal level of bradykinin (0.19 ng/mL) in control plasma samples was determined by calculating the x-intercept. The calculated basal level was then added to the spiked concentration for all standard curve and QC samples to enable accurate quantification. Using 1/x regression, bradykinin was linear with an R2 value of >0.99. A summary of standard curve performance is shown in is shown in Table 3. Results from QC analysis are shown in Table 4. At all levels, QC samples demonstrated very good accuracy and precision, with mean accuracies ranging from 92.7–104.0 and mean %CV’s of 1.21–4.31. These results easily meet the recommended FDA acceptance criteria outlined in the white papers describing best practices in bioanalytical method validation for LC-MS/MS assays.3,4
Accurate quantification of bradykinin in plasma is particularly challenging because it is metabolized rapidly, with a half life of less than 30 seconds, and can be artificially produced during blood sampling and sample preparation, via proteolytic processes.2,5,6 To assess the best preservation of bradykinin in blood, as well as to prevent the formation of bradykinin ex vivo, particular attention was paid to the protocol for blood collection which employed the use of commercially-available blood collection tubes containing proprietary additives that provide enhanced recovery plasma analytes. More specifically, the BD P100, P700, and P800 collection tubes provide a means of preservation of plasma to be used in peptide and protein analysis.7 The original work presented (720004833EN) only assessed the preservation of bradykinin in P100 blood collection tubes. The BD P100 and P700 blood collection tubes contain proprietary mixtures of additives and inhibitors. The BD P100 collection tubes also contain a mechanical separator that allow for ease of collection and separation of the plasma after blood centrifugation. The P700 tubes contain the same inhibitors as the P100 tubes, with an additional inhibitor for stabilization of Glucagon-Like Peptide I (GLP-1) and contains no mechanical separator. P800 blood collection tubes, like the P100 and P700 blood cllection tubes, contain a proprietray cocktail of inhibitors that provide preservation of bioactive peptide in plasma, and contains no mechanical separator. The P800 blood collection tubes are marketed for assays that require quantitation and measurement of the GLP-1, Glucose-Dependent Insulinotropic Polypeptide (GIP), Glucagon, and Ghrelin.
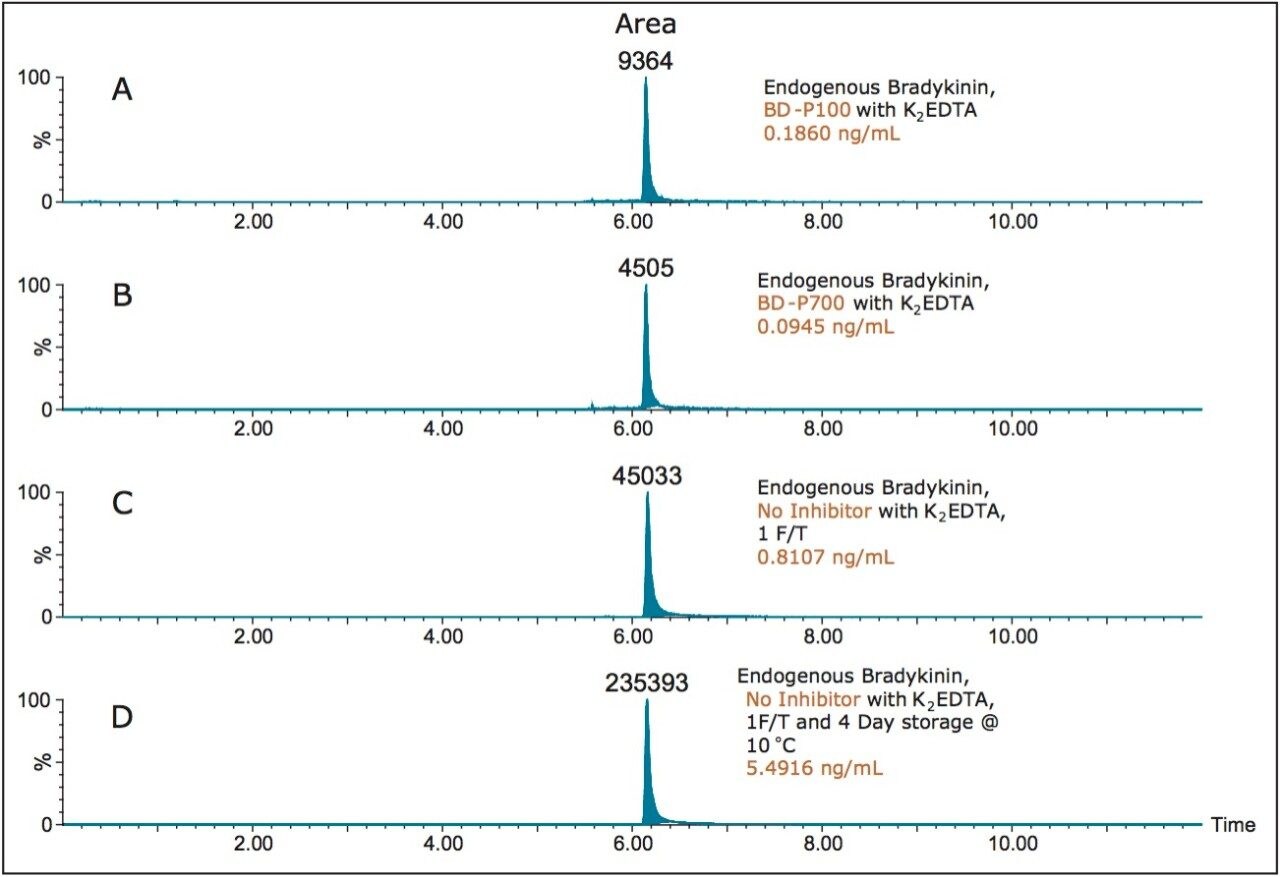
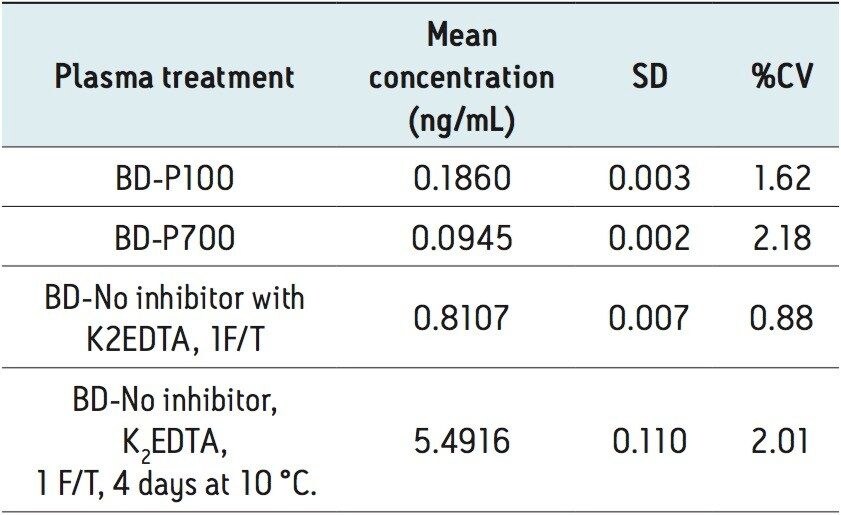
Mean extracted endogenous plasma bradykinin concentrations, in which the blood was collected with (P100, P700), and without protease inhibitors (K2EDTA only, days 1 and 4) are shown in Table 5. Average CV’s of the endogenous bradykinin levels ranged from 0.88–2.18%, indicating a very robust and reproducible method. Representative chromatograms for these results are shown in Figure 8 (panels A-D). Panel A is a representative chromatogram of endogenous plasma bradykinin obtained from blood collected in the P100 tubes, with a mean calculated concentration of 0.1860 ng/mL. P700 blood collection yielded a mean endogenous bradykinin plasma level of 0.0945 ng/mL, and is shown in Panel B. This concentration was approximately half of the concentration determined using the P100 tubes. The artifactual formation of bradykinin in plasma without inhibitor is demonstrated in panels C and D. In these cases, blood was collected in K2EDTA-only blood collection tubes, and the subsequent plasma was brought though 1 freeze/thaw (F/T) cycle. Panel C represents the bradykinin concentration on day 1, where the bradykinin plasma level increased to 0.8107 ng/mL. Panel D represents the bradykinin concentration after 4 days of storage at 10 °C, where bradykinin plasma levels increased to 5.4916 ng/mL. Endogenous levels of bradykinin using the P800 showed relative area counts similar to that of the P700 collection (data not shown), but due to a 10x signal loss of the IS in the P800 tube samples endogenous levels of bradykinin were not calculated for the P800 sample collection. It is assumed that the analogue IS was not protected from metabolism and/or degradation in the P800 tube due to differences in the cocktail of inhibitors. Further, the reduced endogenous bradykinin plasma levels using the P700 collection tube indicated that this cocktail of inhibitors may be more appropriate for stabilization and prevention of ex vivo bradykinin formation. However, another possibility that was not explored was that the presence of the plasma mechanical separator provided a mechanism of bradykinin formation prior or during blood collection and centrifugation. These results further emphasize the need for proper sample collection and storage to accurately quantify endogenous bradykinin plasma levels.
Use of the ionKey/MS System, mixed-mode SPE and higher m/z b or y ion MS fragments provided the level of selectivity and sensitivity necessary to accurately quantify bradykinin and distinguish subtle differences in concentrations. The current analysis uses 100 μL of plasma and provides a significant improvement in sensitivity and S:N over the analytical scale LC-MS analysis which uses twice as much sample. The use of the 150 µm iKey Separation Device enabled the development of a highly sensitive, low flow quantitative MRM method for bradykinin that could distinguish a change of 2.5 pg/mL of bradykinin over the basal level. Standard curves were accurate and precise from 2.5–8,000 pg/mL. QC samples at all levels easily met recommended FDA regulatory criteria4,5 with mean accuracies ranging from 92.7–104.0 and mean %CV’s of 1.20–4.31, indicating an accurate, precise, and reproducible method. Furthermore, an injection of the same volume (10 μL) of sample corresponded to a >10x increase in on-column sensitivity as compared to the traditional analytical flow method for this peptide. In addition to the sensitivity increase the ionKey/MS System provides over the 2.1 mm I.D. scale, it also reduces solvent and sample consumption, thereby reducing cost and allowing for multiple injections of samples for improved accuracy or to meet the guidelines for ISR. This study also demonstrates the importance of proper sample collection with appropriate additives for the stabilization/preservation of bradykinin in plasma to accurately represent endogenous levels. This method shows great promise for high sensitivity quantification of bradykinin in patient samples from PK and clinical studies using the ionKey/MS System if further validation was performed.
We would like to thank:
Jeffrey Widdos and Biological Specialty Corporation for providing excellent service and assistance in the special collection of human plasma used in this work.
David Craft and BD Diagnostics Corporation for providing blood collection tubes for assessment of bradykinin plasma levels, and for the technical discussion regarding the various BD blood collection tubes.
720004945, February 2016