
In this study it was demonstrated that the ProMass software can be adopted into an ACQUITY QDa based MassLynx project for confirmation of analyte mass and yield results consistent with the molecular weight of the target sequence within the default mass tolerance settings.
Research into therapeutic oligonucleotides has received steadily increasing attention from the pharmaceutical industry. This is due to potential applications using deoxyribonucleic acid (DNA) sense/antisense oligonucleotides and interfering ribonucleic acid- (RNAi) based therapies.1,2 The production of oligonucleotides with high yields via automated stepwise synthetic methods is well established. As part of the production process, purification and desalting steps are incorporated to remove byproducts of synthesis, such as failed sequences and production impurities. Characterization of purified synthetic products must be carried out prior to use in therapeutic applications to ensure product identity and purity. Ion Pairing Reversed Phase Liquid Chromatography (IP-RPLC) has become a prevalent technique in the analysis of synthetic oligonucleotides in part due to the selectivity offered by such techniques, as well as its ability to incorporate mass spectrometry-friendly reagents and buffers as first demonstrated by Apffel and colleagues.3,4
Mass information afforded by MS detection offers an efficient means of identifying challenging base modifications for improved productivity in synthetic therapeutic oligonucleotide workflows. Pharmaceutical companies engaged in oligonucleotide research are often investigating numerous potential biotherapeutic candidates, which can negatively impact productivity as the characterization process of synthetic oligonucleotides often requires manual processing of LC-MS data. Previous work demonstrated that incorporation of ProMass (Novatia, LLC) for MassLynx Software enables automated spectrum deconvolution and data analysis for high-throughput screening of UPLC-MS data generated on Waters MS instruments, such as the ACQUITY SQD Detector and SYNAPT.5 As one of the newest mass detectors from Waters, the ACQUITY QDa has been established as an efficient means for obtaining mass information within existing, optically-based LC workflows in the biopharmaceutical manufacturing environment when used as an orthogonal detection technique.6-8
The objective of this application note is to demonstrate that the ACQUITY QDa Detector provides a simple and cost-effective solution in the assessment of identity and purity of synthetic oligonucleotides and that mass spectral data acquired can be readily processed with ProMass for MassLynx Software in an automated fashion.
Triethylamine (99.5% purity) and 1,1,1,3,3,3-hexafluoro-2-propanol (99.8% purity, LC-MS grade) were purchased from Sigma Aldrich. Mass spectrometry grade solvents (Optima series) were purchased from Fisher Scientific. Mobile phase buffers were newly prepared prior to experiments. PolyT oligonucleotide standards were from Waters (P/N 186004135). siRNA upper strand 5'-UCGUCAAGCGAUUACAAGGTT-3' and its complementary lower strand 5' TTCCUUGUAAUCGCUUGACGA-3' were ordered from Integrated DNA Technologies. All samples were prepared at a concentration of 10 pmol/μL. Mass loads on column were kept constant at 50 pmol or 5 μL injections.
|
LC system: |
ACQUITY UPLC H-Class |
|
Detectors: |
ACQUITY UPLC TUV w/Ti flow cell, ACQUITY QDa Detector |
|
Absorption wavelength: |
260 nm |
|
Column: |
ACQUITY UPLC OST BEH C18, 1.7 μm, 2.1 mm x 50 mm (P/N 186003949) |
|
Column temp.: |
60 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
5 μL |
|
Mobile phase A: |
15 mM TEA, 400 mM HFIP prepared in H2O, pH 8.0 |
|
Mobile phase B: |
15 mM TEA, 400 mM HFIP prepared in MeOH |
|
*mobile phases prepared gravimetrically |
|
Time |
Flow (mL/min) |
%A |
%B |
%C |
%D |
|---|---|---|---|---|---|
|
Initial |
0.200 |
81.0 |
19.0 |
0 |
0 |
|
15.00 |
0.200 |
73.5 |
26.5 |
0 |
0 |
|
16.00 |
0.200 |
50.0 |
50.0 |
0 |
0 |
|
17.00 |
0.200 |
81.0 |
19.0 |
0 |
0 |
|
21.00 |
0.200 |
81.0 |
19.0 |
0 |
0 |
|
Time |
Flow (mL/min) |
%A |
%B |
%C |
%D |
|---|---|---|---|---|---|
|
Initial |
0.200 |
82.0 |
18.0 |
0 |
0 |
|
4.00 |
0.200 |
80.0 |
20.0 |
0 |
0 |
|
4.01 |
0.200 |
50.0 |
50.0 |
0 |
0 |
|
6.00 |
0.200 |
50.0 |
50.0 |
0 |
0 |
|
6.01 |
0.200 |
82.0 |
18.0 |
0 |
0 |
|
10.00 |
0.200 |
82.0 |
18.0 |
0 |
0 |
|
Sample rate: |
2 points/sec |
|
Mass range: |
410–1250 Da |
|
Mode: |
ESI negative |
|
Collection mode: |
continuum |
|
Cone voltage: |
20 V |
|
Capillary voltage: |
0.8 kV |
|
Probe temp.: |
600 °C |
MassLynx SCN 9.25 with MaxEnt1
Recently, it was demonstrated that the ACQUITY QDa Detector is capable of providing mass spectral data using traditional IP-RPLC methods, which incorporate ion pairing agents such as TEA buffered in HFIP, as shown in Figure 1A.9 In the previous study, it was also shown that the ACQUITY QDa is capable of providing accurate mass information within its operating specifications. In addition, it was shown that MS spectra processed within MassLynx using the MaxEnt1 deconvolution algorithm provided analysts with straightforward data interpretation of complex spectra (Figure 1B).
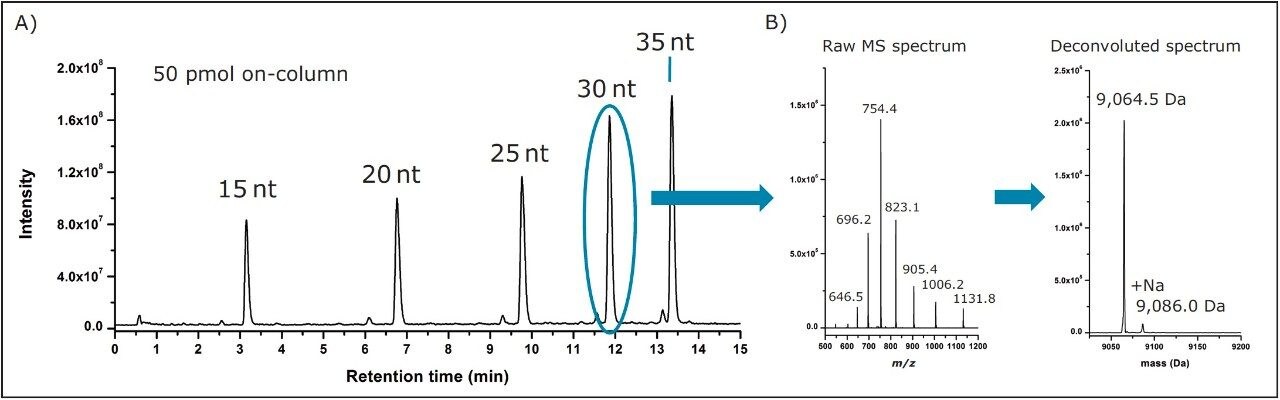
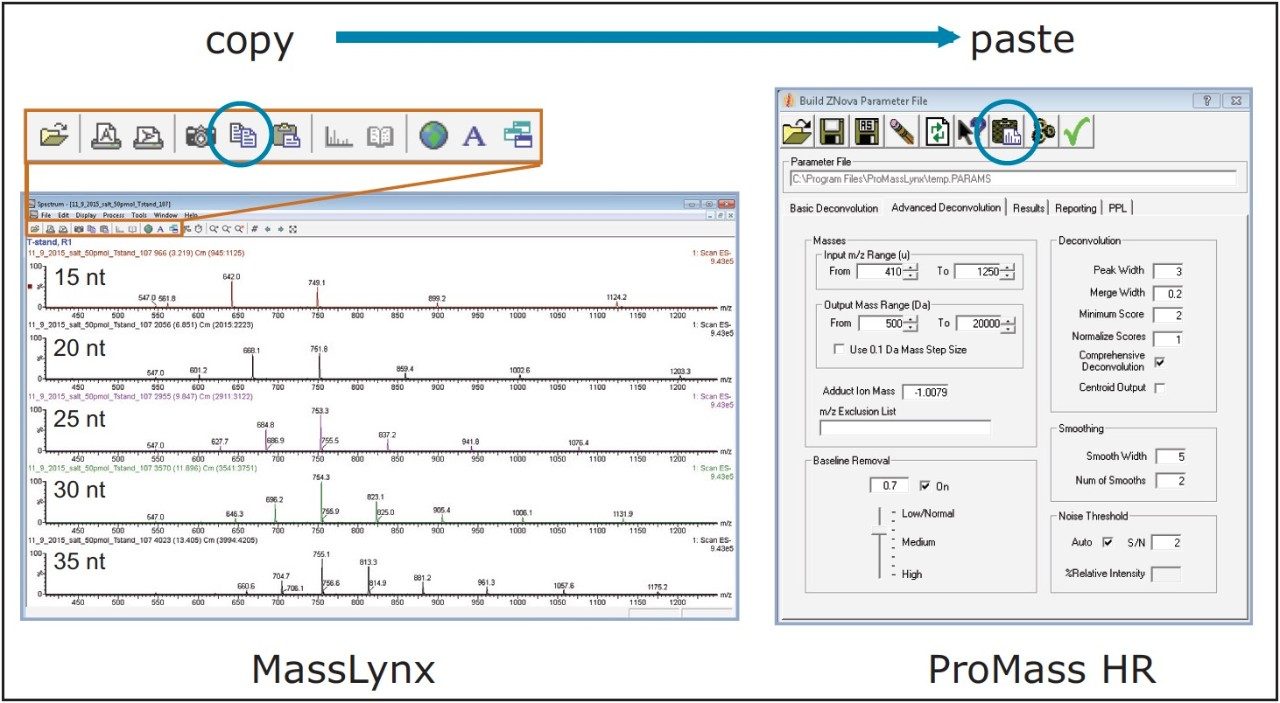
As aforementioned, pharmaceutical companies engaged in oligonucleotide research often incorporate software such as ProMass that enable automated data analysis in a high-throughput manner for improved productivity. A natural extension of the previous study is to evaluate the compatibility of ProMass with average mass data acquired using the ACQUITY QDa Detector. As with the previous work, a one minute window centered across the peak apex of the polyT standards was used to combine the MS spectrum data acquired by the ACQUITY QDa, as shown in the left panel of Figure 2. ProMass, which uses the ZNova deconvolution algorithm, offers a panel of deconvolution parameters for method flexibility in the deconvolution of components/peaks in the mass chromatograms, as shown in the right panel of Figure 2. For this experiment, the default settings used were: Peak Width=3, Merge Width=0.2, Minimum Score=2, and Normalize Scores=1. Spectrum for each polyT standard was copied to the operating system clipboard and processed using the manual processing feature of ProMass, as illustrated in Figure 2.
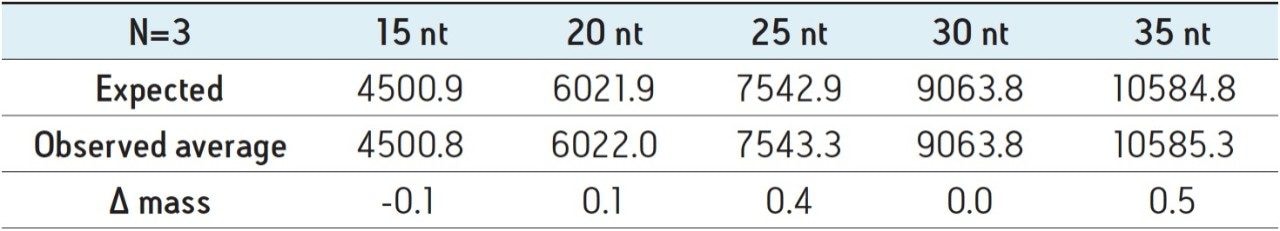
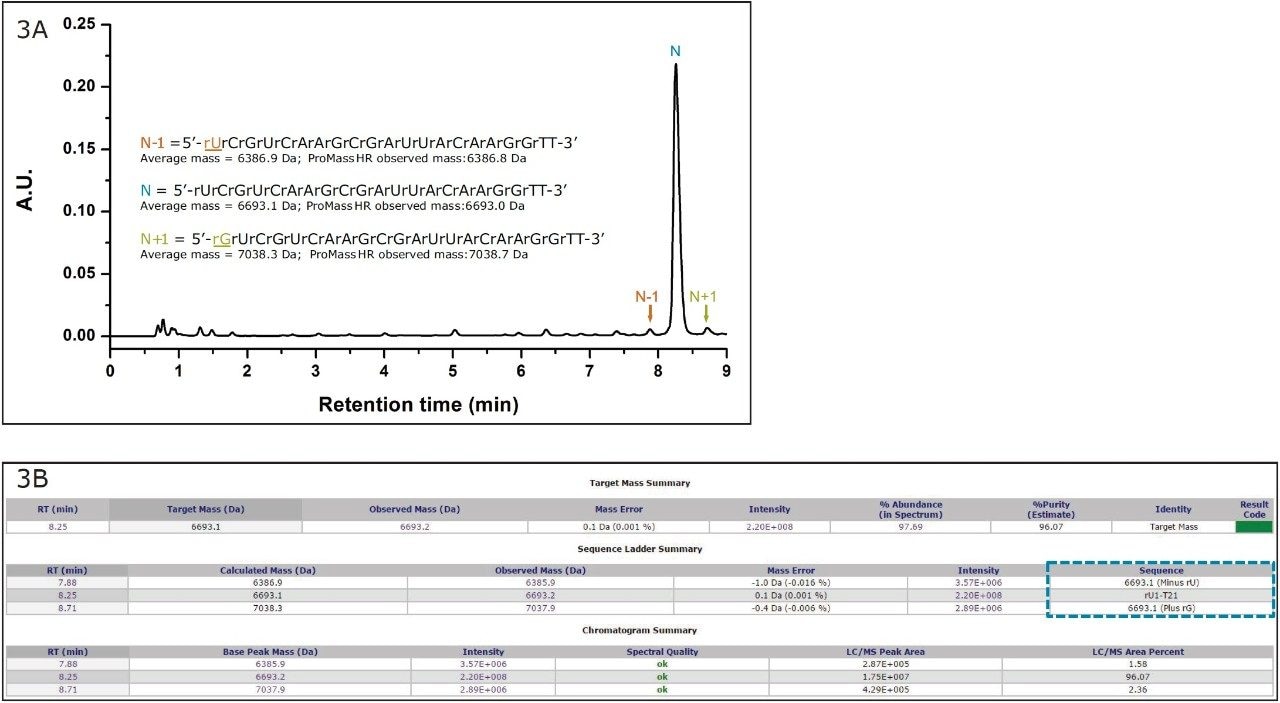
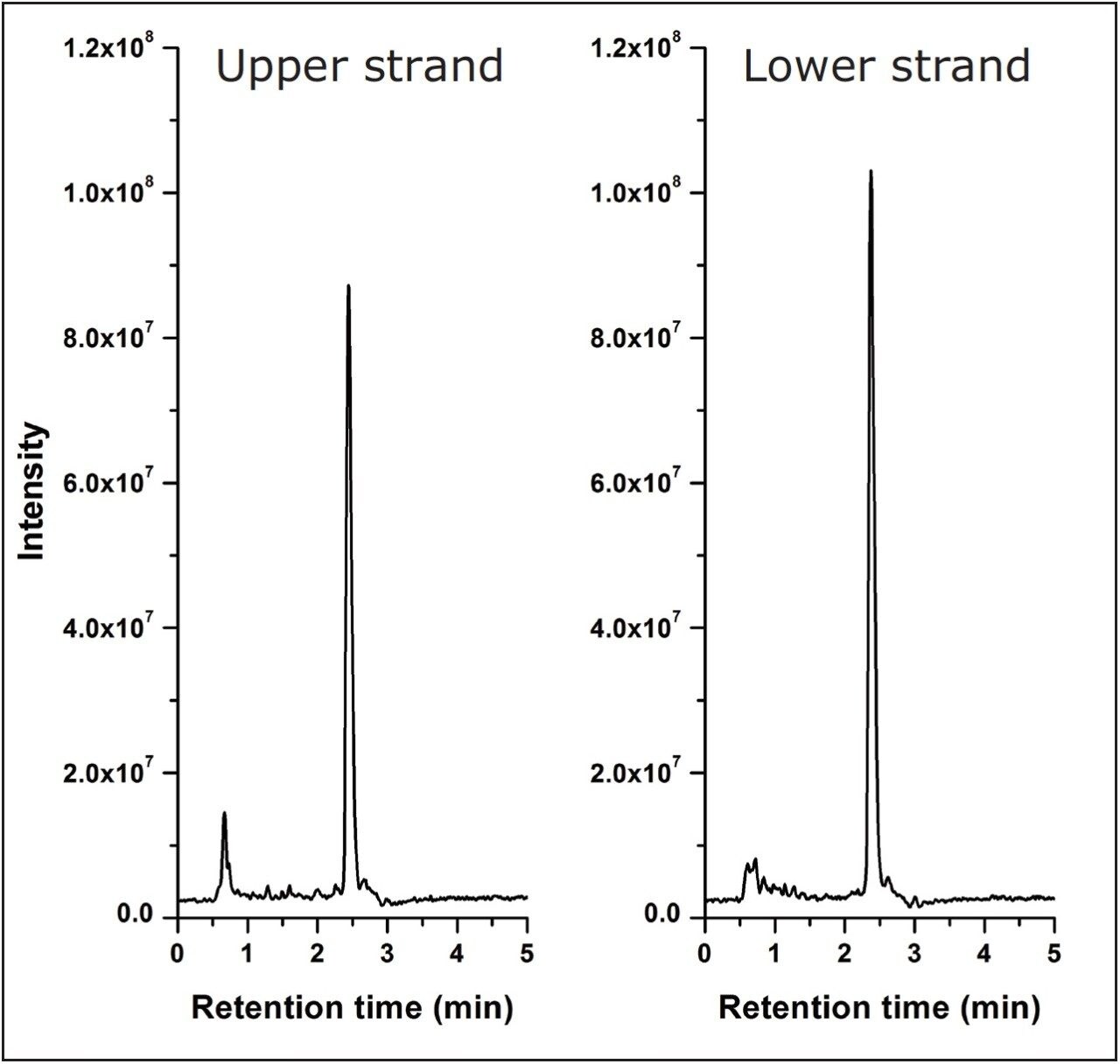
The deconvolution results were observed to be highly reproducible across a technical triplicate with mass accuracy observed between -0.1 Da to +0.5 Da across the polyT standards (Table 1). To further evaluate the mass accuracy of ProMass with a sample more representative of one found in a therapeutic setting, a siRNA sample with a sequence length of 21 nucleotides – exhibiting a double thymine overhang – was separated using a high resolution separation gradient (Figure 3A). The pure oligonucleotide (N) was resolved from the base deletion (N-1) and base insertion (N+1) forms in under ten minutes. Using a one minute integration window from 7.7 minutes to 8.8 minutes, ProMass successfully identified the deletion and insertion as shown in the sequence ladder summary report (Figure 3B). Similar to the polyT analysis, spectrum acquired with the ACQUITY QDa was combined from 7.5 to 9.5 minutes for deconvolution. Mass accuracy of the associated spectrum was observed to range between +0.1 Da to +0.4 Da. This demonstrates that ProMass is compatible with mass spectral data acquired with the ACQUITY QDa and is capable of providing adequate mass information for routine identification and purity assessments in the manufacturing process of synthetic oligonucleotides.
With the performance confirmed of ProMass utilizing mass spectral data acquired with the ACQUITY QDa, the ability to process data in a high-throughput manner was then evaluated. The software package provides a formatted MassLynx sample list, which is designed to allow UPLC-MS data to be processed immediately after data acquisition while the next sample in the queue is analyzed or batch processed offline post-acquisition in an automated fashion (Figure 4). The user provides the raw data, defines the target sequence, and provides details about the target product search (Target Info) – correct configuration of which is essential for successful data processing. Two columns for defining parameter files required by ProMass for processing the LC-MS chromatograms are also provided in the sample list format and are defined by the analyst as shown in Figure 4. The ProMassBridge Parameter File, which is part of the ProMassBridge application, facilitates a connection between MassLynx raw files and ProMass, and contains the user defined parameters (e.g. retention time range, smoothing and subtracting settings) for integration of the MS chromatograms. The ProMass ZNova Parameter File contains user-definable settings for MS data analysis such as the input and output masses, ion polarity, deconvolution settings, target mass, impurities tolerance thresholds, and options for the data report.
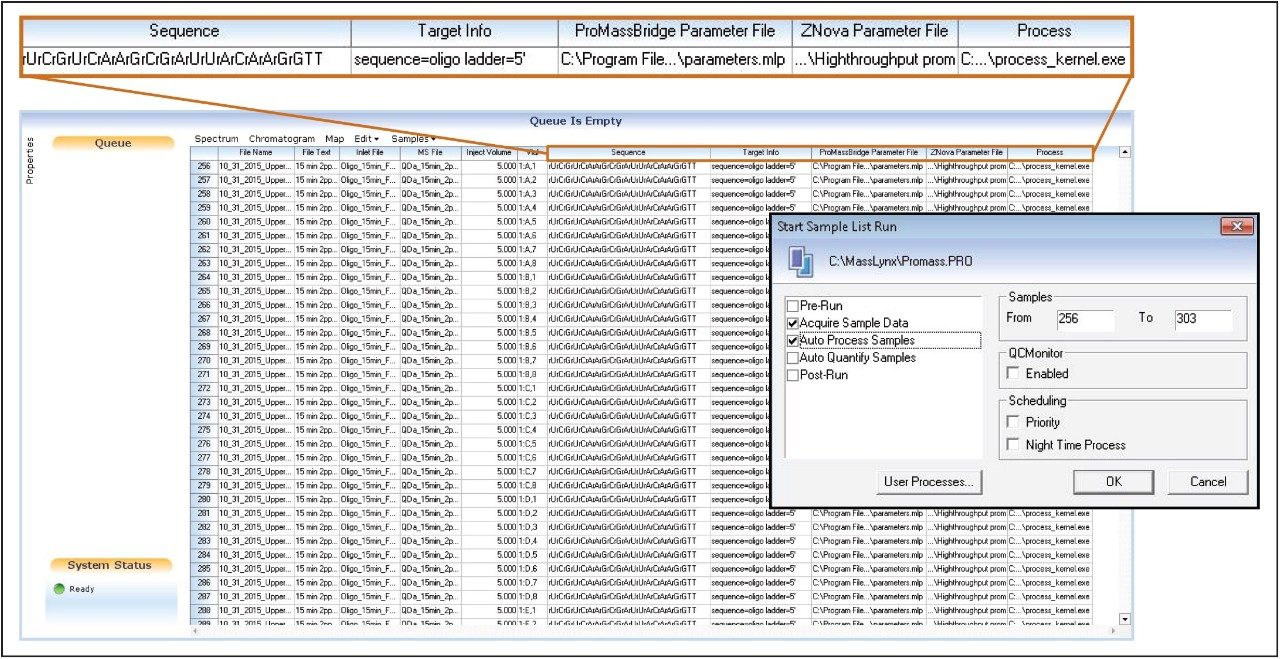
The siRNA sample previously used (upper strand) and its complementary sequence (lower strand) were used to evaluate the batch processing capability of ProMass. A ten minute high-throughput separation method was used for the analysis (Figure 5). The ProMassBridge Parameter File was configured to integrate MS chromatograms between 2.0 and 3.5 minutes with lockspray functionality disabled (this function is used for HRMS data; e.g., QToF MS). All other parameters were left at their default settings. For high-throughput analysis mode, ProMass software has the ability to display the data summary in an interactive sample plate format or Excel sheet.
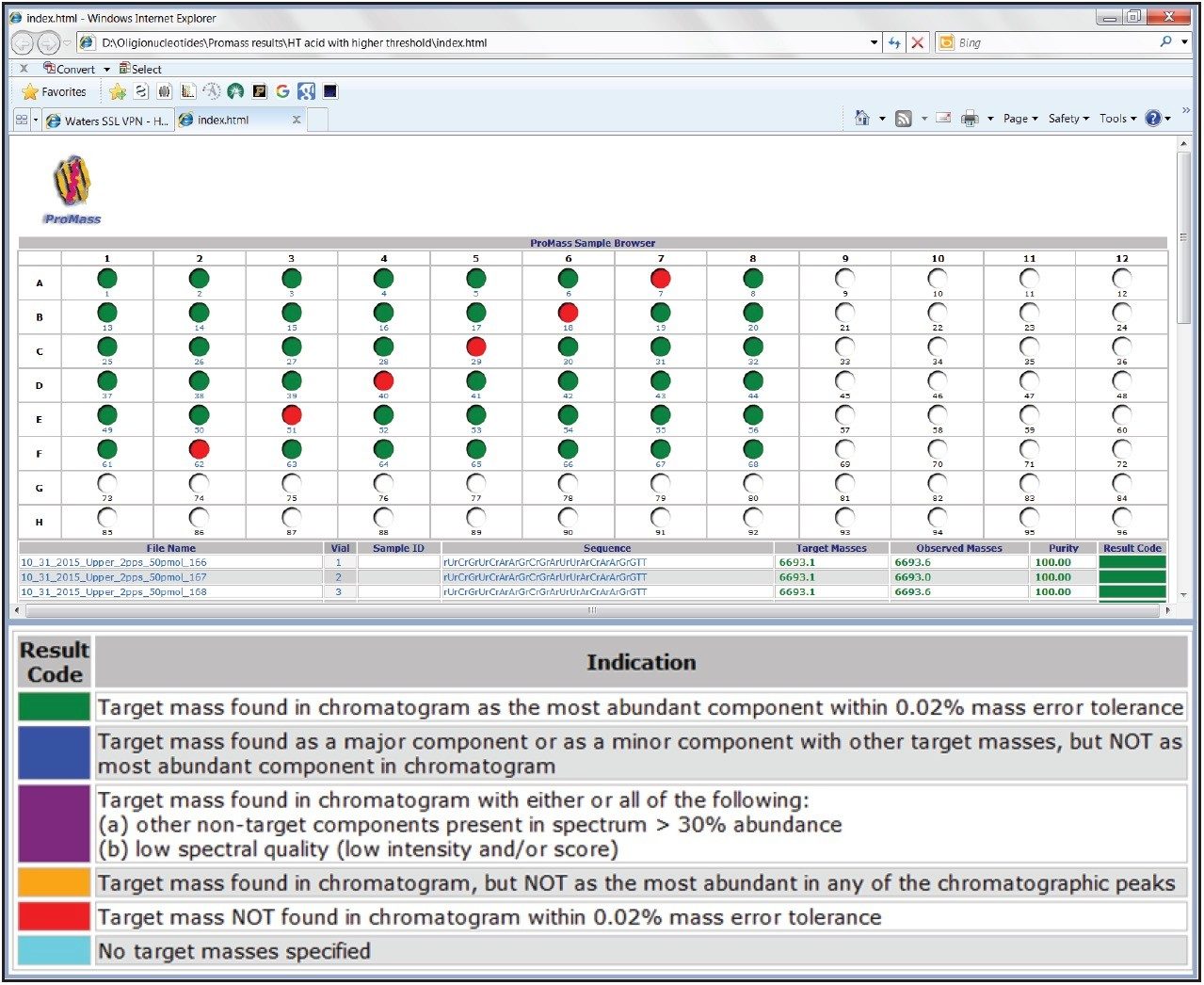
To test the interactive sample plate format display option, upper siRNA samples were loaded in the standard 48-well plate format with every 7th vial position loaded with the lower siRNA complementary sequence as a negative control. As shown in Figure 4, samples from line 256 to line 303 were selected for batch processing with the upper siRNA sequence targeted for identification. After batch processing, the data is displayed in a color-coded, internet browser-based format (Figure 6). The colors, which represent the result of the targeted mass search, are defined in the legend of Figure 6. The plate format display is interactive; the analysis results can be displayed by selecting the actual vial position. Information such as a summary report of the analysis with target peak identification, spectral abundance, and peak purity can be accessed in this manner along with the corresponding spectrum and chromatogram. Additional information includes deconvolution results, identified impurities, and details of associated spectra.
Mass information afforded by MS detection offers an efficient means of identifying challenging base modifications for improved productivity in synthetic therapeutic oligonucleotide workflows. Methods that can be rapidly deployed and automated in the assessment of product identity and purity in the production of synthetic oligonucleotides are highly desirable. In this study it was demonstrated the ProMass software can be adopted into an ACQUITY QDa based MassLynx project for confirmation of analyte mass and yield results consistent with the molecular weight of the target sequence within the default mass tolerance settings. Through the use of ProMass, assignment of target peaks, base modifications, and process impurities can be performed in a high-throughput automated fashion with significantly reduced analysis time for improved productivity. The interactive color-coded results in web browser-based format are user-friendly and enable straightforward data interpretation. This work demonstrates the ACQUITY QDa is an ideal addition to an analyst’s lab for increased productivity and confidence of data analysis for routine identification and purity assessments in the manufacturing process of synthetic oligonucleotides.
720005681, April 2016