
In this work, sample extraction, clean-up, and analysis methods were developed for UPLC-MS/MS determination of a wide variety of veterinary drugs in eggs. The ACQUITY UPLC I-Class System coupled with Xevo TQ-S MS offered good sensitivity for the veterinary drug residues in this study.
In order to insure public health and safety, reliable analytical methods are necessary to determine veterinary drug residue levels in foods. The compounds of interest range from highly polar, water-soluble compounds to very non-polar, fat-soluble compounds. In order to maximize throughput and minimize costs it is desirable to determine the widest possible range of veterinary drug residues in food samples with a single analytical method. Eggs contains significant amounts of proteins, fats, and, lecithin (phospholipids). These components can be detrimental to good instrumental performance and should be reduced or eliminated prior to LC-MS analysis.
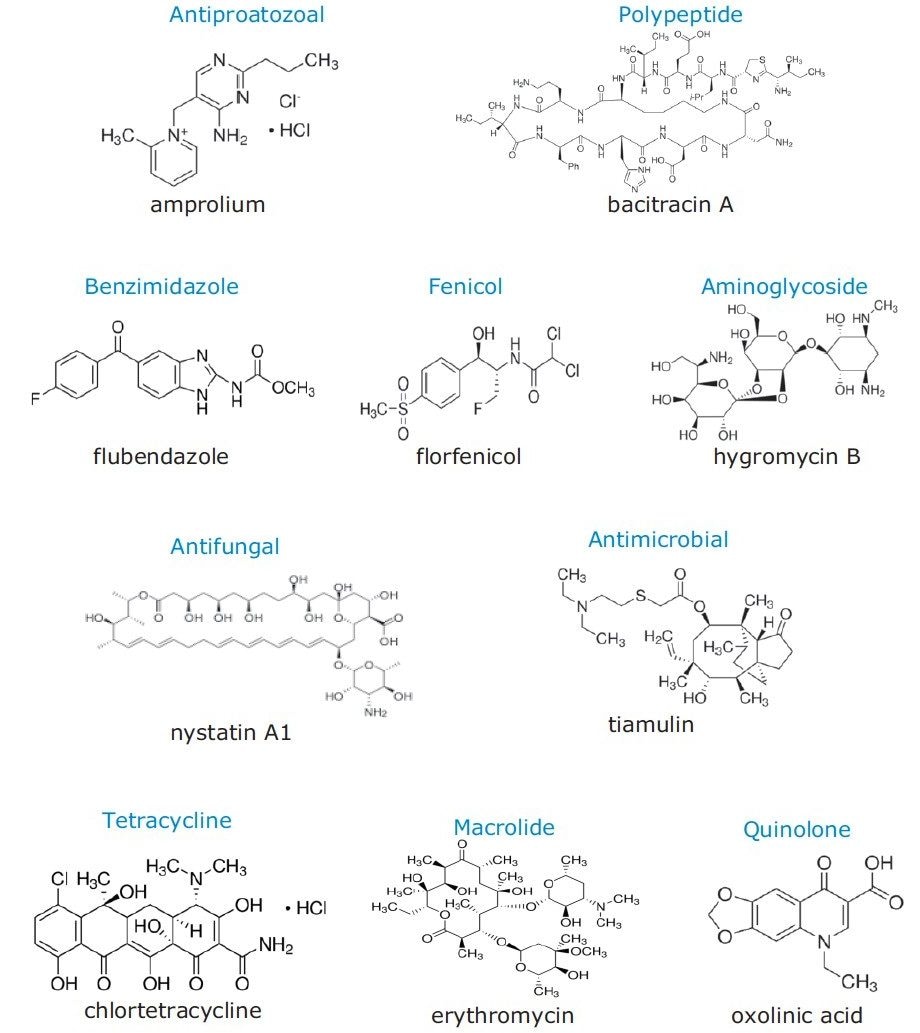
Veterinary drugs are used in chicken farms to control diseases of laying hens. However, these compounds can be transferred to and accumulate in the eggs. The presence of veterinary drug residues in eggs is a potential health risk for the consumer because the residual drugs can provoke allergic reactions or induce pathogen resistance to antibiotics used in human medicine1. Sixteen representative veterinary drugs from twelve classes, most of which have MRLs established in US or China, were chosen for this study.1,2 Figure 1 presents the structures of a subset of these veterinary drugs.
Sample preparation is a challenging task for the multi-residue determination of veterinary drugs in eggs. The analyst must recover a wide variety of drug classes with different physico-chemical properties. Some of the target compounds may bind to proteins or other matrix components. Eggs are among the highest food sources of lecithin (phospholipids) and also have significant amounts of fats; these co-extracted substances can lead to interference and ion suppression in the LC-MS analysis, contamination of the analytical column, and other components of the UPLC system, and contamination of the mass spectrometer itself.
In this work, sample extraction, clean-up, and analysis methods were developed for UPLC-MS/MS determination of a wide variety of veterinary drugs in eggs. Samples were treated with an acidified acetonitrile/water solvent to precipitate proteins, release bound residues, and to extract the veterinary drugs of interest. Then, to remove fats and phospholipids, a simple pass-through clean-up was performed using a novel SPE device, the Oasis PRiME HLB Cartridge.
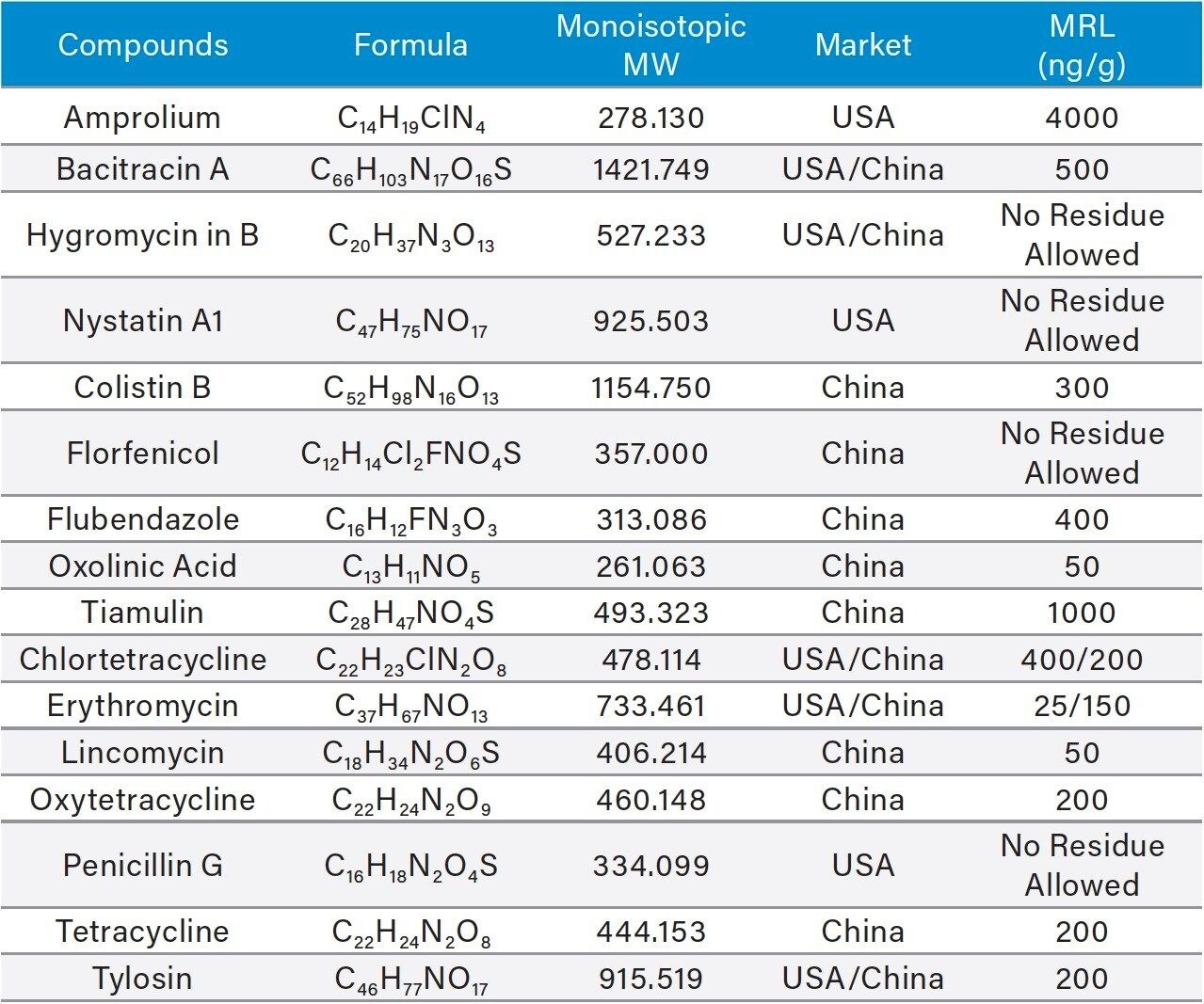
Sixteen veterinary drugs from different classes were chosen for this study. Table 1 lists their formulas, MWs, and MRLs established in USA or China.
|
System: |
ACQUITY UPLC I-Class |
|
Column: |
ACQUITY UPLC BEH C18, 1.7 μm, 2.1 x 100 mm |
|
Column temp.: |
30 °C |
|
Injection volume: |
10 μL |
|
Flow rate: |
0.4 mL/min |
|
Mobile phase A: |
0.1% Formic acid in water |
|
Mobile phase B: |
0.1% Formic acid in acetonitrile |
The initial composition was 85% A and 15% B. Phase B was increased linearly to 40% in the first 2.5 min, and then linear ramp to 95% B in 1.4 min, maintained for 2.3 min, then returned to the initial composition and equilibrated for 2 min.
|
System: |
Xevo TQ-S |
|
Ionization mode: |
ES+ (ES-for Florfenicol) |
|
Capillary voltage: |
3.00 kV (2.50 kV for negative mode) |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
600 °C |
|
Cone gas flow: |
150 L/hr |
|
Desolvation gas flow: |
1000 L/hr |
|
Collision gas flow: |
0.15 mL/min |
|
Nebulizer gas flow: |
7.00 Bar |
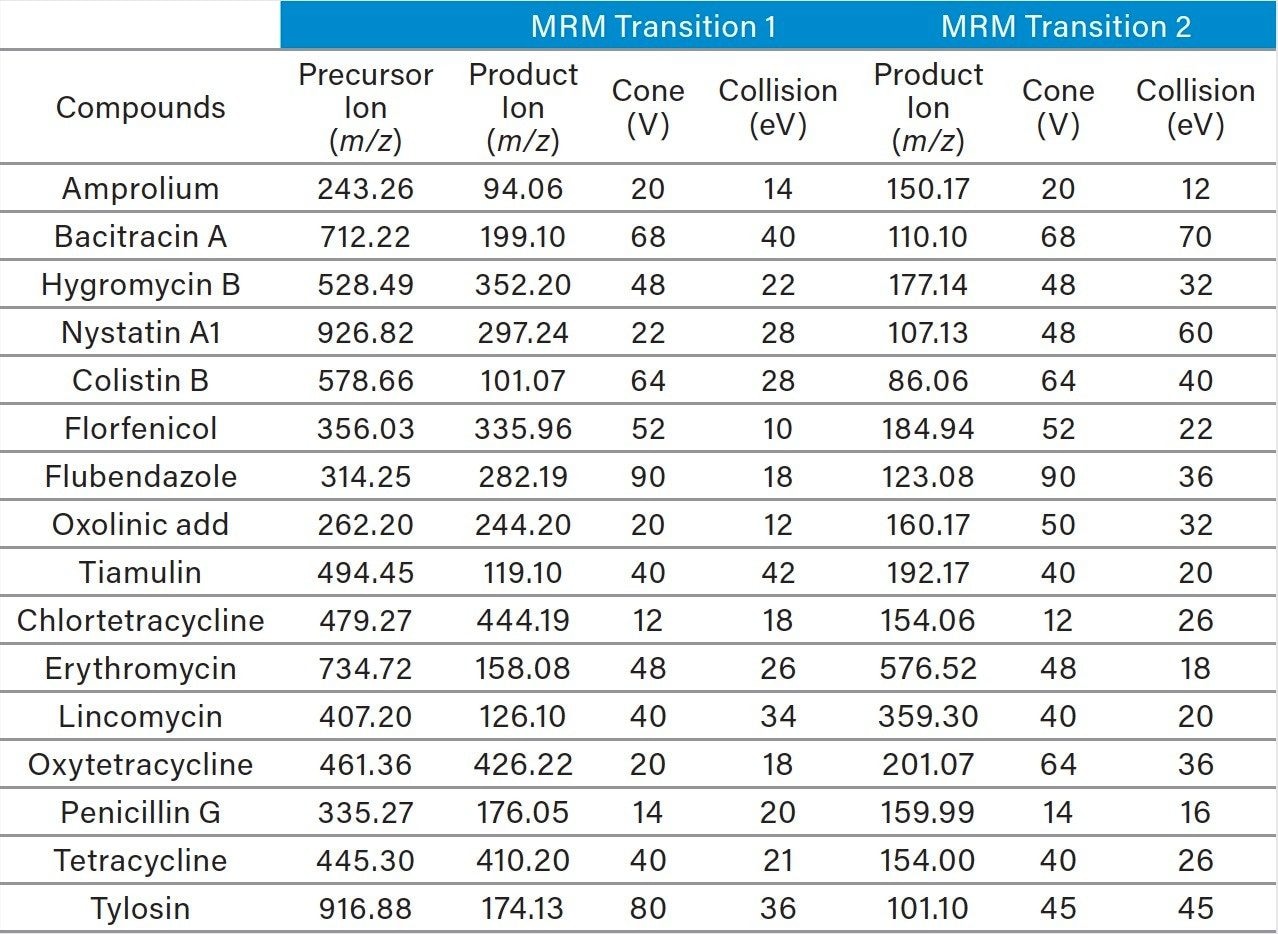
Extraction: Two grams of homogenized whole chicken eggs were weighed into a 50 mL polypropylene centrifuge tube. Recovery samples were fortified with the appropriate amount of standards before 8 mL of 0.2% formic acid in 80:20 acetonitrile/water were added. The samples were vortexed for 30 s, placed on a mechanical shaker for 30 mins, and then centrifuged at 4500 rpm for 10 min. An aliquot of the supernatant was taken for the SPE clean-up.
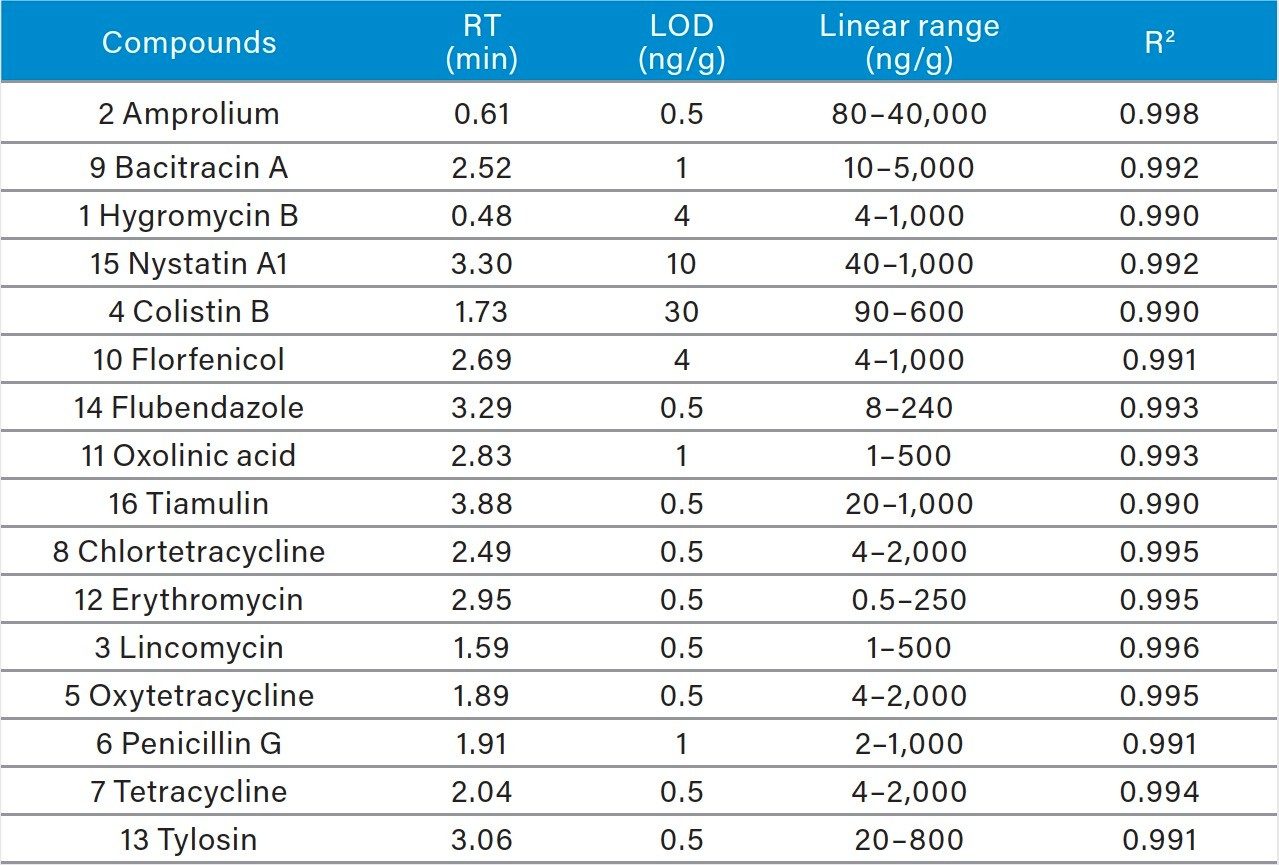
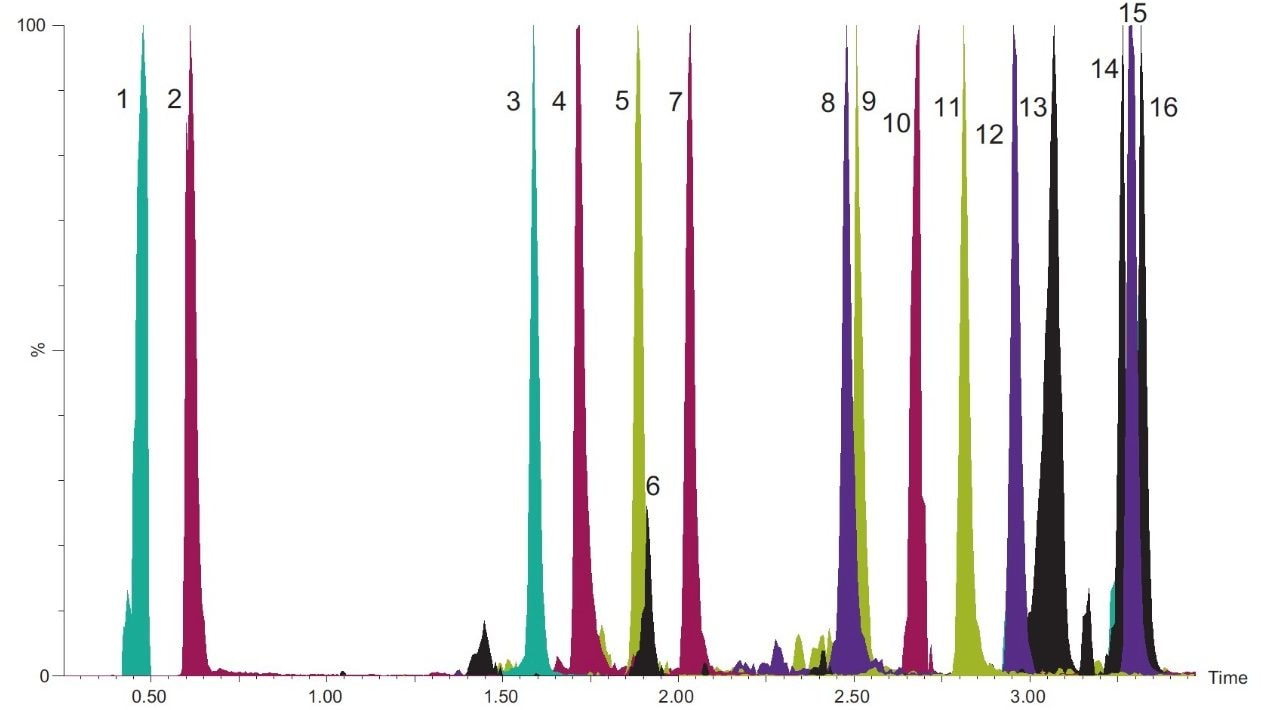
Pass-through SPE clean-up: An Oasis PRiME HLB 3 cc Vac Cartridge, 60 mg, (p/n: 186008056), was mounted on a precleaned vacuum manifold. Cartridge conditioning is not required and was not performed. A 0.5 mL aliquot of the supernatant was passed through the cartridge and collected using 1~2 psi vacuum. 0.2 mL of the collected extract was taken and diluted to 0.6 mL with aqueous 10 mM ammonium formate buffer (pH 4.5) prior to UPLC-MS/MS analysis. Figure 2 shows a typical chromatographic separation obtained for a matrix-matched standard.
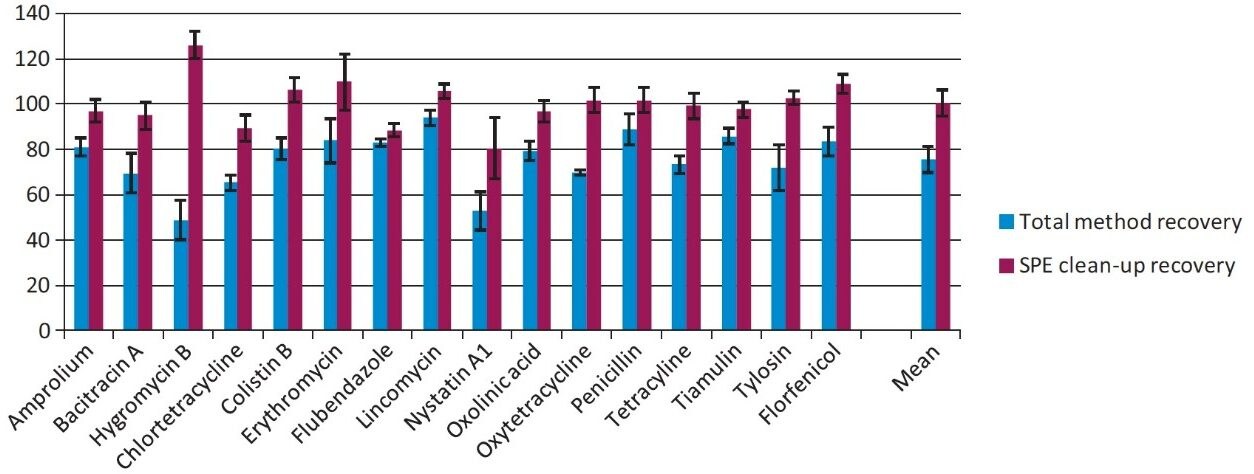
The Oasis PRiME HLB Cartridge was evaluated with respect to analyte recovery and phospholipids removal from egg matrix. The total method recoveries ranged from 50–97%. However, the Oasis PRiME HLB Cartridge clean-up contributes little to any method recovery losses. As shown in Figure 3, the measured recovery for the SPE clean-up step is better than 80% for all compounds, with recovery for most compounds greater than 90%.
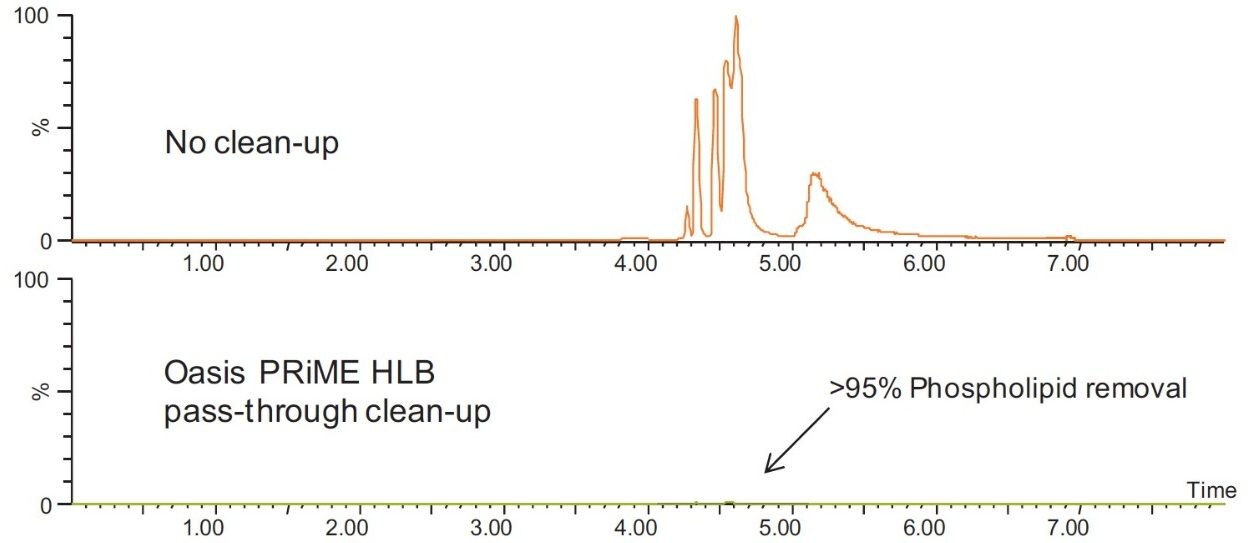
Whole eggs contain significant amounts of fat and are among the highest sources of dietary lecithin (phospholipids). The total lipid content of chicken egg is about 11% by weight (excluding the shell) and the phospholipids content is about 0.35%.4 Significant amounts of these potential interfering substances are extracted along with the target drugs in the initial sample preparation extraction step. Greater than 84% of total lipids were removed from the egg extract after pass-through clean-up with the Oasis PRiME HLB Cartridge. The clean-up step was even more effective for removal of phospholipids. Figure 4 shows that the Oasis PRiME HLB Cartridge clean-up removed greater than 95% of phospholipids from the egg extract.
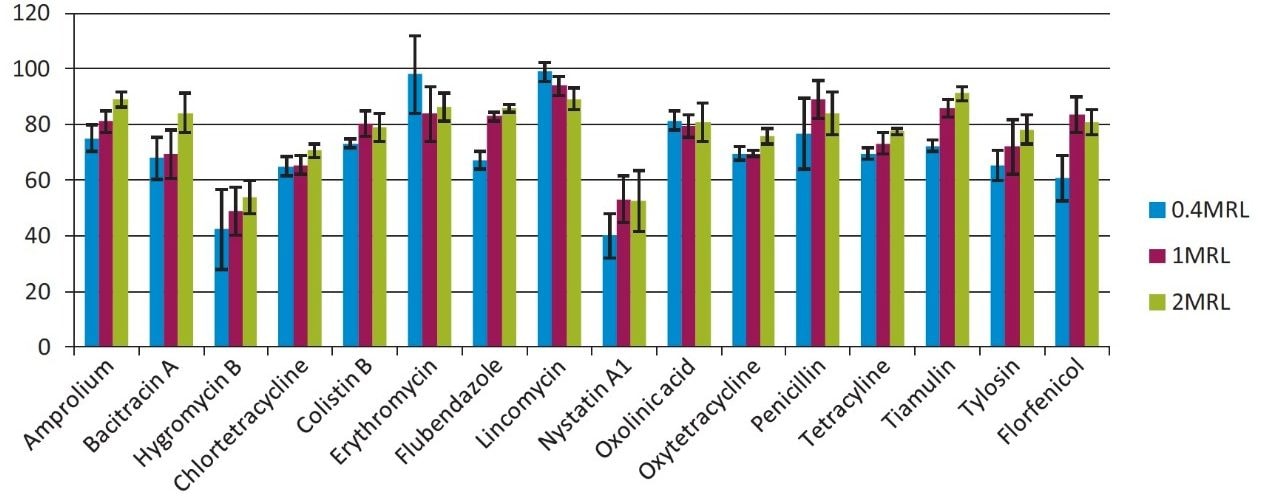
Recovery studies were carried out at three concentration levels (0.4MRL, 1MRL, 2MRL), six replicates per level. Matrix-matched standard calibration curves were used. Figure 5 shows the results. Recovery was greater than 70% for most target compounds (>70%) except for Nystatin and Hygromycin. Reproducibility was acceptable (RSD<20%) for all compounds except for Hygromycin at 0.4 MRL (RSD=34%).
720005794, February 2016