For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
This Application brief accurately quantify vitamin D metabolites in low volume serum samples to facilitate human, rodent, and in vitro research studies.
Measure multiple vitamin D metabolites in a single analysis using only 100 μL of sample.
Total serum 25-hydroxyvitamin D (25(OH)D, the sum of the concentrations of 25(OH)D2 and 25(OH)D3), is the biomarker of vitamin D nutritional status.1 The vitamin D catabolite, 24,25-dihydroxyvitamin D3 (24,25(OH)2D3) is formed from 25(OH)D3 by the action of cytochrome P450 CYP24A1, which is now accepted as a determinant of circulating 25(OH)D levels.2 In current vitamin D research studies, there is renewed interest in quantifying 24,25(OH)2D3, in addition to 25(OH)D as a measure of the rate of clearance of 25(OH)D3. However, the analytical tools to quantify multiple vitamin D metabolites in a single method are not widely available.
To facilitate clinical research studies of vitamin D metabolism, we have developed a new method based on liquid-liquid extraction, chemical derivatization, and UPLC-MS/MS, using an ACQUITY UPLC System with a Xevo TQ-S (Figure 1). The method provides analytically sensitive and selective measurement of 24,25(OH)2D3 and 25(OH)D in the same run, requiring only 100 μL of serum. The recent availability of deuterated internal standard for 24,25(OH)2D3 from various sources enables a truly quantitative approach. The chromatographic separation of 3-epi-25(OH)D3 from 25(OH)D3 enabled by this method ensures that 25(OH)D3 measurements are not confounded by the presence of this naturally-occurring stereoisomer. The method described here can be easily adapted for the analysis of rodent serum,3 and for the study of vitamin D metabolism in in vitro cultured-cell models.

25(OH)D3 and 25(OH)D2 calibrators (6PLUS1) were purchased from Chromsystems. A 6-level calibrator set for 24,25(OH)2D3 was generated in-house using a matrix of 20% human serum in 0.1% bovine serum albumin dissolved in phosphate-buffered saline; supplemented with synthetic 24,25(OH)2D3. Internal standards d3-25(OH)D3 and d3-25(OH)D2 were purchased from Isosciences. d6-24,25(OH)2D3 was supplied by Drs. Antonio Mourino and Miguel Maestro, University of Santiago de Compostela. All LC-MS/MS solvents, additives, and extraction solvents were Optima LC-MS grade and purchased from Fisher Scientific, with the exception of MTBE, which was purchased from Sigma Aldrich. DMEQ-TAD was purchased from Key Synthesis.
In microcentrifuge tubes, 100-μL aliquots of test serum or calibrator were diluted with 200 μL of water and supplemented with the following internal standards: 80 ng/mL d3-25(OH)D3, 65 ng/mL d3-25(OH)D2, and 6 ng/mL d6-24,25(OH)2D3. A 100 μL volume of 0.1M HCl was added, and protein precipitation was carried out by adding 150 μL 0.2 M zinc sulfate and 450 μL of methanol with vortex mixing after addition of each component. The mixture was centrifuged at 12,000 x g for 10 minutes and the supernatant was transferred to borosilicate glass tubes. Organic extraction was carried out by adding 700 μL of hexane and 700 μL of MTBE, with vortex mixing after addition of each component. The upper organic phase was transferred into maximum recovery vials (p/n 186000327c) and dried under a stream of prepurified N2 at 37 °C. Samples were derivatized by redissolving the dry residue in 25 μL of 0.1 mg/mL DMEQ-TAD in ethyl acetate and incubating for 30 minutes at room temperature in the dark. A second aliquot of DMEQ-TAD was added and allowed to incubate for an additional 60 minutes.4 A 40 μL volume of ethanol was added and the derivatized extract was dried, and redissolved in 60 μL of 60:40 (v/v) methanol/water UPLC mobile phase.
|
LC system: |
ACQUITY UPLC |
|
Column: |
ACQUITY UPLC BEH-Phenyl, Å300, 1.7 μm, 2.1 x 50mm (p/n 186002884) |
|
Column temp.: |
40 °C |
|
Sample temp.: |
4 °C |
|
Injection vol.: |
10 μL, full loop mode |
|
Strong wash: |
Methanol |
|
Weak wash: |
60:40 (v/v) methanol/water |
|
Flow rate: |
400 μL/min |
|
Mobile phase A: |
2 mM ammonium acetate plus 0.1% formic acid in water |
|
Mobile phase B: |
2 mM ammonium acetate plus 0.1% formic acid in methanol |
|
Gradient: |
Initial conditions were 35:65 (v/v) mobile phase A:mobile phase B at a flow rate of 400 μL/min. Mobile phase B was increased to 90% over 5 minutes using an exponential gradient (curve 8), before returning to starting conditions for 1 minute for a total run time of 6 minutes. |
|
Mass spectrometer: |
Xevo TQ-S |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
1.0 kV |
|
Desolvation temp.: |
650 °C |
|
Desolvation gas: |
1000 L/h |
|
Cone gas: |
150 L/h |
|
Acquisition mode: |
multiple reaction monitoring (MRM) as shown in Table 1. |
MassLynx Software v4.1 with TargetLynx Application Manager
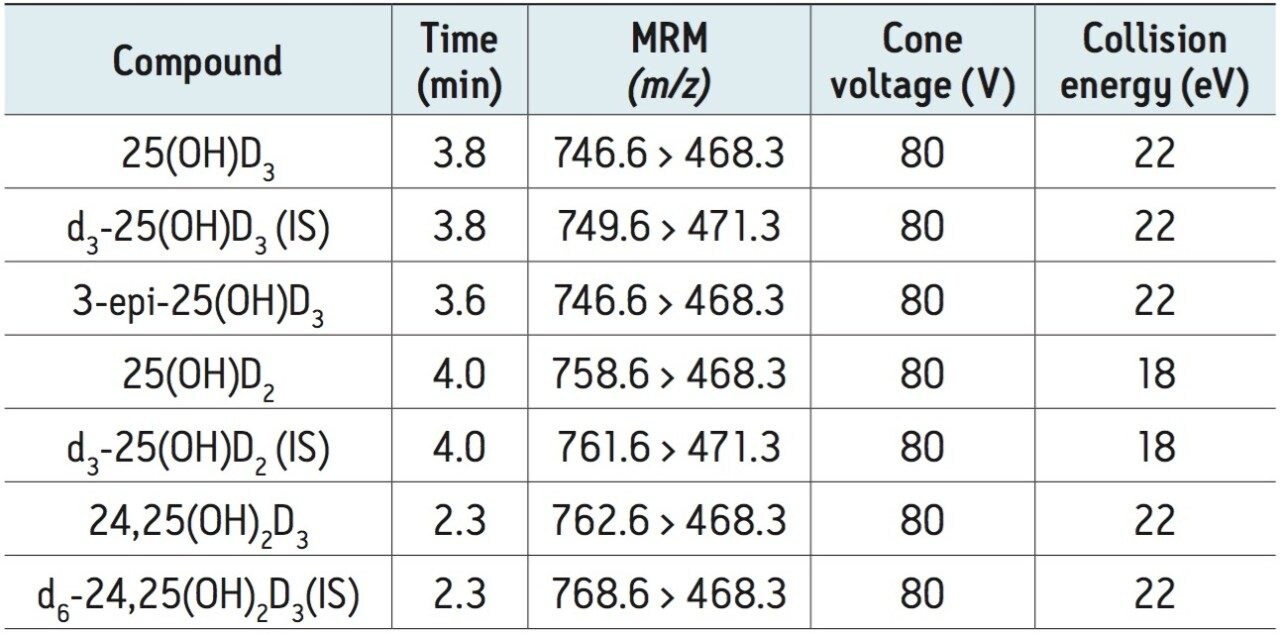
For this clinical research method, derivatization of vitamin D metabolites with DMEQ-TAD offers the advantage of improving ionization efficiency relative to native metabolites. This derivatisation also produces an increase in molecular mass of 336 Da which results in reduced background interference to precursor and product ions.4 The major characteristic ions for derivatized 25(OH)D3, 25(OH)D2, and 24,25(OH)2D3, were the protonated species [M+H]+ at m/z 746.6, 758.6, and 762.6, respectively (Table 1), and when subjected to collision-induced dissociation under optimized conditions yield A-ring/DMEQ-TAD fragment (m/z 468), and DMEQ-TAD fragment (m/z 247) moieties as the major products, as shown for the 25(OH)D3 adduct in Figure 2. The m/z 468 fragment ion was chosen for MRM analysis because of greater selectivity and lower background as compared with m/z 247. DMEQ-TAD adducts of the target analytes consisted of 6R and 6S isomers,4 of which the more abundant 6S isomer was used for quantification.
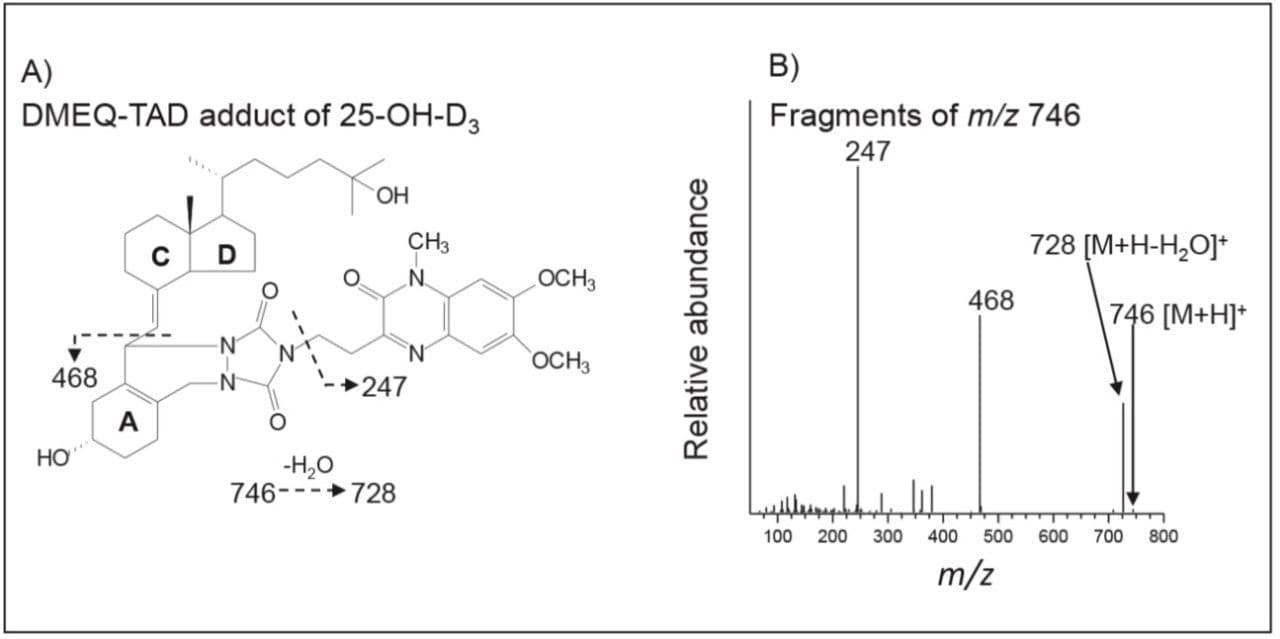
In a representative serum sample, the 6S isomers of the DMEQ-TAD adducts of 25(OH)D3, 25(OH)D2, and 24,25(OH)2D3 eluted at 3.80, 4.02, and 2.30 minutes respectively as shown in Figure 3. The peak eluting at 3.58 co-migrated with synthetic 3-epi-25(OH)D3, characterized by a single broad peak suggesting co-elution of the 6R and 6S isomers for this analyte. 3-epi-25(OH)D3 is a known isobar of 25(OH)D3. Since these two analytes were chromatographically resolved, the presence of 3-epi-25(OH)D3 did not confound 25(OH)D3 measurement using the current system.
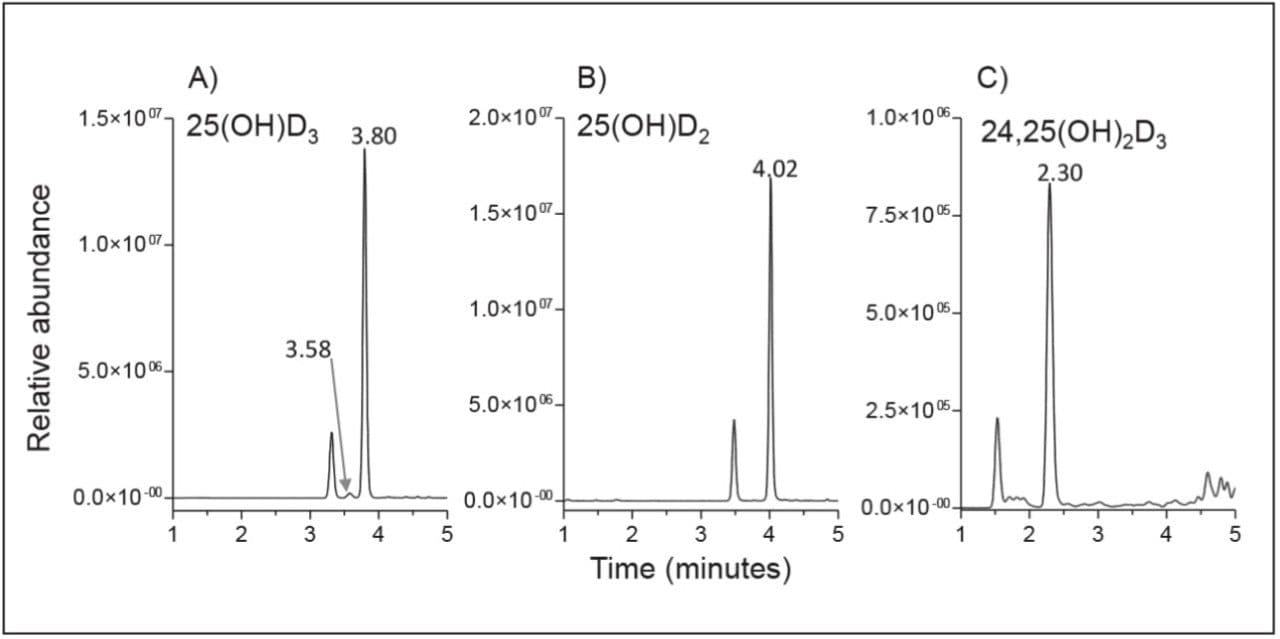
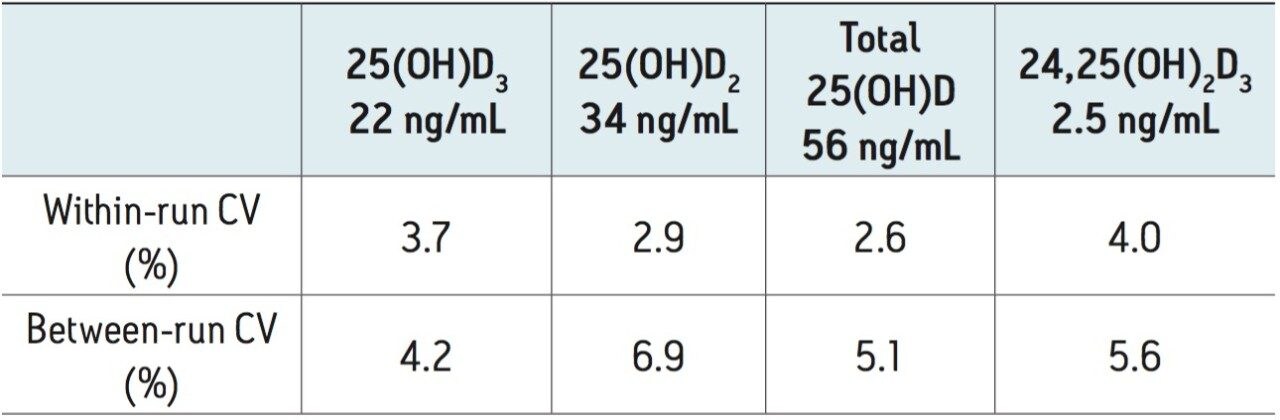
Over the calibration range of 25(OH)D3 (3.8-148 ng/mL), 25(OH)D2 (4.9-120 ng/mL), and 24,25(OH)2D3 (0.4-11.6 ng/mL) the method was shown to be linear for each analyte giving representative r2 values of at least 0.997 (Figure 4). Lower limits of quantification as defined by signal-to-noise ratios of ≥10, were estimated to be within the 0.1-0.2 ng/mL range and lower limits of detection (signal-to-noise ≥3) were estimated to be as low as 0.04 ng/mL. Within-run and between-run imprecision was determined by analyzing five replicates of a serum sample on each of 14 assay days. Mean within-run CVs for the target analytes ranged from 3-4% and between-run CVs ranged from 4-7% (Table 2). For 25(OH)D3 and 25(OH)D2, method accuracy was assessed by analyzing serum samples distributed by the Vitamin D External Quality Assessment Scheme (DEQAS) on each assay day. Mean discrepancies from the all-laboratory trimmed mean, and LC-MS/MS method mean were - 2% and -5% respectively based on 77 DEQAS samples analyzed over 14 days of analysis.
This method demonstrates that 24,25(OH)2D3 can be reliably measured using the Waters ACQUITY UPLC System and Xevo TQ-S Mass Spectrometer. A robust and analytically sensitive method has been developed, using small sample volumes, that is well suited to clinical research studies.
720005364, May 2015