This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the use of HILIC with an ACQUITY UPLC Glycoprotein BEH Amide 300Å Column to assay the glycan occupancy of intact mAbs.
Unprecedented Hydrophilic Interaction Chromatography (HILIC) separations of intact mAb glycan occupancy variants using the ACQUITY UPLC Glycoprotein BEH Amide 300A 1.7μm Column.
Monoclonal antibodies (mAbs) have emerged as some of the most important therapeutics on the market. These mAbs that are prescribed for therapeutic use are most often expressed from eukaryotic cell lines, such as CHO, and, as a result, are N-glycosylated at two consensus site asparagine residues in the Fc portion of their heavy chains. Since glycosylation can be a measure of efficacy, safety and manufacturing conditions, it is often critical to characterize and routinely monitor the N-glycan profile of a mAb drug substance.1-2 The recent introduction of the GlycoWorks RapiFluor-MS N-Glycan Kit has made it significantly easier to perform highly detailed HILIC-based, released N-glycan analyses and to thereby elucidate the heterogeneity of N-glycosylation.3-4 However, it is also critical to determine the extent to which the asparagine linkage sites are occupied with N-glycans, particularly since incomplete glycosylation dramatically changes the effector functions of a mAb.5 Traditionally, this assessment has been performed by a sized-based Capillary Electrophoresis-Sodium Dodecyl Sulfate (CE-SDS) separation of the heavy chains resulting from reduction of a mAb.6,7 To instead directly assess glycan occupancy variants for intact mAbs, we have developed an LC separation based on HILIC. To achieve these unprecedented separations, a new, purposefully designed HILIC column was employed. This new column, the ACQUITY UPLC Glycoprotein BEH Amide 300Å 1.7 µm Column, contains a wide-pore amide bonded stationary phase that ensures that mAb species have access to the porous network of the stationary phase and are not significantly impaired by restricted diffusion.8
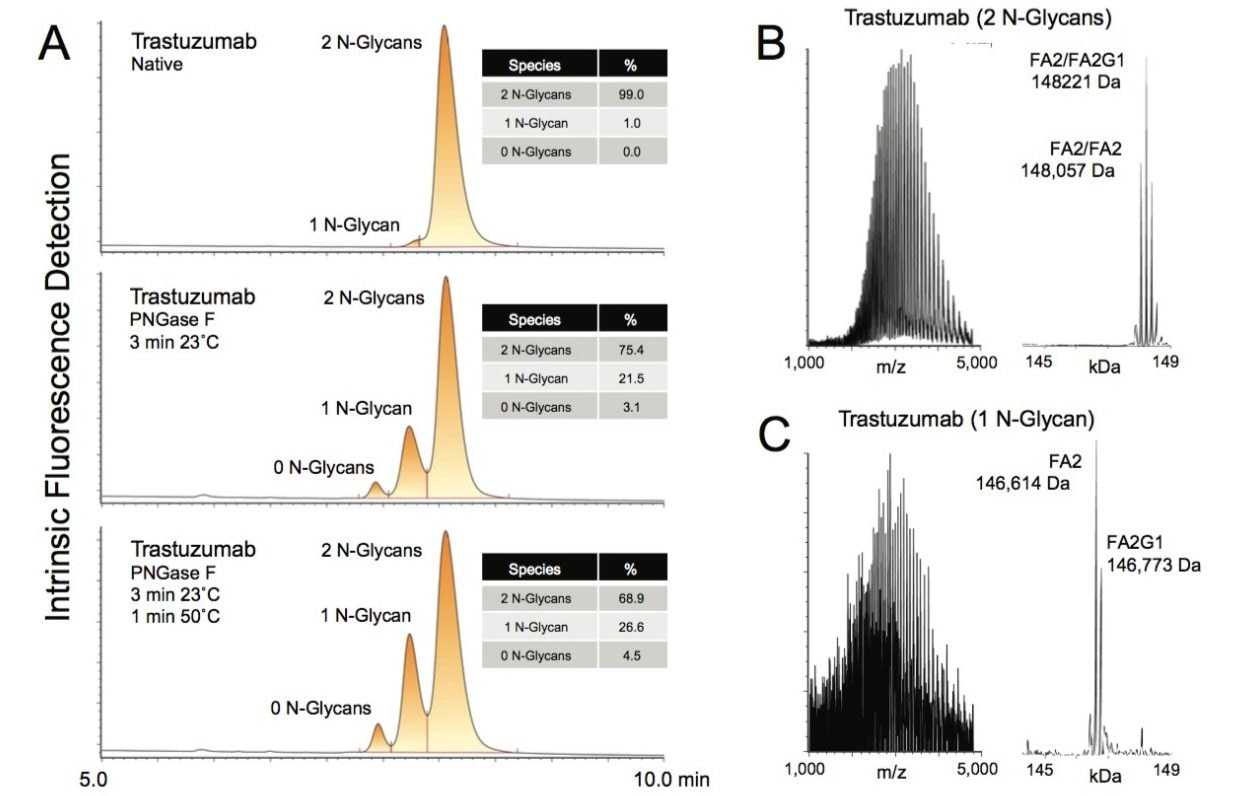
A high-throughput, high resolution HILIC separation was established for intact mAbs using a 2.1 x 150 mm wide-pore BEH amide column, a 0.4 mL/min flow rate, and a column temperature of 80 ˚C. In addition, two mobile phase additives, 0.1% trifluoroacetic acid (TFA) and 0.3% hexafluoroisopropanol (HFIP) were employed to improve the solubility of intact mAbs in the high organic, initial mobile phase conditions of the HILIC gradient. To enhance the sensitivity of this LC method, the intact proteins were detected by means of their intrinsic fluorescence. Excitation and emission wavelengths of 280 and 320 nm were found to provide optimal signal-to-noise and consistently flat chromatographic baselines that are desirable for peak integration. A representative set of chromatograms resulting from this 15 minute LC method is shown in Figure 1A. Three chromatograms are displayed. The top chromatogram shows trastuzumab as injected from a dilution of its formulation, while the other two chromatograms show samples of trastuzumab after being subjected to partial PNGase F deglycosylation. Deglycosylated samples of trastuzumab clearly showed three distinct peaks in their HILIC profiles, as was predicted if the glycan occupancy variants of a mAb were to be resolved. The unadulterated sample of trastuzumab contained measurable levels of only the fully occupied and singly occupied forms (1%) as confirmed by online mass analysis. The deconvoluted mass spectrum corresponding to the main LC peak exhibited several masses, such as 148,057 Da and 148,221 Da, that are within 2 Da of the theoretical molecular weights for the predominant glycoforms of trastuzumab (Figure 1B).9 In contrast, the deconvoluted mass spectrum for the smaller, less strongly retained peak showed reduced heterogeneity and masses that were lighter by approximately 1000 to 2000 Da, consistent with the single occupancy form (Figure 2B). Fully aglycosylated species were not detected in this sample. An interesting observation from these data is that the levels of fully deglycosylated forms are lower than would be predicted if both sites were deglycosylated at the same rate. In these examples, the observed level of fully deglycosylated forms is approximately one-third lower than would be predicted. This may indicate that either the digestion rate for one of the N-glycans is slower than the other, or that upon removal of the first N-glycan, the digestion rate of the remaining N-glycan is reduced.
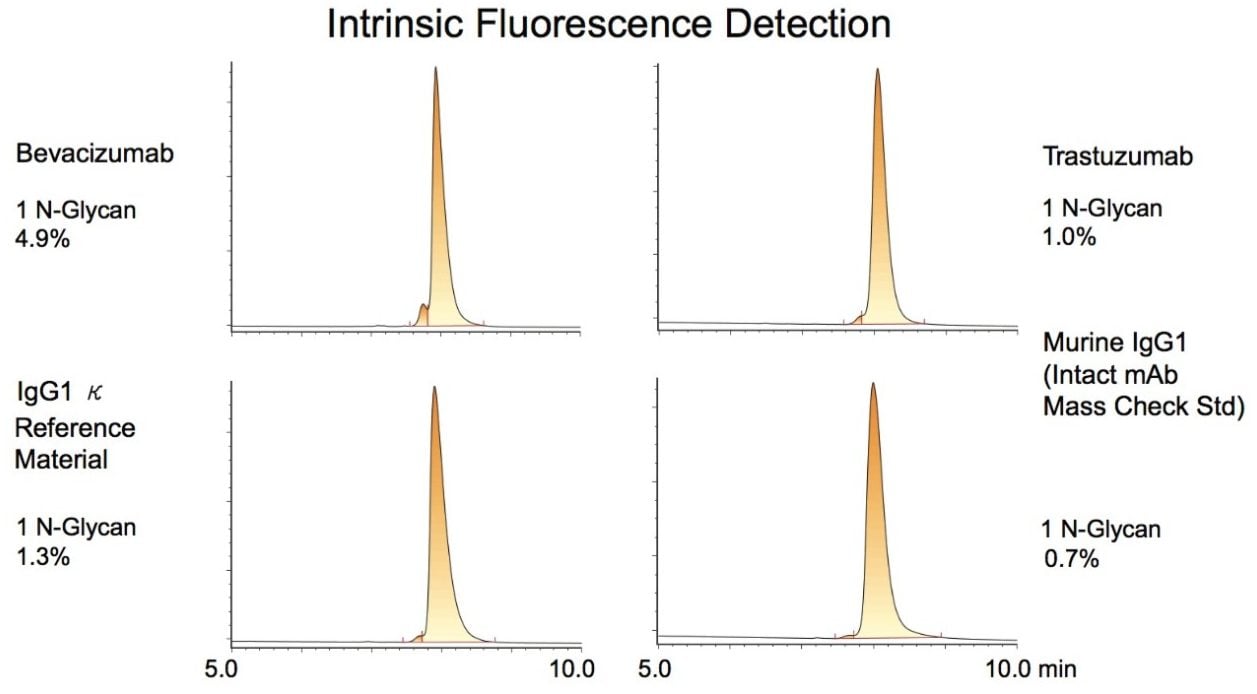
To assess the applicability of this technique to other mAbs, we analyzed three additional IgG samples. Results on trastuzumab, bevacizumab, a candidate IgG1κ reference material, and a murine IgG1 (Intact mAb Mass Check Standard, p/n 186006552) are shown in Figure 2. Integrations on the HILIC-fluorescence chromatograms indicated that these samples contain 1 N-glycan (incomplete glycosylation) variants at relative abundances ranging from 0.7 to 4.9%. These observations suggest that this HILIC method could be an attractive technique for assaying incomplete glycosylation of mAbs down to levels well below 1% for the 1 N-glycan form. Detection limits for the 0 N-glycan form may perhaps be even lower. Of these samples, bevacizumab might be predicted to have the highest abundance of the 0 N-glycan form. Indeed, the bevacizumab profile presented a peak, albeit very minor, with a retention time consistent with a 0 N-glycan species, the area of which would contribute to the overall profile at a level of only 0.05%. Such an observation suggests that the 0 N-glycan level is lower than would be statistically predicted; however, rigorous determination of the quantitative limits of this analysis and the identity of this putative 0 N-glycan peak would need to be evaluated to confirm that this is indeed the case.
This strategy for measuring mAb glycan occupancy is most appealing in that it requires minimal, if any, sample preparation. We have observed that some interferences can be encountered that are due to partially fragmented and/or reduced mAb species. In which case, as with the Intact mAb Mass Check Standard, a simple, centrifugal filtration clean-up step with a 100kDa MWCO polyethersulfone membrane was sufficient to minimize such interferences. Given the unique selectivity of the HILIC separation, it should also be possible to address potential interferences by performing offline or online 2D-LC, wherein a size exclusion or reversed phase separation could be coupled to the wide-pore amide HILIC separation. Future investigations could also include reducing glycan heterogeneity (via a sialidase or other exoglycosidase) to allow the mAb glycan occupancy variants to be more discretely resolved. Similarly, there is an opportunity to use this separation in combination with enzymes that generate Fc subunits.
In addition to profiling the heterogeneity of glycosylation, it is also critical to assay glycan occupancy. Here, we demonstrate that a Glycoprotein BEH Amide column, purposefully designed for large molecule HILIC separations, can be used to directly quantify incomplete N-glycan occupancy in intact mAb samples. Unlike a conventional CE-SDS separation of reduced mAbs, this technique provides a non-inferred assessment on the nature (i.e. 1 N-glycan versus 0 N-glycans) of glycan occupancy for the intact mAb. The proposed HILIC methodology is also MS-compatible, making it possible to readily confirm the assignments of observed peaks.
720005435, June 2015