
In this application note, we illustrate the transfer of a robust HPLC monosaccharide method to the ACQUITY UPLC H-Class Bio System running as an HPLC, using monosaccharide standards as well as bovine fetuin and the commercially available monoclonal antibody cetuximab as analytes.
Evidence for reliable and consistent glycosylation of glycoprotein therapeutics is typically obtained through LC-based analysis of N-linked glycans. On occasion, regulatory agencies request information of monosaccharide content as an orthogonal technique for confirming the glycan composition. Beyond profiling changes in total glycan composition, monosaccharide analysis can also be used as an exploratory technique for identifying various monosaccharide modifications, including phosphorylation and sulfation, which can be particularly challenging to discern when analyzing at the released glycan level in the absence of mass spectrometry.
Initial investigation of monosaccharide analyses resulted in a reliable and informative HPLC-based approach using 2-aminobenzoic acid (2AA)-derivatized glycan hydrolysates where each of the individual monosaccharides were successfully separated using reversed-phase chromatography. Since this time, the analytical demands placed on development and QC labs that are tasked with characterizing and monitoring glycoprotein therapies have increased dramatically. Assays used to monitor changes in glycosylation therefore need to be updated in order to maximize productivity through improved analytical efficiency.
In this application note, we illustrate the transfer of a robust HPLC monosaccharide method to the ACQUITY UPLC H-Class Bio System running as an HPLC, using monosaccharide standards as well as bovine fetuin and the commercially available monoclonal antibody cetuximab as analytes. Geometric scaling of the presented method is then performed in a stepwise manner to demonstrate the advantages of transferring legacy HPLC monosaccharide analyses to UPLC technology as a means of decreasing assay time.
|
Waters ACQUITY UPLC H-Class Bio System, comprised of: |
ACQUITY UPLC H-Class Bio Quaternary Solvent Manager (QSM) ACQUITY UPLC H-Class Bio Sample Manager (FTN) ACQUITY UPLC H-Class Bio Column Heater (CH-A) ACQUITY UPLC FLR Detector Extension loop: 100 μL |
|
Waters columns: |
XBridge C18 5 μm, 130 Å, 4.6 x 100 mm XBridge C18 3.5 μm, 130 Å, 2.1 x 100 mm XBridge C18 XP 2.5 μm, 130 Å, 2.1 x 100 mm ACQUITY UPLC BEH C18 1.7 μm, 130 Å, 2.1 mm x 100 mm |
|
Mobile phase A: |
0.2% N-butylamine, 0.5% phosphoric acid, and 1% THF in H2O |
|
Mobile phase B: |
50% mobile phase A in acetonitrile |
|
Excitation wavelength: |
360 nm |
|
Emission wavelength: |
425 nm |
|
Column temp.: |
30 °C |
|
Injection vol.: |
4.6 mm x 100 mm format, 4.8 μL, 2.1 mm x 100 mm format, 1 μL |
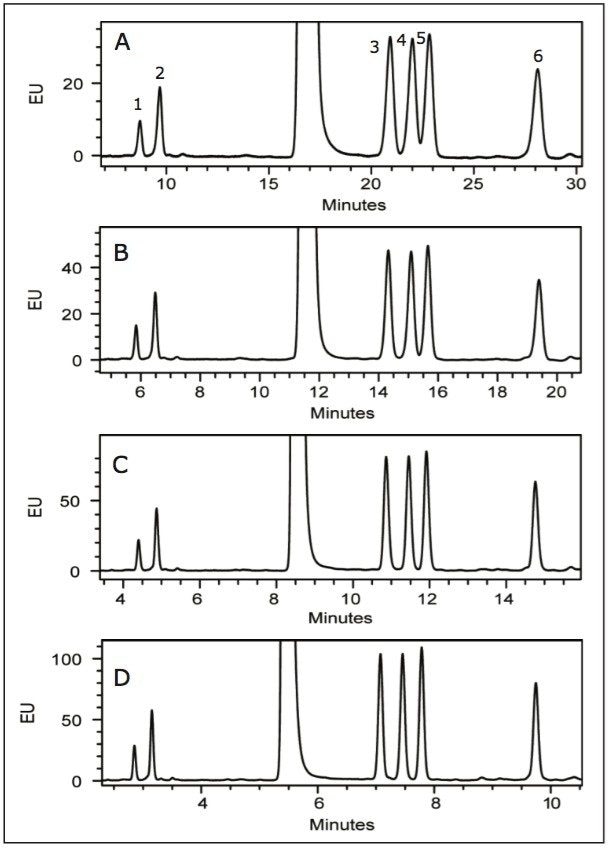
Following the described approach, typical run times for HPLC-based monosaccharide analysis are reduced from 45 minutes to just 17 minutes. Importantly, chromatographic resolution between measured critical peak pairs is observed to improve with migration to smaller column particle sizes. Selectivity is unaffected due to the availability of reversed-phase column chemistries in a number of particle sizes and dimensions. The data presented here indicate migration to UPLC technology offers significant advantages for improving monosaccharide chromatographic quality.
Derivatization of monosaccharides was performed as previously described,1, 2 with a number of minor modifications as recommended by Stepan and Staudacher.3 Monosaccharides from bovine fetuin were released by acid hydrolysis using 2 M TFA with hydrolysis occurring for 3 h at 100 °C. Resulting hydrolysates were then dried by centrifugal evaporation followed by reconstitution in 5 μL of 80 mg/mL sodium acetate trihydrate. A 2AA labeling solution was prepared by dissolving 30 mg of 2AA in 1 mL of 2% (w/v) boric acid in methanol. This suspension was then used to dissolve 30 mg of sodium cyanoborohydride. Of this preparation, 10 μL was added to each of the monosaccharide mixtures. Monosaccharides were labeled at 80 °C for 60 min. Upon completion of labeling, serial dilutions were performed to generate a 1000 fold dilution of the labeled material. For preparation of monosaccharide standards, labeling was performed as outlined above with the omission of acid hydrolysis.
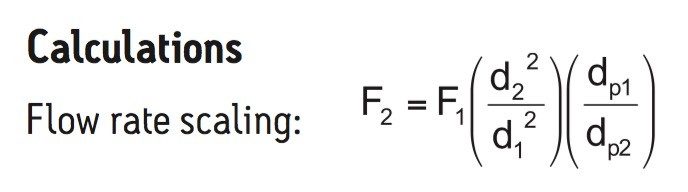
F refers to flow rate, d refers to column I.D., and dp refers to particle diameter. In each case, 2 refers to the new column and 1 refers to the original column.

Vi refers to injection volume, r refers to column internal radius, and L refers to column length. In each case, 2 refers to the new column and 1 refers to the original column.
To verify the ability of the ACQUITY UPLC H-Class Bio to run legacy analyses of 2AA-derivatized monosaccharides, we first established an HPLC separation using method conditions based on previously described chromatographic conditions1,2 To evaluate the proposed method, a reference standard mix was prepared by combining individual monosaccharides into a common mix. This mix included the typical monosaccharides expected in biologically relevant samples, namely N-acetylglucosamine (GlcNAc), N-acetylgalactosamine (GalNAc), glucose (Glc), mannose (Man), galactose (Gal), xylose (Xyl), and fucose (Fuc). In addition to these standards, two glycoprotein samples were also selected to determine the accuracy of this approach in determining monosaccharide composition. The first glycoprotein selected was bovine fetuin, a protein known to contain both N- and O-glycosylation sites. The second glycoprotein selected was the commercial monoclonal antibody cetumixab.
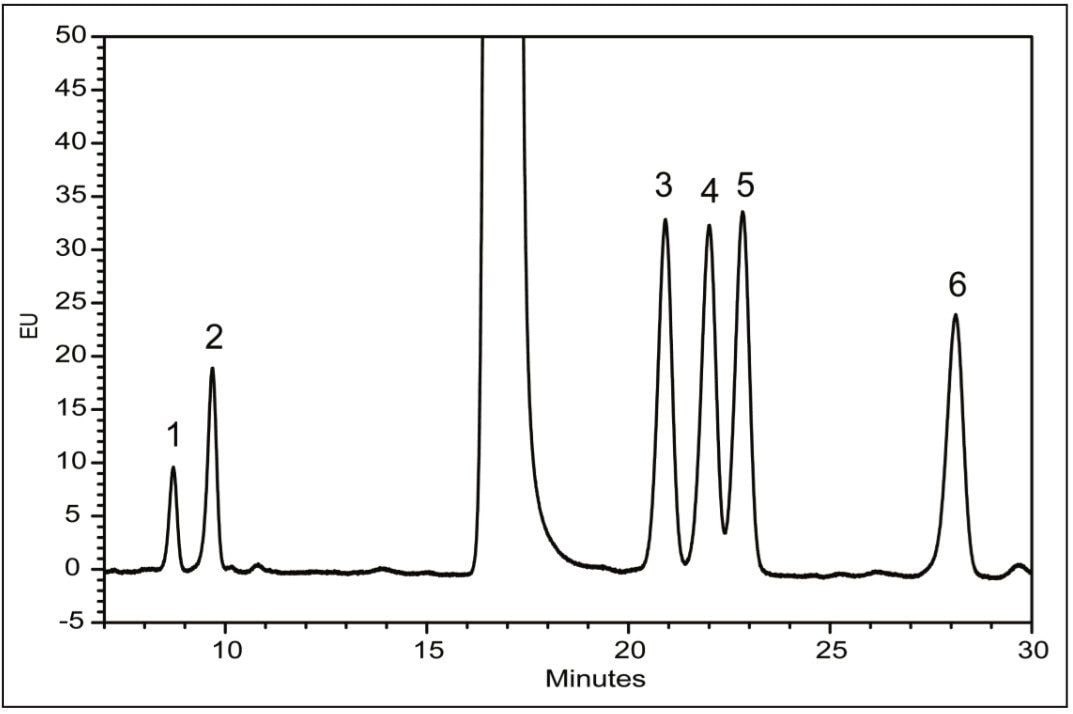
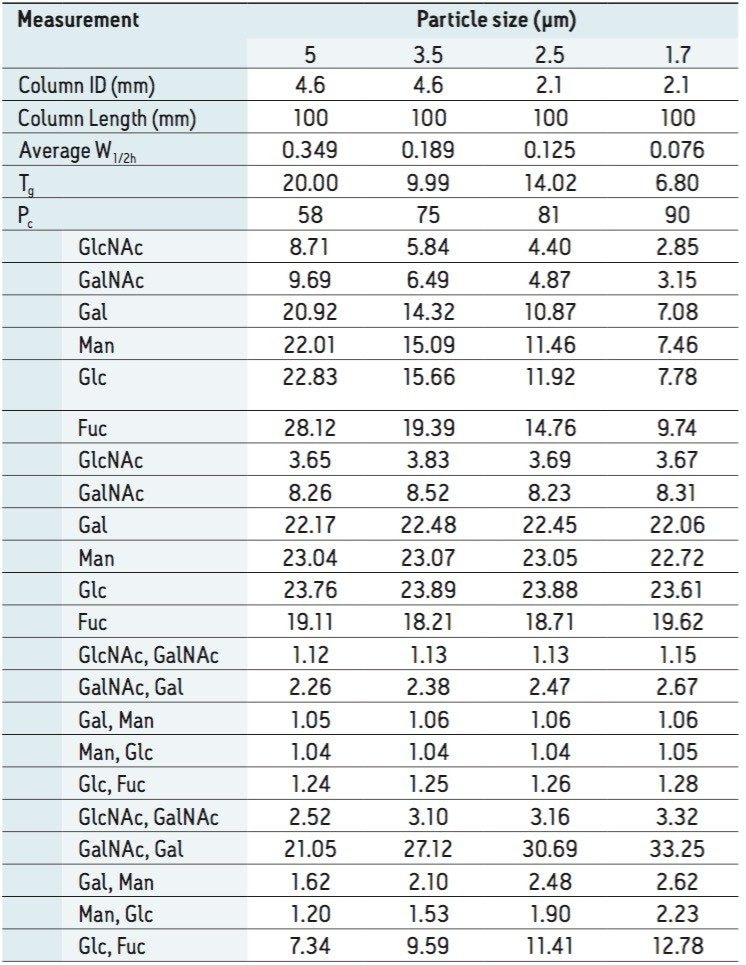
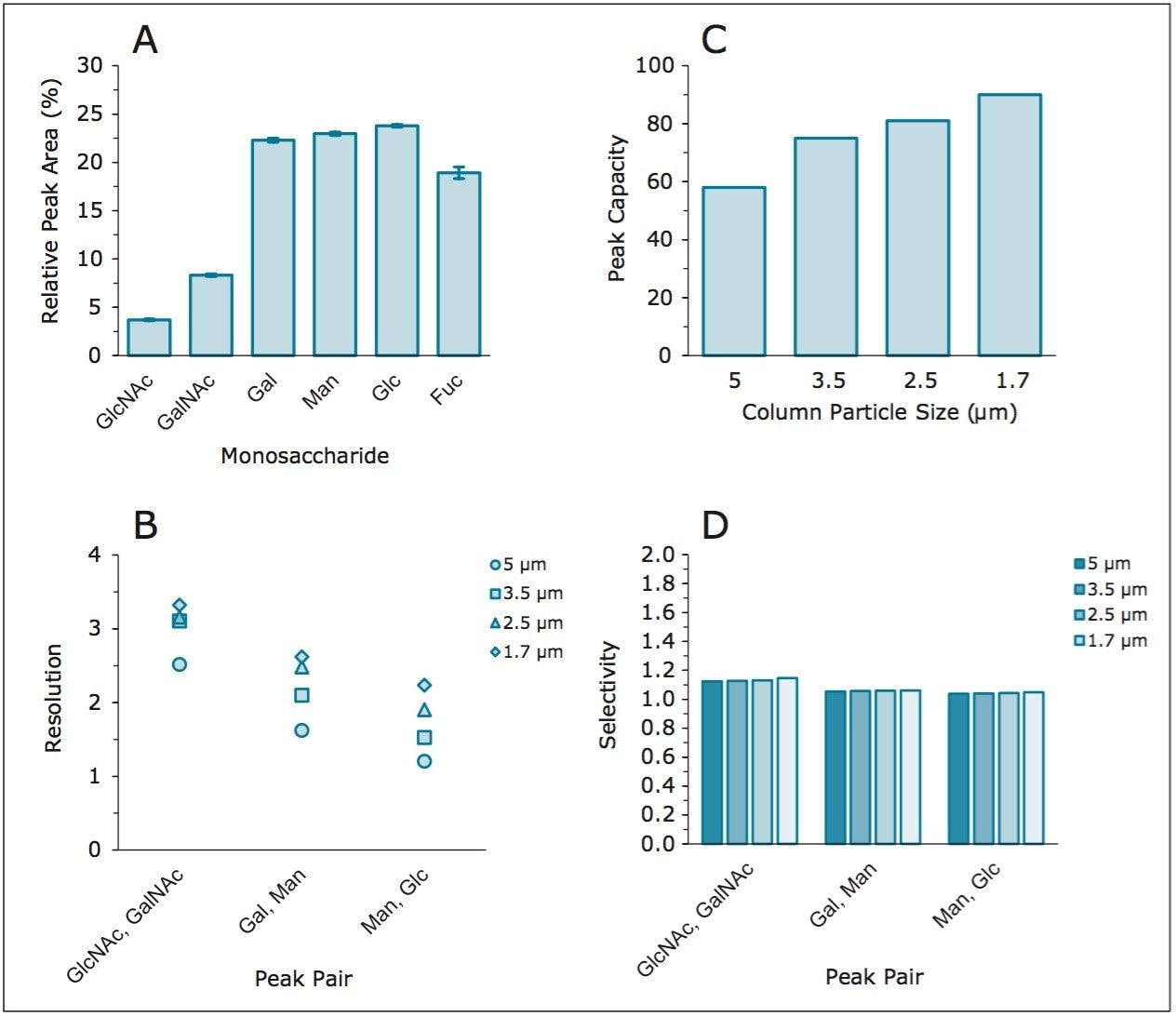
Following hydrolysis from the glycoprotein samples and 2AA derivatization, monosaccharides were separated using the aforementioned method. The resulting HPLC chromatogram (Figure 1) acquired on the ACQUITY UPLC H-Class Bio is consistent with previously published data.2 In terms of chromatographic performance, peak capacity was measured together with selectivity and resolution between critical peak pairs. These data are summarized in Table 1. Consistent peak area was observed across separations using all particle sizes (Figure 2d). These data confirmed the separation of all relevant components and, therefore, established a suitable method for monosaccharide method scaling to UPLC technology.
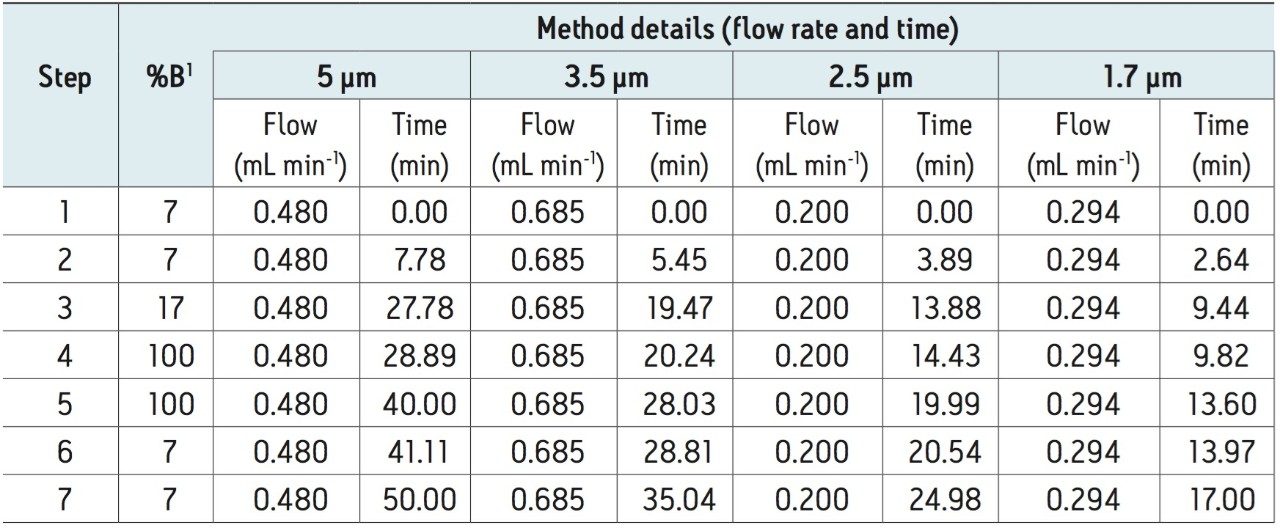
In an effort to improve throughput of analyses and general chromatographic quality, the above described method was geometrically scaled in a stepwise manner to UPLC column technology. This involved scaling the flow rate to the new column dimensions and adjusting individual steps in the gradient method to deliver equivalent column volumes as itemized in the original recommended HPLC method. Details of the UPLC method can be found in the experimental section of this application note.
Several particle sizes of identical chemistry ranging from 1.7 to 5 μm were used in the scaling exercise. In the case of the 1.7 μm and 2.5 μm particles, a 2.1 mm x 100 mm column dimension was used while 4.6 mm x 100 mm column dimensions were used for the 3.5 μm and 5 μm particles. Flow rate and injection volume scaling calculations specific for individual column dimensions were determined using the appropriate equations defined in the experimental section of this application note. The duration for each step in the gradient table was subsequently modified based on the new flow rate and column volume to ensure consistent delivery of equivalent column volumes per change in organic composition when compared to the original method. The results of these calculations are summarized in the experimental section of this application note where flow rates and gradient step durations are itemized with respect to each column particle size.
To evaluate the results of method scaling, the monosaccharide mix was separated under the new gradient conditions for each particle size and column dimensions (Figure 2). Method scaling to UPLC column technology reduced the total required run time of the method from 50 min (in the case of the 5 μm column, Figure 2a) to just 17 min (in the case of the 1.7 μm column, Figure 2d), an improvement in efficiency of approximately 66%. Details of chromatographic performance are presented in Table 1.
In general, peak capacity was shown to increase with decreasing particle size (Figure 3a), an expected outcome based on the narrower peaks achieved with UPLC technology. The reduction in run time was not at the cost of resolution, where a general improvement was observed across all critical peak pairs as column particle size decreased (Figure 3b). Selectivity remained unaffected mainly due to the availability of identical column chemistry across multiple particle sizes (Figure 3c). Changes in column particle size did not impact relative peak area determination, evidenced by averaged peak areas for each monosaccharide across all column formats (Table 1 and Figure 3d). Taken together, scaling of the original monosaccharide method produced improved resolution in a shorter amount of time, with negligible impact to selectivity.
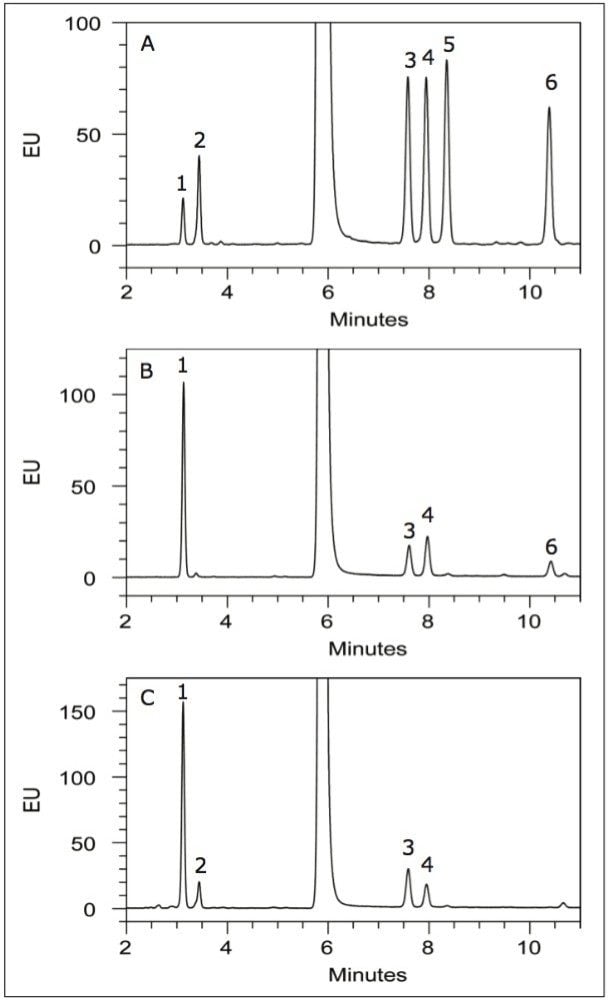
To verify this approach in determining monosaccharide composition, we analyzed both fetuin and cetuximab 2AA-labeled monosaccharides. Fetuin is known to contain O-glycosylation and therefore should demonstrate the presence of GalNAc within its profile. Cetuximab, on the other hand, contains no O-glycosylation and should therefore be absent of any GalNAc.
To perform the analysis, the 1.7-μm BEH C18 particle chemistry was used and results of the separations were compared to the monosaccharide standard (Figure 4a). GalNAc is clearly detected in the fetuin sample (Figure 4b), as evidenced by a chromatographic peak with similar retentivity to the GalNAc standard. Also consistent with literature is the absence of core fucosylation in fetuin, largely evidenced by the absence of a peak with similar retentivity to the fucose standard. This finding is consistent with previous reports of bovine fetuin N-glycosylation characterization.4,5 Analysis of cetuximab reveals all individual monosaccharides with the exception of xylose, a monosaccharide not typically associated with mammalian N-linked glycosylation (Figure 4c). Mannose was found to be in higher quantity than galactose, a result consistent with previous literature indicating the presence of several high mannose structures in cetuximab.6-8
As a complementary approach to released glycan analysis, monosaccharide profiling allows analysts to verify glycan composition determined in traditional HILIC-based separations. A well-established assay for monosaccharide analysis uses reversed-phase chromatography to separate 2AA-derivatized monosaccharides. In this application note, we have demonstrated the ability of the ACQUITY UPLC H-Class Bio System for running both HPLC and UPLC methods for monosaccharide analyses. This HPLC assay provided sufficient resolution of individual monosaccharides but was restricted in part by the time required to perform the separation. A significant reduction in runtime was obtained by transferring the legacy HPLC method to UPLC technology. With the modernized, UPLC-based separation, a higher throughput assay for monosaccharide analysis was thereby achieved.
720005255, March 2015