In the following application, orthogonal detection techniques – namely, PDA and mass detection – in combination with HPLC, were employed to identify and quantitate Irbesartan and related compound A. An automated solvent manager with flow splitting was included in the system configuration to align the dynamic ranges of the two detectors. The identity of the peaks was confirmed by mass detection with the ACQUITY QDa Detector. This chromatographic configuration displayed the required area and retention time reproducibility, in addition to reliable quantitation.
High Performance Liquid Chromatography (HPLC) is one of the most widely applied analytical techniques, and can serve as a powerful separation technique for chemical mixtures consisting of a wide range of compounds including isomers. HPLC, with its choice of detection techniques, can separate analytes from a mixture, but provides little information about what a compound might be. Using photodiode array (PDA) detection can provide some information about peak homogeneity or presence of coeluting peaks, but can only provide identification by comparing standards in the same mobile phase. Adding a mass detector to an HPLC-PDA system can provide information for peak identification, for recognizing coeluting chromatographic peaks, and for confirming peak homogeneity. Mass spectrometry is, however, often perceived to be too complicated, time consuming, and expensive. Moreover, the two detectors have different sensitivity levels, and therefore combining the two detection techniques may require analyzing the sample on two different systems.
In the following application, orthogonal detection techniques – namely, PDA and mass detection – in combination with HPLC, were employed to identify and quantitate Irbesartan and related compound A. An automated solvent manager with flow splitting was included in the system configuration to align the dynamic ranges of the two detectors. The identity of the peaks was confirmed by mass detection with the ACQUITY QDa Detector. This chromatographic configuration displayed the required area and retention time reproducibility, in addition to reliable quantitation. The Alliance HPLC System, with orthogonal detection, and Waters CDS (Empower or MassLynx Software), capable of combining data from orthogonal detectors, help increase confidence in compound identification of pharmaceutical products.
An accurately weighed quantity of Irbesartan standard was dissolved in methanol to obtain a 100 mL stock solution of 1 mg/mL. A series of different concentrations of Irbesartan-related compound A in the presence of a constant concentration of Irbesartan was analyzed. The calibration curve was run with different concentrations of Irbesartan-related compound A prepared at six levels (0.0005 mg/mL, 0.0010 mg/mL, 0.005 mg/mL, 0.010 mg/mL, 0.050 mg/mL, and 0.250 mg/mL), and a fixed amount of Irbesartan standard at 0.100 mg/mL in methanol.
|
LC system: |
Alliance HPLC with ISM and Alliance Splitter Kit, 2998 PDA Detector, and ACQUITY QDa Detector |
|
Run time: |
25 min |
|
Column: |
XSelect HSS T3, 4.6 mm x 250 mm, 5 μm (L1) (p/n 186004793) |
|
Column temp.: |
40 °C |
|
Mobile phase: |
Water (Solvent line A) Acetonitrile (Solvent line B) 1% Formic acid in water (Solvent line C) |
|
Separation Mode: |
Gradient |
|
Wash solvent: |
70:30 water/acetonitrile |
|
Flow rate: |
1.0 mL/min |
|
Injection volume: |
10 μL |
|
Time(min) |
%A |
%B |
%C |
|---|---|---|---|
|
- |
70 |
10 |
20 |
|
5.0 |
30 |
50 |
20 |
|
8.0 |
30 |
50 |
20 |
|
12.0 |
0 |
80 |
20 |
|
18.0 |
0 |
80 |
20 |
|
18.1 |
70 |
10 |
20 |
|
25.0 |
70 |
10 |
20 |
|
Wavelength range |
210 nm to 400 nm at 1.2 nm bandwidth |
|
Detection wavelength |
254 nm |
|
Data rate |
5 points/s |
|
Time constant |
0.4 s (normal) |
|
ISM conditions Solvent |
90:10 water/acetonitrile with 0.1% formic acid |
|
Flow rate |
300 μL/min |
|
Splitter kit |
Alliance Splitter Kit (p/n 205000917) |
|
MS: |
ACQUITY QDa |
|
Detector Ionization mode: |
ESI+ |
|
Capillary voltage: |
0.8 V |
|
Cone voltage: |
15 V |
|
Sampling rate: |
2 points/s |
|
Probe temp: |
300 °C |
|
Scan range: |
100 – 600 Da |
|
SIR masses [M + H]: |
429.4 (Irbesartan API) 447.3 (Irbesartan-related compound A) |
|
Data management: |
Empower 3 FR2 CDS |
|
USP System Suitability Criteria: |
% Area RSD ≤1.5 for 5 replicate injections |
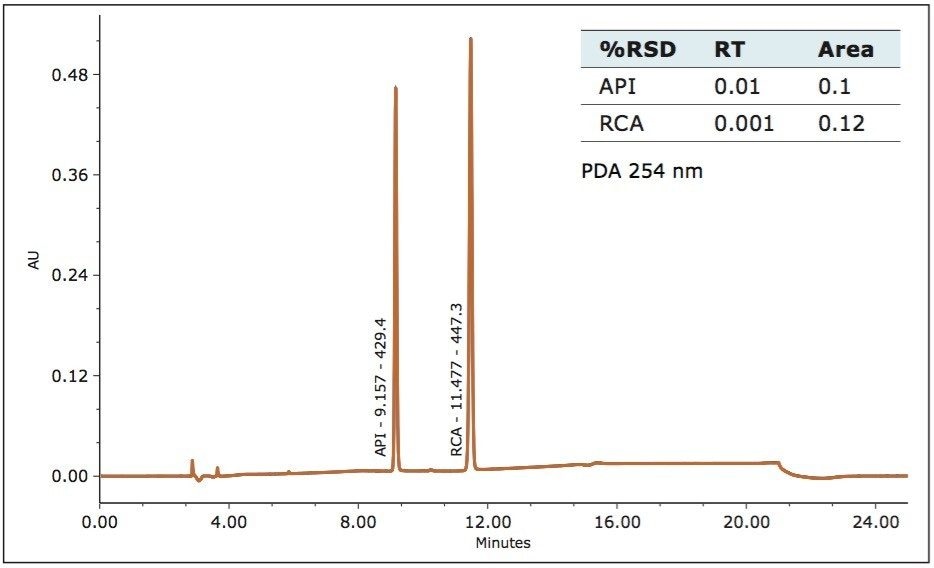
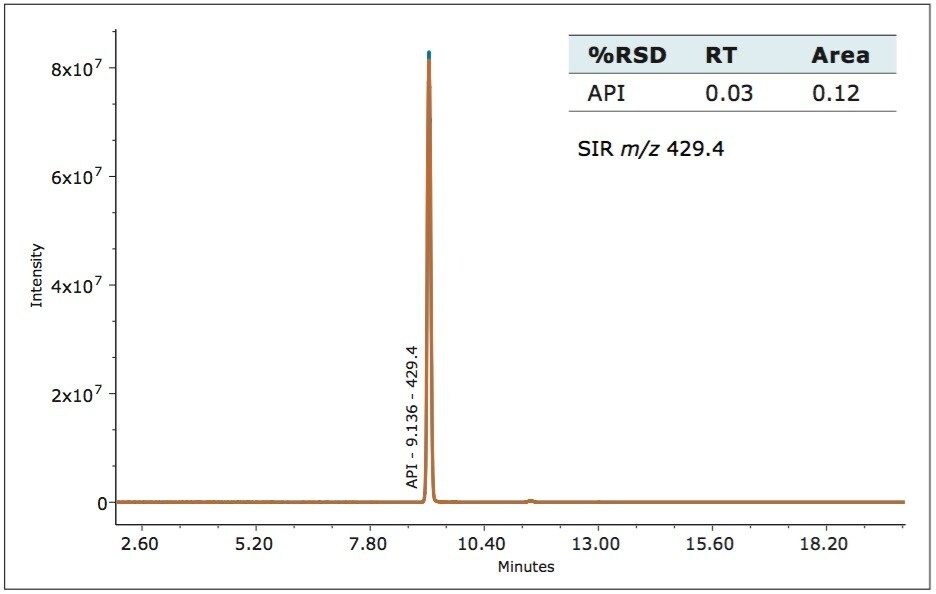
The ACQUITY QDa Detector is a mass detector that detects a different physical property of an analyte, as compared to the Waters 2998 PDA Detector. In order to employ mass detection, the non-volatile USP-specified buffer was replaced with a volatile mobile phase containing formic acid, (see gradient table). The analytical method conditions separated irbesartan-related compound A from the standard. The identity of the peaks was confirmed using m/z of the analyte by the ACQUITY QDa Detector. The system’s routine performance was evaluated using six injections prepared at 0.100 mg/ml concentration for the irbesartan API and related compound A. Retention time and area reproducibility are shown in figures 2, 3, and 4. All values meet the typical assay requirements. The area of the related compound A in the MS trace falls within acceptable limits (Figure 4). It is, however, slightly more variable than irbesartan API because the signal is smaller in intensity by a factor of 10 compared to the API.
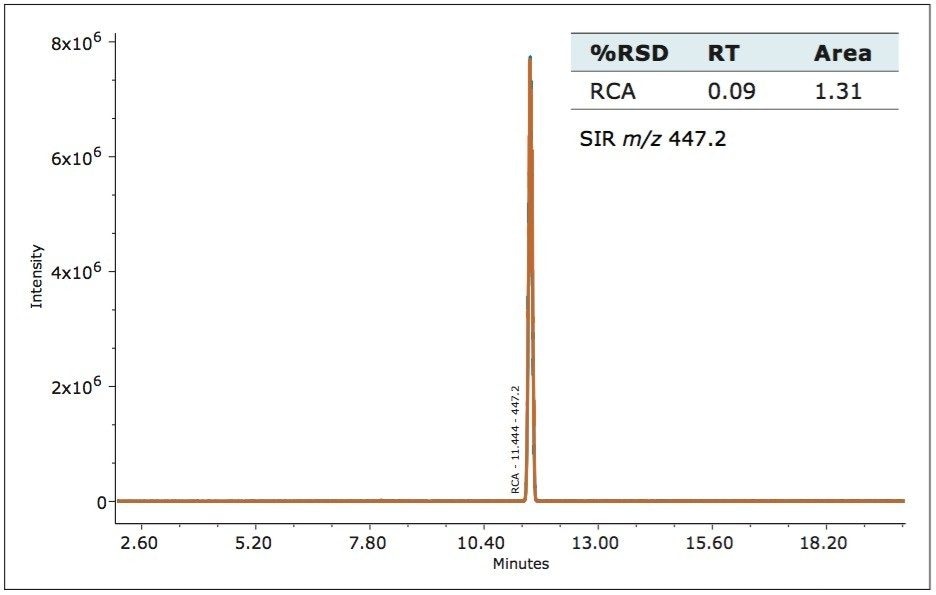
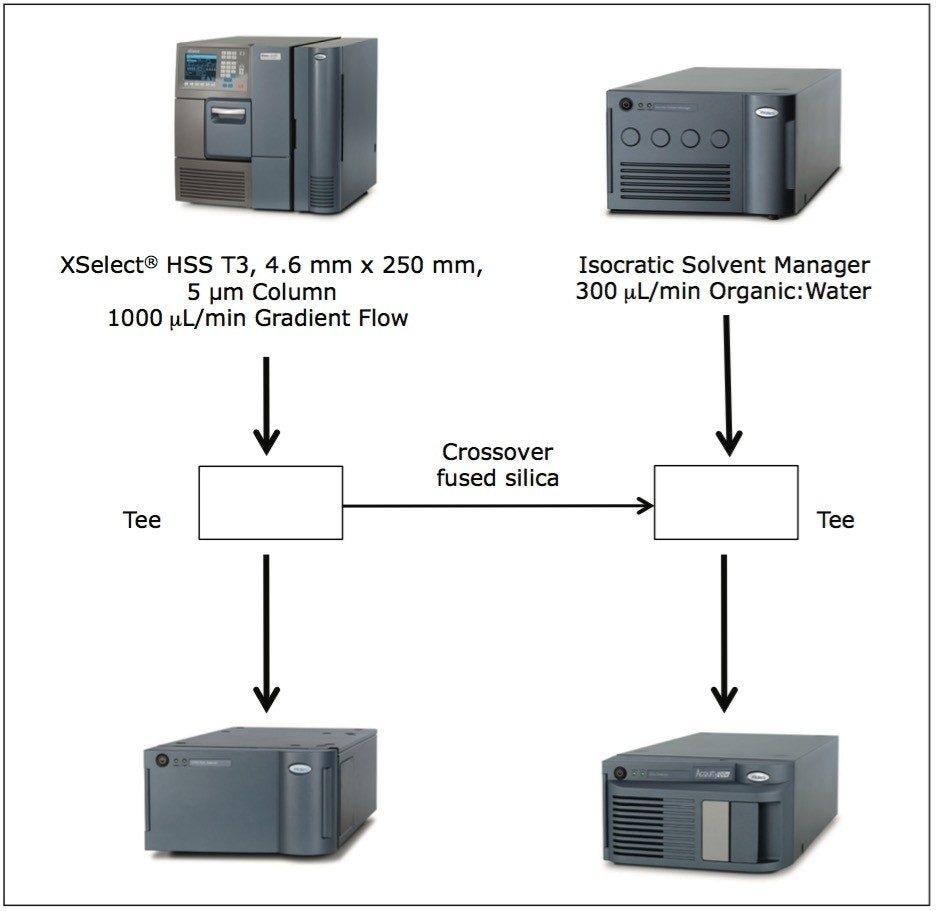
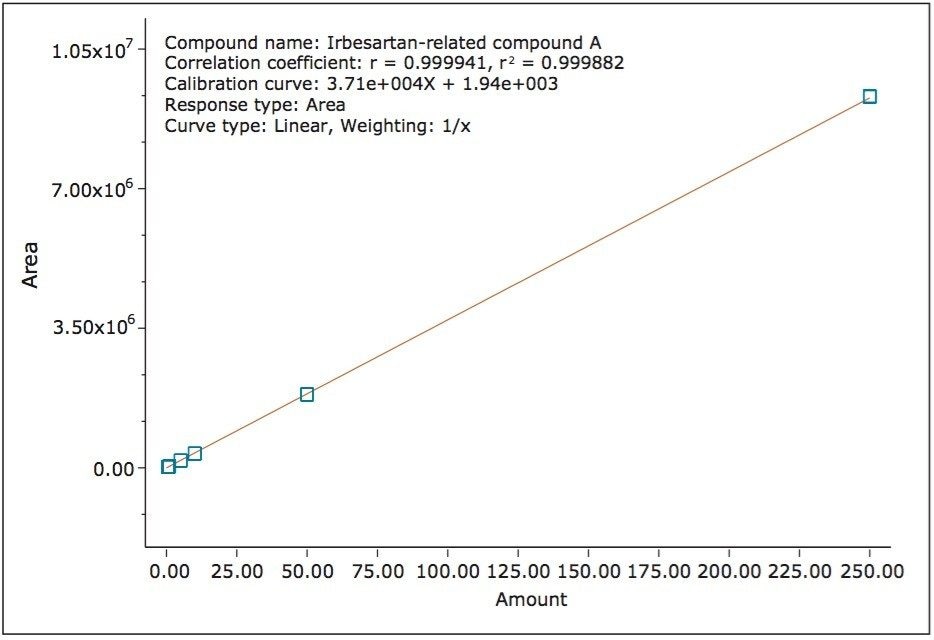
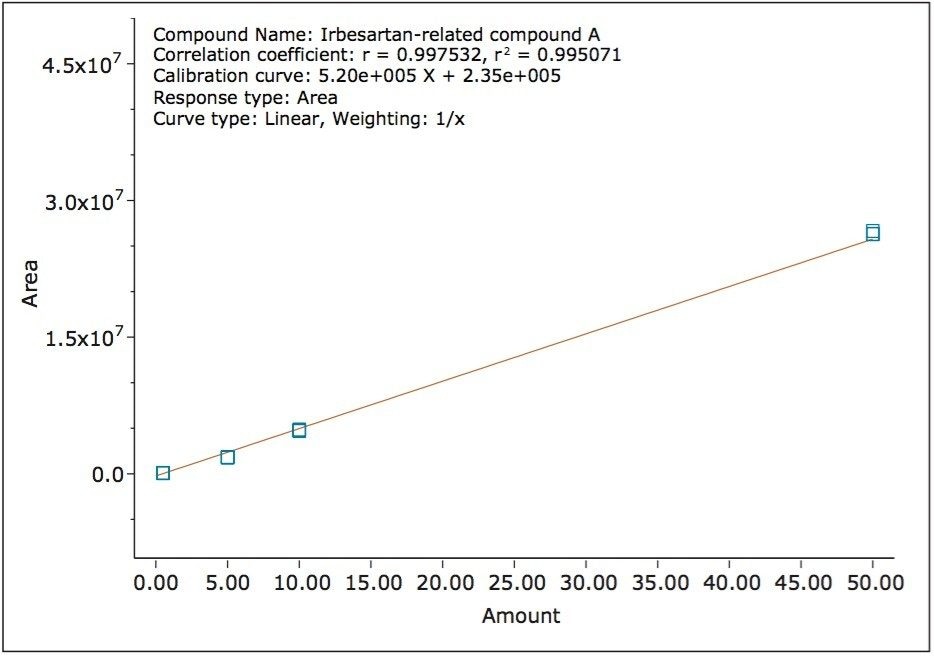
The ISM with restrictor module was used to divide the flow from the column between the 2998 PDA Detector and the ACQUITY QDa Detector. With this split and dilute configuration (Figure 1), the dynamic linear ranges of the two detectors were aligned. Linearity was evaluated using triplicate injections prepared at six levels on the PDA detector and five levels on the mass detector. The resulting calibration was linear across the range of concentrations, 0.0005 mg/mL to 0.250 mg/mL with a correlation coefficient (r2) of 0.9998 on the PDA detector and 0.0005 mg/mL to 0.050 mg/mL with a correlation coefficient (r2) of 0.995 on the mass detector, (Figure 5 and 6). The use of split and dilute resulted in achieving equivalent sensitivity for irbesartan related compound A on the PDA and the mass detectors.
720004946, February 2014