This application illustrates the use of a generic LC-MS method to provide high-sensitivity qualitative and quantitative information using high resolution time-of-flight mass spectrometer.
A three-day qualification study shows that the method achieves stringent bioanalytical criteria comparable to tandem quadrupole methods.
Quadrupole time of flight (QTof) mass spectrometers have long been a staple in the DMPK lab for their utility as screening workhorses. Due to key enhancements in detector sensitivity, StepWave Technology’s increase in signal-to-noise and reduction of neutrals, and QuanTof Technology’s improved resolution and dynamic range, the capacity for a quan/qual approach is maturing. This makes the QTof platform appealing as a tool in both discovery and development groups that are looking to provide qualitative and quantitative program information more routinely.
QTofs can provide additional information for compounds (metabolites) for which authentic standards and multiple reaction monitoring (MRM) optimization workflows are not always applicable. The ability to quantify both the parent compounds as well as determine relative levels of metabolites (or accurate concentrations, if authentic standards are available) allows the DMPK scientist to better assess the quality of preclinical candidate molecules moving through the drug pipeline.
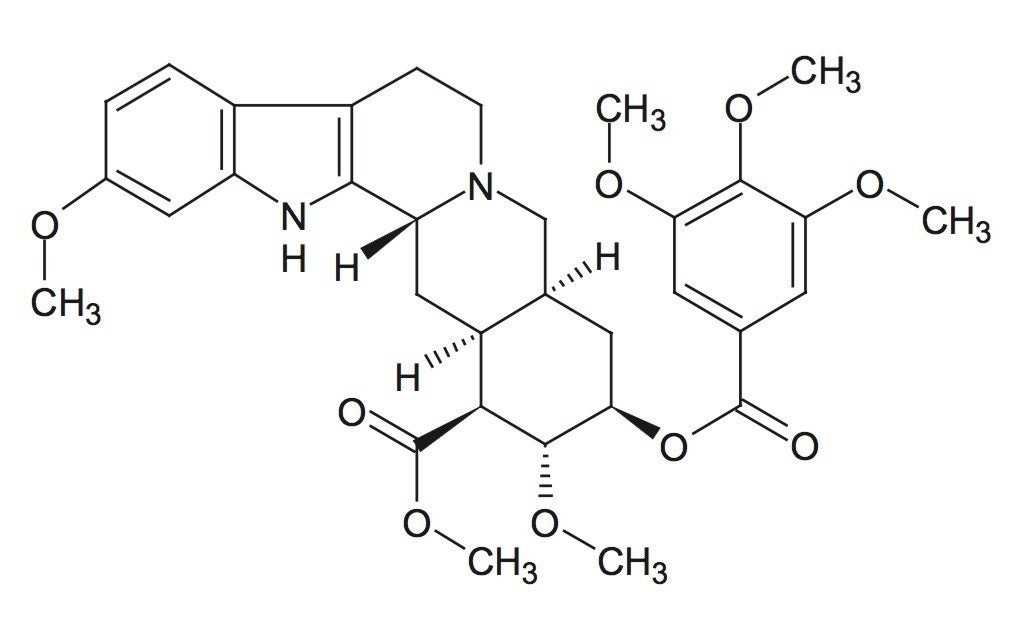
Reserpine is a complex indole alkaloid derived from the plant species Rauwolfia serpentina. Although now considered a second-line therapeutic agent, reserpine has been employed alone or in combination with other drugs (hydralazine, hydrochlorothiazide) for management of hypertension.
A stock solution of reserpine (Sigma Aldrich, St. Louis, MO, USA) was prepared at 100 ng/mL in quenched human plasma, and was serial diluted 1:1 (v:v) with quenched human plasma to a final concentration of 0.003 ng/mL (3 pg/mL). The serial diluted samples were used as the calibration standard solutions. The quality control solutions were prepared by serial dilution of the 100 ng/mL stock solution 1:2 (v:v) with the quenched human plasma to 0.005 ng/mL (5 pg/mL). Quenched human plasma was prepared by adding acetonitrile 3:1 (v:v) to human plasma (source: Bioreclamation) to precipitate protein. After centrifugation at 15,000 rpm at 10 °C for 20 min, the supernatant was transferred and diluted 1:1 (v:v) with H2O prior to injection.
The analysis was performed on an ACQUITY UPLC I-Class System. A 10-μL aliquot of each sample was injected onto an ACQUITY UPLC C18 2.1 x 100 mm, 1.7 μm Column. The column was operated under gradient conditions over nine minutes at a flow rate of 600 μL/min. This is a standard method for analyzing samples containing a large number of compounds, facilitating, for example, simultaneous identification of drug metabolites.
The column effluent was monitored using a Xevo G2-S QTof Mass Spectrometer operated in MS scan mode. Leucine encephalin was employed as a LockSpray reference mass standard, with a scan acquired every 15 seconds. Mass spectrometric data were acquired in continuum mode, mass-corrected, and centroided post-run. Extracted mass chromatograms for protonated reserpine (609.28 ± 0.05 Th) were integrated for quantification.
Data acquisition and processing were performed with MassLynx Software with TargetLynx Application Manager.
The sample set was analyzed on three consecutive days.
This application note reports qualified quantification of reserpine spanning more than three orders of linear magnitude in concentration using QTof mass spectrometry. Using a generic LC-MS method, this high resolution time-of-flight instrument provides accurate and robust mass measurements to permit highly selective quantification based on mass chromatograms.
For DMPK groups also running these methods for metabolite identification, this methodology can serve several purposes, including:
This generic method was designed to support a large number of DMPK discovery assays in a routine and harmonized manner. Using this method, polar compounds such as buspirone elute at 3.94 minutes, while on the other end of the spectrum, clopidogrel elutes at 6.96 minutes.
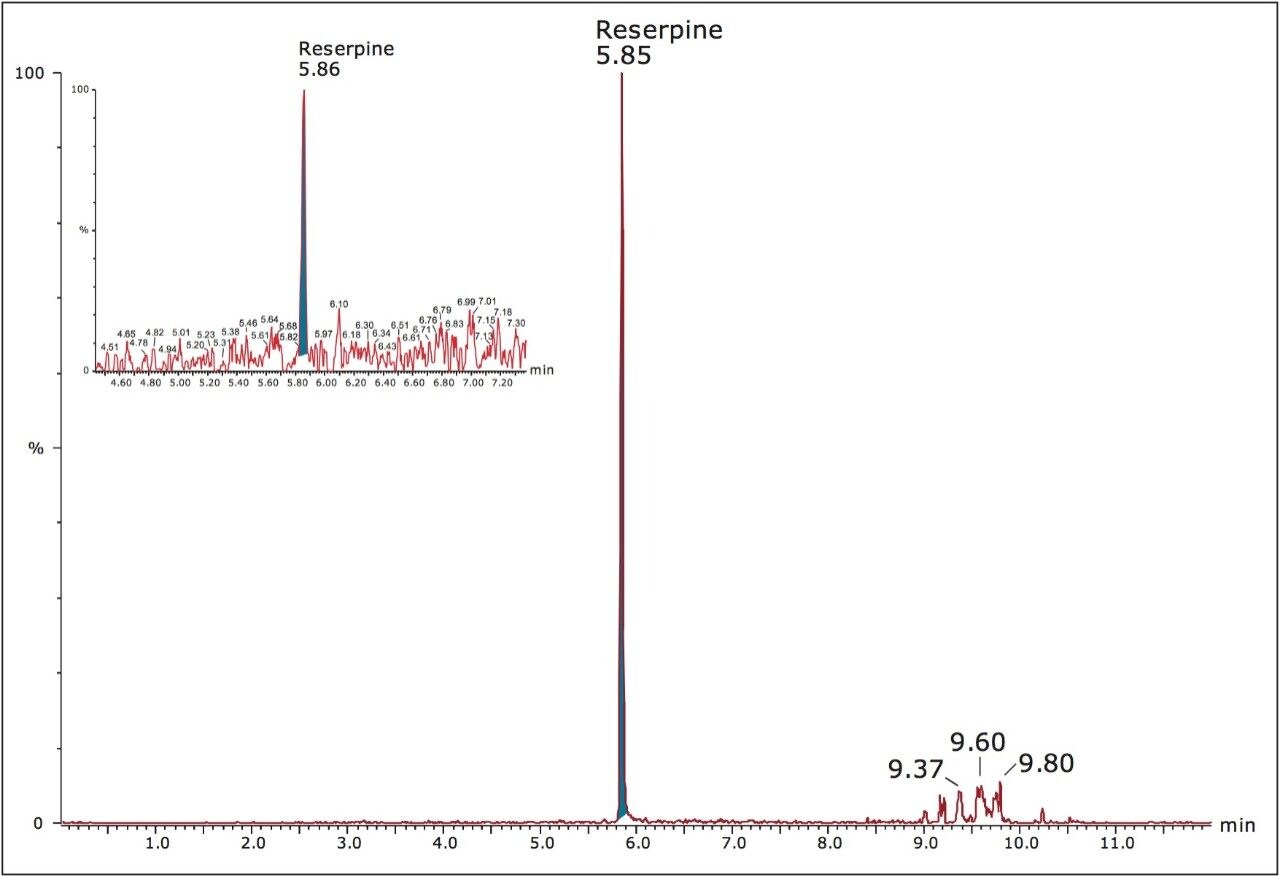
Reserpine was eluted at a retention time of 5.85 minutes and a sharp, symmetrical peak (peak width = 1.3 second at half height, before smoothing prior to integration) was observed in the extracted mass chromatogram, enabling accurate processing and peak integration irrespective of the complex matrix of protein-precipitated human plasma. A typical run is shown in Figure 2. The peak representing the lower limit of quantification (LLOQ) in the insert has a signal-to-noise ratio of 10.3.
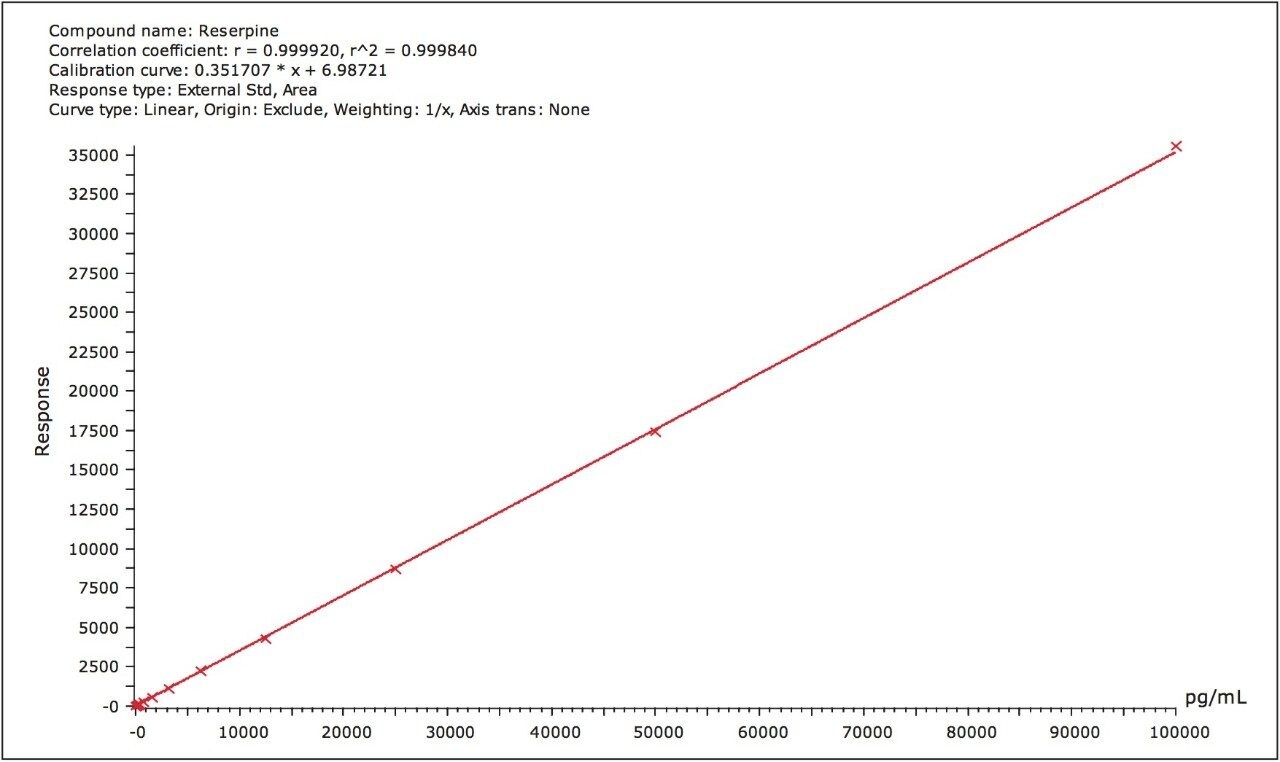
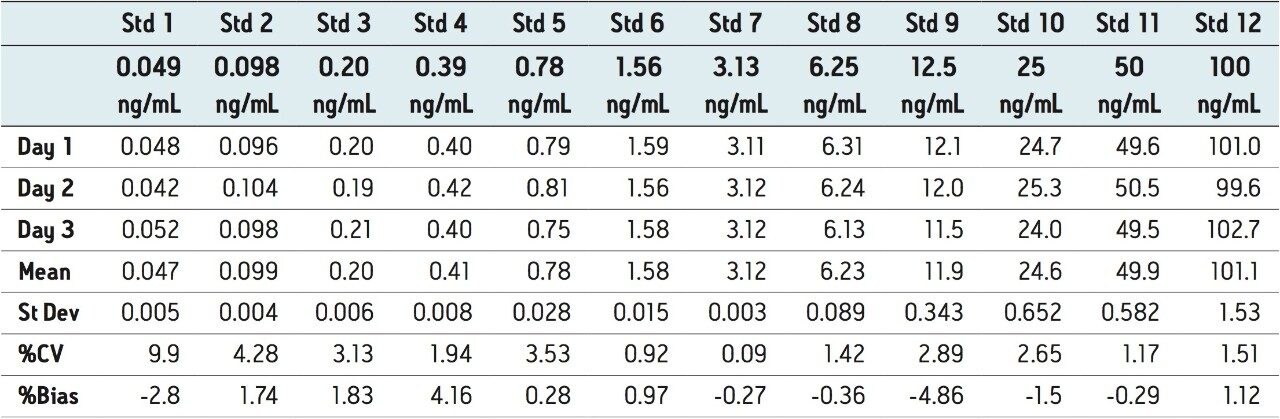
The LLOQ was determined to be 0.049 ng/mL, with a limit of detection of 0.024 ng/mL or less. A typical calibration line for the assay is shown in Figure 3. The correlation coefficients (r2) for each of the three days were 0.9998, 0.9998, and 0.9989 for day 1, day 2, and day 3, respectively, using a 1/x weighted linear regression.
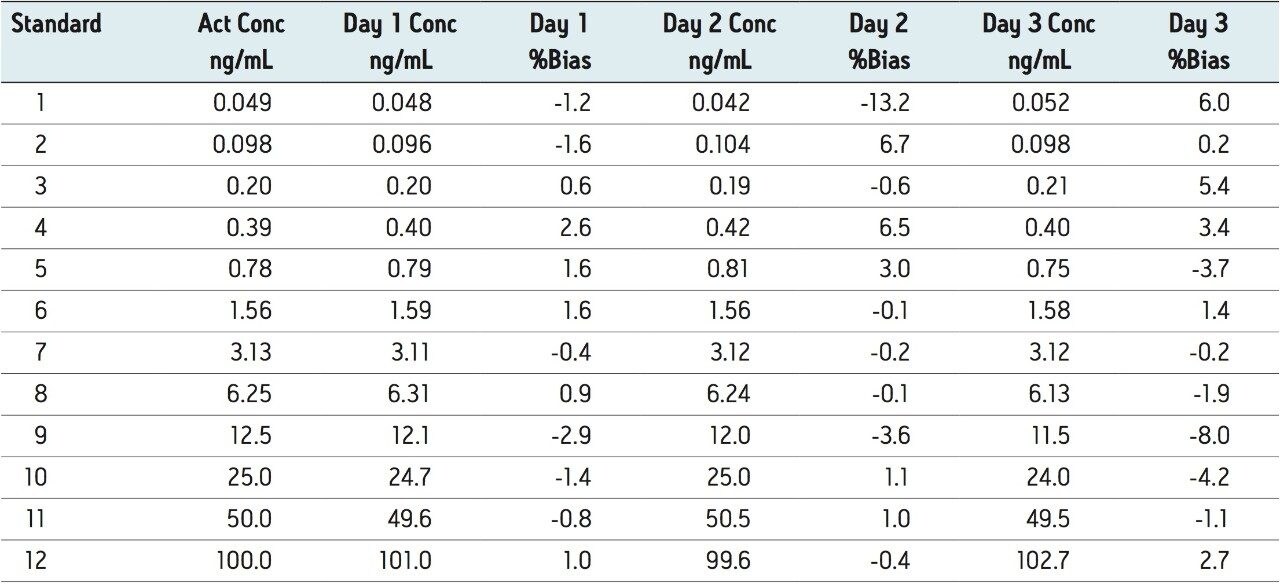
The accuracy and precision qualification data are displayed in Tables 1 and 2. The percent bias values are less than 15% (range of -13.2% to 6.0%) for each individual standard measurement. The bias valued for the interday results range from -4.9% to 4.2%.
These results indicate a linear range of 3.3 orders of magnitude ranging from 0.048 ng/mL to 100 ng/mL.
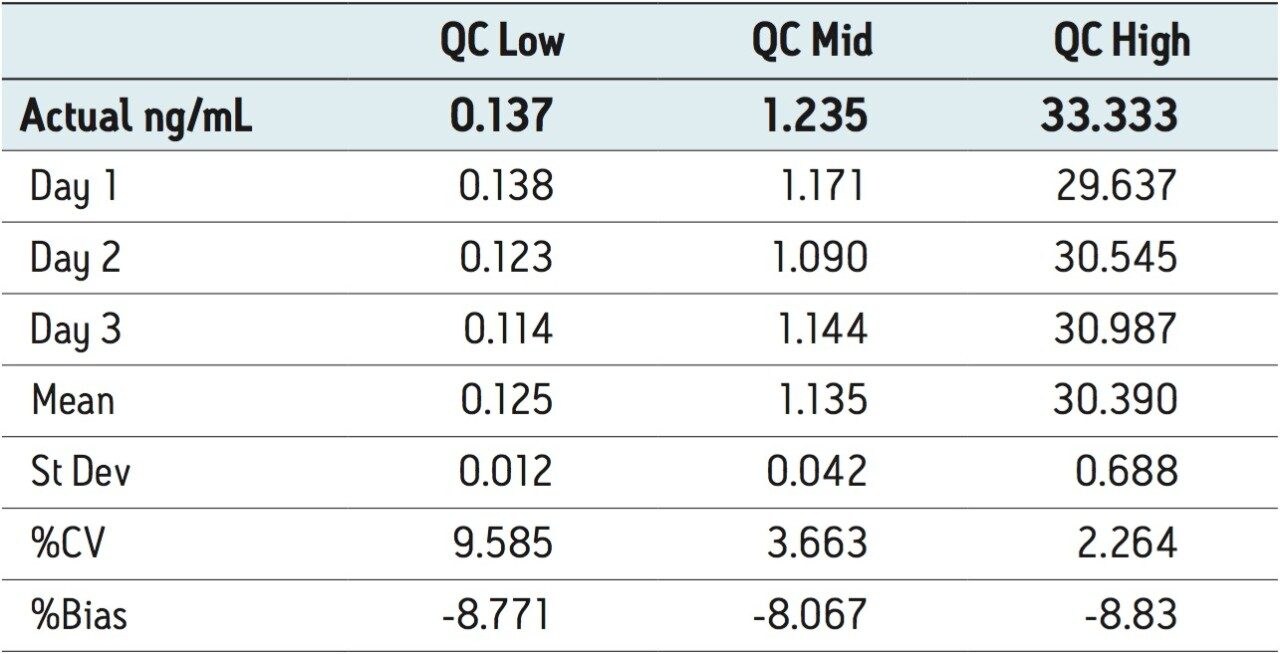
A second series of dilutions were run as QC samples at concentrations near the LLOQ, midrange, and at the upper range of standard curve concentration. The inter-day precision and accuracy results for these samples are displayed in Table 3. The coefficient of variation was 9.6%, 2.3%, and 3.7% at the high and mid concentrations. The bias was -8.8% near the LLOQ and -8.1% to -8.8% at low, mid, and high QC concentrations, respectively. All these values are within typical bioanalysis guidelines used for tandem quadrupole validations.
720004688, May 2013