
For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates efficient analysis of aldosterone in plasma by LC-MS.
Efficient measurement of aldosterone with LC-MS in plasma.
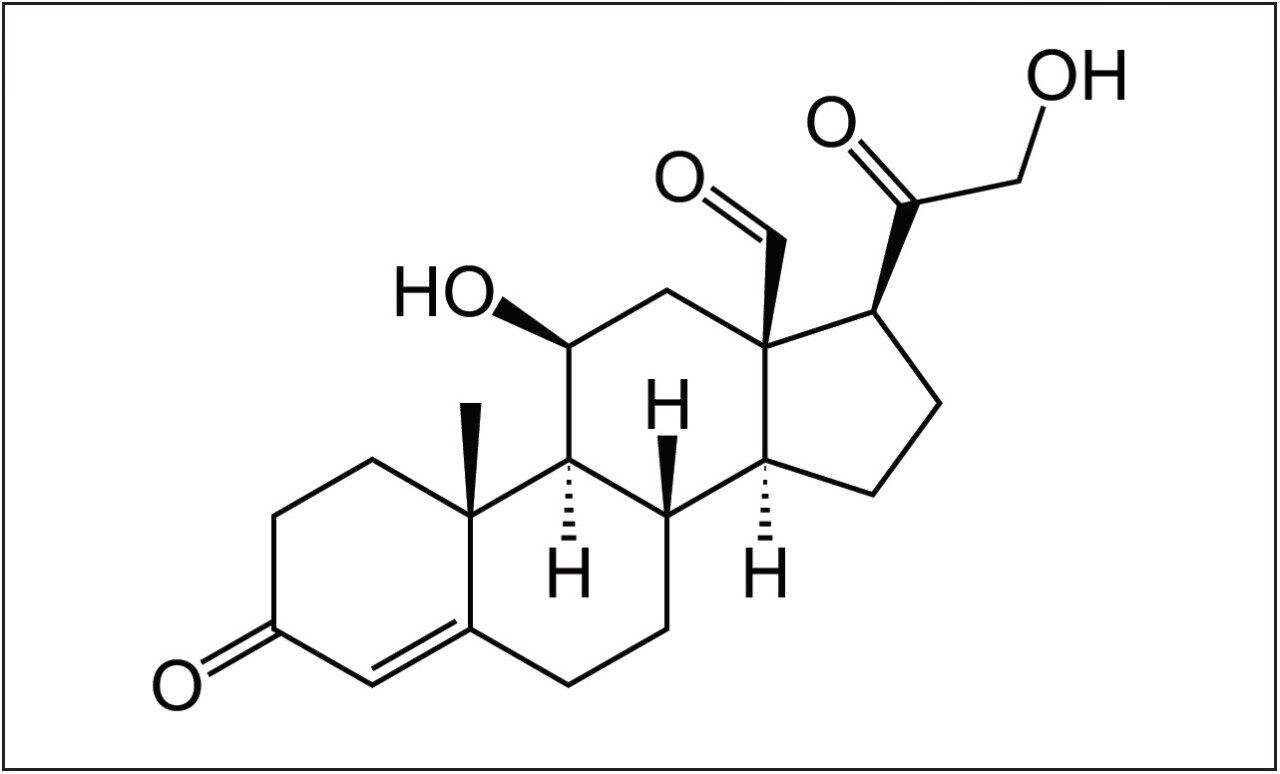
Aldosterone (a steroid hormone made by the adrenal gland) helps regulate sodium and potassium levels in the body to exert hormonal control on blood pressure and the balance of fluids and electrolytes in the blood.
Accurate, low level measurement of steroid hormones, like aldosterone, in plasma has proven to be difficult. Many existing assays suffer from a lack of specificity due to antibody cross reactivity with other closely structurally related steroids, and also tend to be highly variable at lower steroid concentrations. Other methods typically require extensive and time consuming extraction and purification steps, followed by derivitization for analysis. As a result of these issues with older methods, many laboratories are turning to LC-MS to provide the sensitivity and specificity required to measure steroid hormones. Even when utilizing LC-MS, the measurement of aldosterone is challenging as the compound is often found in very low levels in plasma and poorly ionizes in a mass spectrometer, making it particularly difficult to analyze.
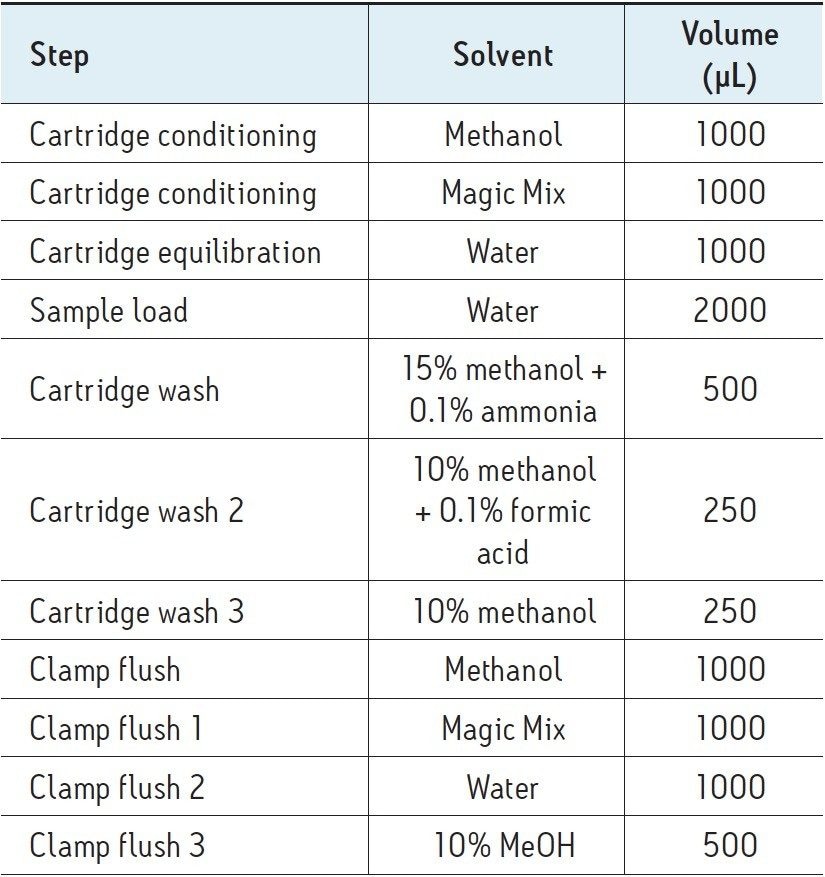
Plasma samples were diluted 4:1 with ZnSO4 to precipitate protein and centrifuged. After protein removal, a 25 μL aliquot of sample was injected into an LC-MS system comprised of a classic ACQUITY UPLC with a Xevo TQ-S Mass Spectrometer equipped with an Online SPE Manager (OSM). SPE was performed as follows:
After SPE, samples were analyzed by LC-MS using the following gradient conditions: 15% water/MeOH + 0.01% ammonia (100 - >10%; 3 min) on an ACQUITY UPLC BEH C18 Column, 130Å, 1.7 μm, 2.1 mm X 50 mm.
In this technology brief, a clinical research method for analyzing aldosterone from plasma was developed. An online SPE system was incorporated into the LC-MS system used for aldosterone measurement. This sample preparation module allows for full integration and automation of sample prep with LC-MS analysis.
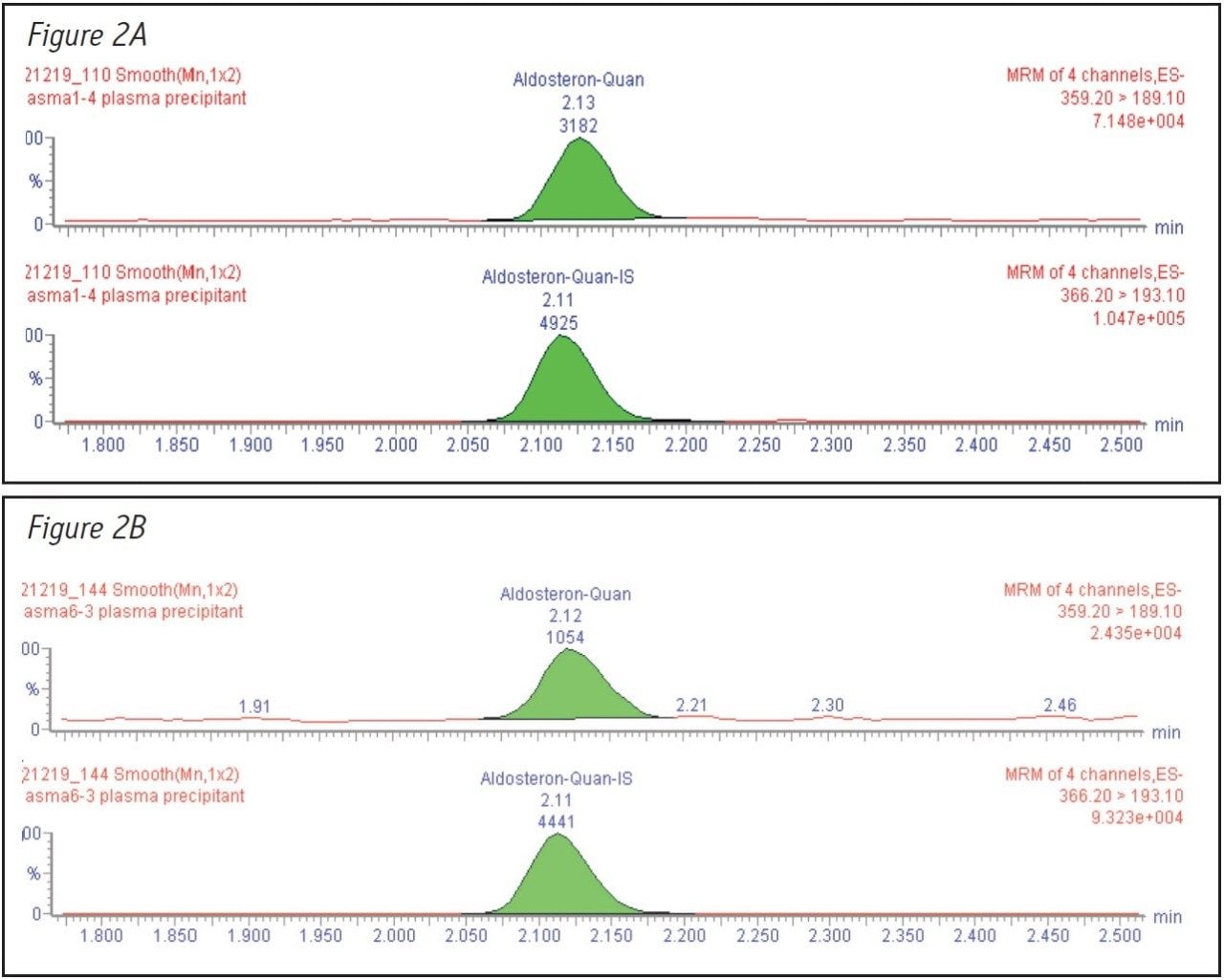
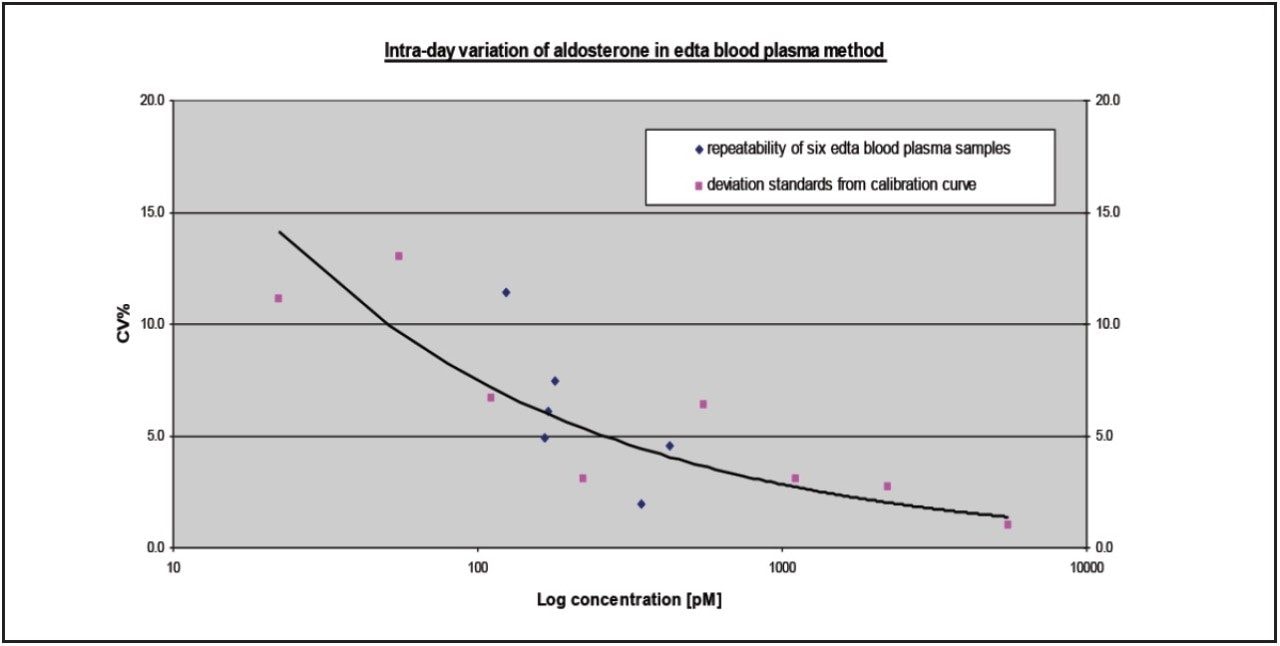
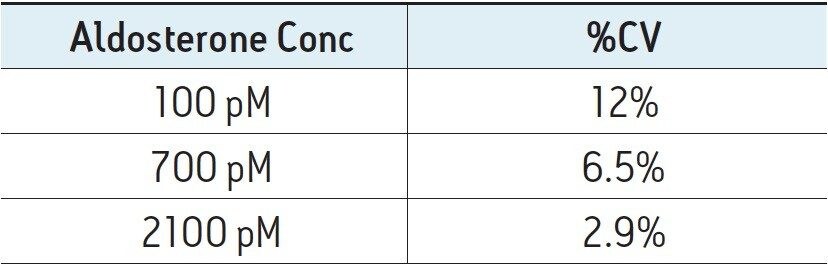
The reproducibility and stability of the method over time was also tested by injecting samples from three different plasma pools containing three different levels of aldosterone (100, 700 and 2000 pM). Each sample was injected 20 times to determine if the peak areas remained constant. Results for these experiments are shown in Figure 4.
In this work, a very efficient method for clinical research has been developed for the measurement of aldosterone from plasma. The method utilizes LC-MS and an online SPE system. This combination of SPE sample preparation coupled with the analytical power of LC-MS provides the necessary sensitivity and productivity required to routinely measure aldosterone in plasma for this important steroid.
The method developed here provides:
720004654, October 2013